
An Unusual Case of Thromboembolic Stroke - Silent Myocardial Infarction from Spontaneous Coronary Artery Dissection
*Corresponding Author(s):
Ying X GueDepartment Of Cardiology, Royal Derby Hospital, Uttoxeter Road, Derby, DE22 3NE, United Kingdom
Tel:+01332 340131,
Email:y.gue@nhs.net
Abstract
An incidental finding on a patient who was followed up after presenting to the hospital with a cerebrovascular event. The angiogram performed showed Spontaneous Coronary Artery Dissection (SCAD) of Left Anterior Descending (LAD) artery. This has resulted in thrombus formation within the left ventricle which led to a thromboembolic stroke. The patient did not have any typical associated conditions for SCAD and it is theorized that heavy smoking and secondary polycythaemia might be contributing factors to the findings.
Keywords
INTRODUCTION
CASE PRESENTATION
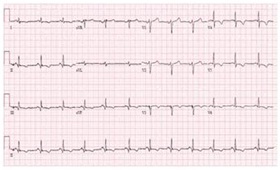 Figure 1: ECG showing inferior and lateral lead changes.
Figure 1: ECG showing inferior and lateral lead changes.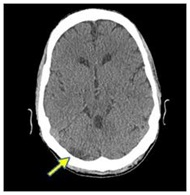 Figure 2: CT brain showing low attenuation in right cerebellar (Arrow).
Figure 2: CT brain showing low attenuation in right cerebellar (Arrow).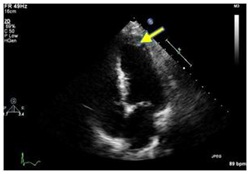 Figure 3: Echocardiogram showing thrombus in apex (Arrow).
Figure 3: Echocardiogram showing thrombus in apex (Arrow).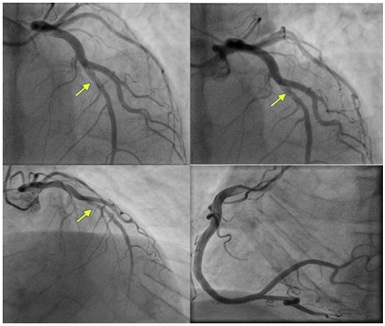 Figure 4: Angiogram showing SCAD in mid LAD (arrows) and normal RCA.
Figure 4: Angiogram showing SCAD in mid LAD (arrows) and normal RCA.Given that the patient was stable, he was treated conservatively with anticoagulation and followed up in outpatient cardiology clinic.
DISCUSSION
Stroke is most commonly caused by carotid artery disease and in the younger population, clinicians should always be mindful towards embolic causes for stroke. This triggered a host of investigations in this gentleman’s case which led to the findings of SCAD as the underlying cause for his symptoms.
It has been thought that SCAD starts out with the development of an intramural haematoma that usually runs in the outer media or between the media and adventitia of the coronary artery, resulting in a false lumen. This lumen expands through blood or clot propagation which then compresses the real lumen, causing ischemia [9].
It is thought that the increased blood volume and cardiac output during pregnancy and labour is one of the contributing factors which makes females in the peri-partum and post-partum period more prone in particular [9]. We understand through studies of patients with polycythaemia vera that blood rheological variables like haematocrit and blood viscosity can promote raised intima media thickness endothelial dysfunction and altered arterial compliance [10-12]. There are also a few case reports on SCAD and polycythaemia vera [13,14]. However, there are no reported cases SCAD with secondary polycythaemia.
Although smoking is one of the well-known and established cardiovascular risk factor by promoting vasomotor dysfunction, thermogenesis and thrombosis it is not typically associated with SCAD. Given the proposed pathogenesis of SCAD, the combination of heavy smoking and secondary polycythaemia could potentially be associated risks [15].
In this case, the differentials included an atherosclerotic plaque event resulting in a similar presentation or a Lotus-Root like lesion causing the angiographic findings. Motreff et al., [16,17] identified 5 angiographic features which could assist clinicians towards the diagnosis of SCAD. The patient had 3 out of the 5 criteria fulfilled i.e., absence of atheroma on other arteries, radiolucent flap generating two lumens and ending of angiographic ambiguity on a side branch. This makes the diagnosis of SCAD more probable. The diagnosis could be confirmed by optical coherence tomography which was not performed in this case.
SUMMARY
COMPETING INTERESTS
REFERENCES
- Vanzetto G, Berger-Coz E, Barone-Rochette G, Chavanon O, Bouvaist H, et al. (2009) Prevalence, therapeutic management and medium-term prognosis of spontaneous coronary artery dissection: results from a database of 11,605 patients. Eur J Cardiothorac Surg 35: 250-254.
- Hampole CV, Philip F, Shafii A, Pettersson G, Anesi GL, et al. (2011) Spontaneous coronary artery dissection in Ehlers-Danlos syndrome. Ann Thorac Surg 92: 1883-1884.
- Tweet MS, Hayes SN, Pitta SR, Simari RD, Lerman A, et al. (2012) Clinical features, management, and prognosis of spontaneous coronary artery dissection. Circulation 126: 579-588.
- El?Sherief K, Rashidian A, Srikanth S (2011) Spontaneous coronary artery dissection after intense weightlifting UCSF Fresno Department of Cardiology. Catheter Cardiovasc Interv 78: 223-227.
- Thompson E, Ferraris S, Gress T (2005) Gender differences and predictors of mortality in spontaneous coronary artery dissection: A review of reported cases, J Invasive Cardiol 17: 59–61.
- Koul AK, Hollander G, Moskovits N, Frankel R, Herrera L, et al. (2001) Coronary artery dissection during pregnancy and the postpartum period: two case reports and review of literature. Catheter Cardiovasc Interv 52: 88-94.
- Appleby CE, Barolet A, Ing D, Ross J, Schwartz L, et al. (2009) Contemporary management of pregnancy-related coronary artery dissection: A single-centre experience and literature review. Exp Clin Cardiol 14: 8-16.
- Mortensen KH, Thuesen L, Kristensen IB, Christiansen EH (2009) Spontaneous coronary artery dissection: a Western Denmark Heart Registry study. Catheter Cardiovasc Interv 74: 710-717.
- Vrints CJM (2010) Spontaneous coronary artery dissection. Heart 96: 801-808.
- Lee AJ, Mowbray PI, Lowe GD, Rumley A, Fowkes FGR, et al. (1998) Blood viscosity and elevated carotid intima-media thickness in men and women: the Edinburgh Artery Study. Circulation 97: 1467-1473.
- Neunteufl T, Heher S, Stefenelli T, Pabinger I, Gisslinger H (2001) Endothelial dysfunction in patients with polycythaemia vera. Br J Haematol 115: 354-359.
- Nemets A, Isakov I, Huerta M, Barshai Y, Oren S, et al. (2006) Evaluation of arterial compliance in polycythemia vera patients: short and long-term influence of phlebotomy. Isr Med Assoc J 8: 845-847.
- Kay IP, Williams MJ (1999) Spontaneous coronary artery dissection: long stenting in a patient with polycythemia vera. Int J Cardiovasc Intervent 2: 191-193.
- Fabregat-Andrés Ó, Berenguer-Jofresa A, Estornell-Erill J (2012) Chronic spontaneous coronary dissection: evaluation by cardiac magnetic resonance. Biomedical Imaging & Intervention J 8: 1-4.
- Ambrose JA, Barua RS (2004) The pathophysiology of cigarette smoking and cardiovascular disease: an update. J Am Coll Cardiol 43: 1731-1737.
- Kadowaki H, Taguchi E, Kotono Y, Suzuyama H, Yoshida M, et al. (2016) A lotus root-like appearance in both the left anterior descending and right coronary arteries. Heart Vessels 31: 124-128.
- Motreff P, Malcles G, Combaret N, Barber-Chamoux N, Bouajila S, et al. (2016) How and when to suspect spontaneous coronary artery dissection? Novel insights from a single-center series on prevalence and angiographic appearance. EuroIntervention.
Citation: Gue YX, Chitkara K (2017) An Unusual Case of Thromboembolic Stroke - Silent Myocardial Infarction from Spontaneous Coronary Artery Dissection. J Angiol Vasc Surg 2: 006.
Copyright: © 2017 Ying X Gue, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

