
Endovascular Treatment with the pCONus Device: Initial Experience in a Single UK Centre
*Corresponding Author(s):
Fozia SaeedDepartment Of Neurosurgery, Leeds General Infirmary, Great George Street, Leeds LS1 3EX, United Kingdom
Tel:+44 7701344725,
Email:fozia.s@doctors.org.uk
Abstract
Background: Wide-necked intracranial bifurcation aneurysms are complex and difficult to treat. Several novel devices have been developed for treatment using endovascular approaches. We present the results of our initial experience of using the pCONus device in our centre.
Methods: This retrospective study includes 7 patients who underwent endovascular treatment for wide-necked (≥4mm) intracranial aneurysms. 4 aneurysms were unruptured and 3 were acutely ruptured at the time of treatment. Initial and follow up radiological and clinical outcomes are presented.
Results: A total of 6 aneurysms were successfully treated with the pCONus device whilst one aneurysm resulted in barrel stent assisted coiling due to the inability to deploy pCONus. There were two complications with the development of a thrombus during the procedure-resolution was observed following further administration of anti-coagulation. At six month follow-up, 83% of the aneurysms showed complete occlusion or stable sub-total occlusion on radiological imaging and all patients had a mRS score of 0.
Conclusion: In this initial case series, pCONus stent assisted coiling for wide-necked bifurcation aneurysms is safe and provides good clinical and radiological outcomes. Long term follow-up is required to determine efficacy of this device.
Keywords
INTRODUCTION
Despite these developments the treatment of intracranial Wide-Necked Bifurcation Aneurysms (WNBA) remains complex, primarily due to the technical difficulties of preserving both bifurcation branches. More recently a novel device the pCONus bifurcation aneurysm implant (Phenox, Germany) has been designed for WNBA. The pCONus device is an advancement of the waffle cone technique which involves inserting a self-expanding stent into the parent vessel whilst its distal tip is implanted into the neck of the aneurysm to hold the coils in place [8].
The aim of our study is to report our initial experience of using the novel pCONus device for EVT of intracranial aneurysms in a series of consecutive cases.
Methods
Device description: The pCONus device (Phenox GmbH, Bochum, Germany) is a laser cut stent-like structure specifically designed for WNBA (Figure 1). The distal end (‘crown’) comprises of 4 petals and a nylon net which is deployed into the aneurysm to support the coil mass and prevent it from protruding into the parent vessel. The petals are available in 6 diameters ranging from 5mm to 15mm. The shaft remains within the parent vessel and has favourable qualities that lower thrombogenicity. The diameter of the shaft is 4mm and is available in two lengths (20mm, 25mm). The device is detached through an electrolytic detachment process but may be completely recovered. Another advantage of this device includes a proximal marker and four radio-opaque markers on each of the petals, allowing for accurate positioning of the device.
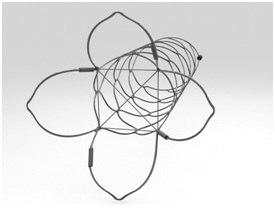 Figure 1: Illustration of the pCONus bifurcation aneurysm implant. Image used from Phenox (Germany), with permission.
Figure 1: Illustration of the pCONus bifurcation aneurysm implant. Image used from Phenox (Germany), with permission.Antiplatelet/Anticoagulation regime: Before treatment, all patients received antiplatelet therapy as shown in table 1. During the procedure, patients received a bolus of 5000 units Heparin followed by a further dose if required to keep the ACT at or above twice baseline. All elective patients were given a bolus dose of 300 mg of Aspirin and Clopidogrel one week before the procedure. Post-procedure patients were continued on Aspirin 75mg indefinitely and Clopidogrel 75mg for 6 weeks.
| PatientNo. | Presentation | AneurysmsTreated | Location | Parent Vessel Diameter (mm) | Aneurysm Size (mm) | Aneurysm neck (mm) | Device | Anti-plateletBT | Anti-platelet AT | |
| 1 | Unruptured | 1 | R MCA | 2-3 | 6 x 6.8 x 10.3 | 4.6 | pCONus | asp+clop | clop 6/52 ?asp indefinitely | |
| 2 | Ruptured/Recurrence | 1 | L AcomA | 2-3 | 5 x 5.4 x 5.7 | 6.1 | pCONus | asp+clop | cbp 6/12 asp indefinitely | |
| 3 | Unruptured | 1 | R MCA | 2-3 | 11.8 x 12.3 x 12.6 | 12.7 | pCONus | asp+clop | cbp 6/52 asp indefinitely | |
| 4 | Unruptured | 2 | L MCA + Lpericallosal | 2-3 | 7.3 x 8 x 9.22 | 8.6 | barrel | asp+clop | cbp 6/52 asp indefinitely | |
| 5 | Ruptured | 2 | L MCA + L PcomA | 2-3 | 4.4 x 5.7 x 6.4 | 4.4 | pCONus | dcagrelor ISOmg+asp Ig | ticagralor 180mg 6/52 asp indefinitely | |
| 6 | Ruptured | 1 | Basilar tip | 3 | 7.5 x 9.4 x 9.6 | 6.3 | pCONus | prasugrel 60mg+isp Ig | prasugrel 10mg 6/12 asp indefinitely | |
| 7 | Ruptured | 1 | L AcomA | 2-3 | 6.3 x 9.3 x 8.1 | 6.0 | pCONus | asp lg | cbp 6/52 asp indefinitely |
R - Right; L - Left; MCA - Middle Cerebral Artery; AcomA - Anterior communication Artery; PcomA - Posterior communicating Artery; BT - Before Treatment; AT - After Treatment; asp - aspirin; clop – Clopidogrel
Different regimes were used for patients with acutely ruptured aneurysms (Table 1). One patient developed a clot in the parent vessel and received 10mg Abciximab during the procedure which successfully dissolved the clot. Post-procedure patients were commenced on Aspirin 75mg once daily indefinitely and either Clopidogrel 75mg, Prasugrel 10mg or Ticegrelor 180mg daily ranging from either 6 weeks to 6 months (Table 1).
Data collection: Ethical approval was sought but not required for anonymised retrospective data collection. Data collected included patient’s age and gender, clinical presentation, aneurysm characteristics (location, size, dome to neck ratio) and technical and clinical complications. Post procedure angiographic results were classified based on the Raymond classification scale: complete occlusion, residual neck or residual aneurysm [9]. The patients were clinically examined pre and post-procedure for any neurological symptoms/signs. The clinical course of each patient was recorded and 6 month outcomes were graded using the modified Rankin Scale (mRS).
Patient follow-up: Patients were reviewed in follow-up clinic at 6 weeks and underwent 6 month follow-up imaging with Magnetic Resonance angiography (MRI 3T, Siemens) with and without gadolinium contrast agent.
RESULTS
Aneurysm characteristics
As a result of the device characteristics only aneurysms with a neck of ≥4mm were treated with the pCONus device. The aneurysms ranged in size with the average dome size as 8mm (range 4.4-12.6mm) and average neck size as 7mm (range 4.4-12.7mm). Aneurysm characteristics are summarized in table 1.
Complications
Post-procedure, one patient returned with a small retroperitoneal haematoma adjacent to the puncture site. This was managed conservatively and resolved spontaneously. Another patient developed transient expressive dysphasia which spontaneously resolved within a few hours. There were no deaths.
In one case, there was procedural difficulty placing the pCONus device across the MCA aneurysm bifurcation branches and the device was therefore retrieved and replaced with a barrel stent (Medtronic Inc.).
Immediate outcome
Follow-up
| Patient No. | Intra-opComplications | ProcedureComplications | Postangiography Result | 6 Month Follow-up(MRA) | mRS |
| 1 | nil | nil | small remnant | complete occlusion | 0 |
| 2 | nil | nil | small remnant | complete occlusion | 0 |
| 3 | nil | nil | small remnant | neck remnant | 0 |
| 4 | nil | unable to deploy pCONus -barrel stent used | complete | neck recurrence | 0 |
| 5 | Thrombus | nil | complete | complete occlusion | 0 |
| 6 | nil | nil | small remnant | neck recurrence | 0 |
| 7 | Thrombus | nil | small remnant | small remnant | 0 |
Illustrative cases
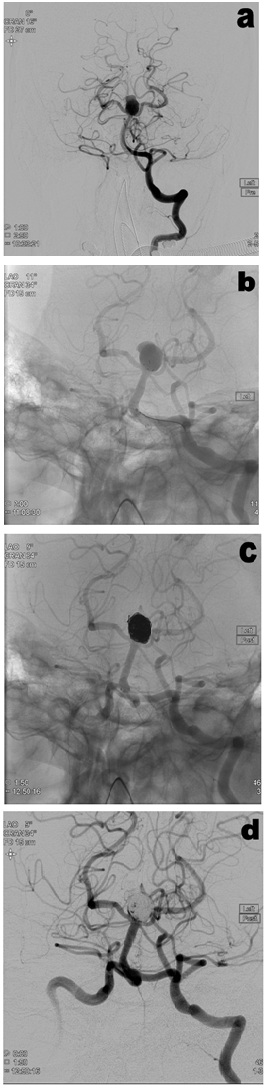 Figure 2: Case of a patient with an acutely ruptured basilar tip aneurysm treated with pCONus (A before treatment, B pCONus device in-situ before coil deployment, C Coils deployed and D after treatment). At 6 months follow-up, MRA showed a small neck recurrence which is undergoing surveillance.
Figure 2: Case of a patient with an acutely ruptured basilar tip aneurysm treated with pCONus (A before treatment, B pCONus device in-situ before coil deployment, C Coils deployed and D after treatment). At 6 months follow-up, MRA showed a small neck recurrence which is undergoing surveillance.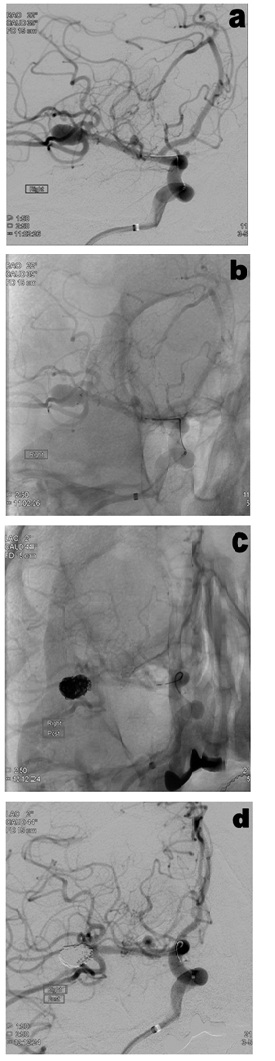 Figure 3: Case of a patient with an unruptured right MCA aneurysm treated with pCONus (A before treatment, B pCONus device in-situ before coil deployment, C Coils deployed and D after treatment). At 6 months follow-up, MRA showed complete occlusion of the aneurysm.
Figure 3: Case of a patient with an unruptured right MCA aneurysm treated with pCONus (A before treatment, B pCONus device in-situ before coil deployment, C Coils deployed and D after treatment). At 6 months follow-up, MRA showed complete occlusion of the aneurysm.DISCUSSION
In order to overcome some of the difficulties in treating WNBA, various techniques have been developed including balloon remodelling and stent assisted techniques. Both CLARITY and ANTENA studies compared conventional coil embolisation with balloon remodelling techniques in ruptured and unruptured aneurysms respectively and found no significant increase in morbidity or mortality [9,10]. However, aside from adding complexity to the procedure, limitations of this technique also include prolonged procedure duration and fluoroscopic time [10].
Stent assisted techniques to treat WNBA include X, Y and T stenting, linear/horizontal stenting and the waffle cone technique [5,8,11,12]. The waffle cone technique involves placing the proximal end of the stent in the parent vessel and the distal end into the aneurysm sac but it is associated with higher rates of recanalisation [13,14]. X, Y, T, and linear stenting involve a complex delivery technique as they all require the use of more than one stent [11,15,16]. More recent advances include the use of a Woven EndoBridge (WEB) device which is a braided nitinol mesh that functions as an intrasaccular flow disruptor [17-20]. One of our patients with an AComA aneurysm treated with pCONus had a recurrence following utilisation of the WEB device previously.
Another recently CE-approved device which can be used to treat wide-necked bifurcation aneurysms is the PulseRider (Pulsar Vascular, San Jose, California, USA). There are limited reports of experience with this device [21,22]. Very recently, Gory et al., 2016 published results of PulseRider stent assisted coiling in 15 patients with unruptured bifurcation aneurysms. This international series found low complication rates but were unable to provide data on efficacy of the device [21].
The pCONus device is also a novel stent which has been specifically designed to assist coiling of wide-necked intracranial bifurcation aneurysms. There are several studies that have assessed the effectiveness of the pCONus device in unruptured aneurysms but only one currently published study on its use in ruptured aneurysms [23-26]. Aguilar-Perez et al., 2016, has shown encouraging results in 21 patients with ruptured aneurysms that were treated with the pCONus device [23]. The authors reported the device as safe and effective for use in ruptured aneurysms but based on short term follow-up results alone.
In our study, EVT with pCONus was successfully performed in six of the seven patients. In one patient the device had to be retrieved due to the inability to place it safely across the middle cerebral artery. The complexity of this case was a result of asymmetrical branches making it difficult to place the pCONus device at the aneurysmal neck in a satisfactory position. This could possibly represent a limitation of this device. Immediate angiogram results showed complete occlusion in one patient and 5 small aneurysm and neck remnants. In line with other studies, the overall performance of the device has so far produced satisfactory clinical results [23-27]. There were no significant clinical complications associated with the use of the device and all patients were discharged with no permanent neurological deficits.
However a significant limitation of stent-assisted coiling with the pCONus device is the need for anticoagulation and anti-platelet therapy peri- and post-procedure. More often, anti-platelets are continued lifelong as the risk of thrombo-embolic complications is reported as ranging between 7-15% [28-30]. This indeed can be problematic, particularly in acute cases of ruptured intracranial aneurysms where risk of bleeding-related complications are higher following SAH sequelae such as hydrocephalus needing surgical intervention or vasospasm resulting in cerebral ischaemia predisposing to haemorrhagic transformation [31-35]. This controversial issue remains a challenge for neurointerventionalists. Bodily et al., 2011, reported an overall procedure-related risk of up to 13% in ruptured cases versus 6-7% in unruptured cases [30,31,36]. Though it is unknown if this difference in risk is truly in relation to the inherent differences or due to confounding factors such as technical differences.
More commonly encountered longer term problems in EVT include recannalisation and recurrence rates of 20% and 10% respectively [37]. Follow-up result of our patients with the pCONus device at 6 months demonstrated 83% with stable complete or sub-total occlusions. The patient who underwent barrel-assisted embolisation was found to have a recurrence of the MCA aneurysm and is awaiting further treatment. In addition, the lack of consistency in pre-procedure antiplatelet regime may have contributed to the intra-procedural complication of thrombus encountered in two of the cases.
The limitations of our study are typical for a single-centre retrospective analysis. We also experienced difficulty with imaging as the stem of the pCONus device appears to cause an artefact on MRA and therefore catheter angiogram may be required for follow-up imaging. Further limitations of this study include the small patient cohort included and unavailable long term follow-up results. However as this is a novel EVT, preliminary results are, in our opinion, vital to engage in the safety and efficacy of this device. We also propose the need for large multi-centre studies to accurately determine the risks of stent-assisted EVT in ruptured aneurysms before final conclusions are made.
CONCLUSION
ACKNOWLEDGEMENT
FS: Acquisition and analysis of the data, interpretation of data, manuscript preparation
FS, AJPG, TP: Drafting the work and critical revision
FS, AJPG, TP: Manuscript preparation and critical revision of the manuscript
ETHICS APPROVAL
REFERENCES
- Molyneux A, Kerr R, Stratton I, Sandercock P, Clarke M, et al. (2002) International Subarachnoid Aneurysm Trial (ISAT) of neurosurgical clipping versus endovascular coiling in 2143 patients with ruptured intracranial aneurysms: a randomised trial. Lancet 360: 1267-1274.
- McDougall CG, Spetzler RF, Zabramski JM, Partovi S, Hills NK, et al. (2012) The Barrow Ruptured Aneurysm Trial. J Neurosurg 116: 135-144.
- Cognard C, Pierot L, Anxionnat R, Ricolfi F; Clarity Study Group (2011) Results of embolization used as the first treatment choice in a consecutive nonselected population of ruptured aneurysms: clinical results of the Clarity GDC study. Neurosurgery 69: 837-841.
- Moret J, Cognard C, Weill A, Castaings L, Rey A (1997) The "Remodelling Technique" in the Treatment of Wide Neck Intracranial Aneurysms. Angiographic Results and Clinical Follow-up in 56 Cases. Interv Neuroradiol 3: 21-35.
- Bartolini B, Blanc R, Pistocchi S, Redjem H, Piotin M (2014) "Y" and "X" stent-assisted coiling of complex and wide-neck intracranial bifurcation aneurysms. AJNR Am J Neuroradiol 35: 2153-2158.
- Fischer S, Vajda Z, Aguilar Perez M, Schmid E, Hopf N, et al. (2012) Pipeline Embolization Device (PED) for neurovascular reconstruction: initial experience in the treatment of 101 intracranial aneurysms and dissections. Neuroradiology 54: 369-382.
- Lubicz B, Mine B, Collignon L, Brisbois D, Duckwiler G, et al. (2013) WEB device for endovascular treatment of wide-neck bifurcation aneurysms. AJNR Am J Neuroradiol 34: 1209-1214.
- Sychra V, Klisch J, Werner M, Dettenborn C, Petrovitch A, et al. (2011) Waffle-cone technique with Solitaire™ AB remodeling device: endovascular treatment of highly selected complex cerebral aneurysms. Neuroradiology 53: 961-972.
- Raymond J, Guilbert F, Weill A, Georganos SA, Juravsky L, et al. (2003) Long-term angiographic recurrences after selective endovascular treatment of aneurysms with detachable coils. Stroke 34: 1398-1403.
- Pierot L, Cognard C, Anxionnat R, Ricolfi F; CLARITY Investigators (2011) Remodeling technique for endovascular treatment of ruptured intracranial aneurysms had a higher rate of adequate postoperative occlusion than did conventional coil embolization with comparable safety. Radiology 258: 546-553.
- Pierot L, Spelle L, Leclerc X, Cognard C, Bonafé A, et al. (2009) Endovascular treatment of unruptured intracranial aneurysms: comparison of safety of remodeling technique and standard treatment with coils. Radiology 251: 846-855.
- Thorell WE, Chow MM, Woo HH, Masaryk TJ, Rasmussen PA (2005) Y-configured dual intracranial stent-assisted coil embolization for the treatment of wide-necked basilar tip aneurysms. Neurosurgery 56: 1035-1040.
- Horowitz M, Levy E, Sauvageau E, Genevro J, Guterman LR, et al. (2006) Intra/extra-aneurysmal stent placement for management of complex and wide-necked- bifurcation aneurysms: eight cases using the waffle cone technique. Neurosurgery 58: 258-262.
- Lee S-M, Kim Y-J, Ko JH (2015) The effectiveness of the waffle-cone technique in treating complex intracranial aneurysms. Interv Neuroradiol 21: 470-478.
- Lubicz B (2011) Linear stent-assisted coiling: another way to treat very wide-necked intracranial aneurysms. Neuroradiology 53: 457-459.
- Chow MM, Woo HH, Masaryk TJ, Rasmussen PA (2004) A novel endovascular treatment of a wide-necked basilar apex aneurysm by using a Y-configuration, double-stent technique. AJNR Am J Neuroradiol 25: 509-512.
- Lubicz B, Klisch J, Gauvrit JY, Szikora I, Leonardi M, et al. (2014) WEB-DL endovascular treatment of wide-neck bifurcation aneurysms: short- and midterm results in a European study. AJNR Am J Neuroradiol 35: 432-438.
- Anil G, Goddard AJ, Ross SM, Deniz K, Patankar T (2016) WEB in Partially Thrombosed Intracranial Aneurysms: A Word of Caution. AJNR Am J Neuroradiol 37: 892-896.
- Caroff J, Mihalea C, Klisch J, Strasilla C, Berlis A, et al. (2015) Single-Layer WEBs: Intrasaccular Flow Disrupters for Aneurysm Treatment—Feasibility Results from a European Study. AJNR Am J Neuroradiol 36: 1942-1946.
- Liebig T, Kabbasch C, Strasilla C, Berlis A, Weber W, et al. (2015) Intrasaccular Flow Disruption in Acutely Ruptured Aneurysms: A Multicenter Retrospective Review of the Use of the WEB. AJNR Am J Neuroradiol 36: 1721-1727.
- Gory B, Spiotta AM, Mangiafico S, Consoli A, Biondi A, et al. (2016) PulseRider Stent-Assisted Coiling of Wide-Neck Bifurcation Aneurysms: Periprocedural Results in an International Series. AJNR Am J Neuroradiol 37: 130-135.
- Mukherjee S, Chandran A, Gopinathan A, Putharan M, Goddard T, et al. (2016) PulseRider-assisted treatment of wide-necked intracranial bifurcation aneurysms: safety and feasibility study. J Neurosurg 30: 1-8.
- Aguilar Pérez M, Bhogal P, Martinez Moreno R, Wendl C, Bäzner H, et al. (2016) Use of the pCONus as an adjunct to coil embolization of acutely ruptured aneurysms. J NeuroIntervent Surg 9: 1-8.
- Lubicz B, Morais R, Alghamdi F, Mine B, Collignon L, et al. (2016) The pCONus device for the endovascular treatment of wide neck bifurcation aneurysms. J Neurointerv Surg 8.
- Fischer S, Weber A, Titschert A, Brenke C, Kowoll A, et al. (2016) Single-center experience in the endovascular treatment of wide-necked intracranial aneurysms with a bridging intra-/extra-aneurysm implant (pCONus). J Neurointerv Surg 8: 1186-1191.
- Gory B, Aguilar-Pérez M, Pomero E, Turjman F, Weber W, et al. (2015) pCONus device for the endovascular treatment of wide-neck middle cerebral artery aneurysms. AJNR Am J Neuroradiol 36: 1735-1740.
- Ulfert C, Pfaff J, Schönenberger S, Bösel J, Herweh C, et al. (2016) The pCONus Device in Treatment of Wide-necked Aneurysms : Technical and Midterm Clinical and Angiographic Results. Clin Neuroradiol.
- Benitez RP, Silva MT, Klem J, Veznedaroglu E, Rosenwasser RH (2004) Endovascular occlusion of wide-necked aneurysms with a new intracranial microstent (Neuroform) and detachable coils. Neurosurger 54: 1359-1367.
- Fiorella D, Albuquerque FC, Han P, McDougall CG (2004) Preliminary experience using the Neuroform stent for the treatment of cerebral aneurysms. Neurosurgery 54: 6-16.
- Piotin M, Blanc R, Spelle L, Mounayer C, Piantino R, et al. (2010) Stent-assisted coiling of intracranial aneurysms: clinical and angiographic results in 216 consecutive aneurysms. Stroke 41: 110-115.
- Bodily KD, Cloft HJ, Lanzino G, Fiorella DJ, White PM, et al. (2011) Stent-assisted coiling in acutely ruptured intracranial aneurysms: a qualitative, systematic review of the literature. AJNR Am J Neuroradiol 32: 1232-1236.
- dos Santos Souza MP, Agid R, Willinsky RA, Cusimano M, Montanera W, et al. (2005) Microstent-assisted coiling for wide-necked intracranial aneurysms. Can J Neurol Sci 32: 71-81.
- Katsaridis V, Papagiannaki C, Violaris C (2006) Embolization of acutely ruptured and unruptured wide-necked cerebral aneurysms using the neuroform2 stent without pretreatment with antiplatelets: a single center experience. AJNR Am J Neuroradiol 27: 1123-1128.
- Mocco J, Snyder KV, Albuquerque FC, Bendok BR, Alan SB, et al. (2009) Treatment of intracranial aneurysms with the Enterprise stent: a multicenter registry. J Neurosurg 110: 35-39.
- Biondi A, Janardhan V, Katz JM, Salvaggio K, Riina HA, et al. (2007) Neuroform stent-assisted coil embolization of wide-neck intracranial aneurysms: strategies in stent deployment and midterm follow-up. Neurosurgery 61: 460-468.
- Liang G, Gao X, Li Z, Wei X, Xue H (2010) Neuroform stent-assisted coiling of intracranial aneurysms: a 5 year single-center experience and follow-up. Neurol Res 32: 721-727.
- Ferns SP, Sprengers ME, van Rooij WJ, Rinkel GJ, van Rijn JC, et al. (2009) Coiling of intracranial aneurysms: a systematic review on initial occlusion and reopening and retreatment rates. Stroke 40: 523-529.
Citation: Saeed F, Patankar T, Goddard AJP (2017) Endovascular Treatment with the pCONus Device: Initial Experience in a Single UK Centre. J Angiol Vasc Surg 2: 008.
Copyright: © 2017 Fozia Saeed, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

