
A Rare Case of Hepatic Portal and Mesenteric Venous Gas in a Patient with Gastric Dilatation Secondary to Gastroparesis and Gastric Bezoar
*Corresponding Author(s):
Sriharsha TalluriGeisinger Commonwealth School Of Medicine, York Hospital, Pennsylvania, United States
Tel:+1 7176597710,
Email:stalluri@tcmc.edu
Abstract
Hepatic Portal Venous Gas (HPVG) is a rare and grave radiological finding, usually associated with a poor prognosis. It is caused by a number of acute abdominal conditions and is most commonly associated with ischemic bowel. There are a number of factors that play a role in HPVG, including changes in intestinal wall distension of bowel and infection. Endoscopic procedures have also been implicated in the formation of HPVG. Plain radiography, ultrasound and CT scan can detect HPVG. Surgery is usually required in majority of cases and mortality rates are high, especially those associated with intestinal necrosis. We report the first case of HPVG secondary to gastric distension secondary to gastroparesis and gastric bezoar in a patient with cirrhosis and Hepatitis C. Our patient did not require surgery and was managed with small frequent meals and metoclopramide, resulting in a favourable outcome. Our case highlights the awareness of conservative management of HPVG if there is no evidence of bowel ischemia or necrosis.
Keywords
INTRODUCTION
Hepatic Portal Venous Gas (HPVG) is a rare disease entity that is usually indicative of grave prognosis. It is reported to be associated with many pathological conditions [1]. We report the first finding of extensive hepatic portal, mesenteric and gastric venous gas in a patient with gastric distension secondary to gastric bezoar as a result of underlying gastroparesis. It is pivotal to determine the underlying etiology as this is the primary determinant of mortality in HPVG. This disease can be treated conservatively if there is no evidence of intestinal ischemia or necrosis.
CASE PRESENTATION
A 41-year-old male with a history of alcohol abuse and hepatitis C with cirrhosis (Child-Pugh Class A and MELD Score 8) presented with a 3-day history of chest pain. He was also feeling depressed and admitted to using cocaine. His other past medical history included type 2 diabetes mellitus. His vital signs and physical examination was normal. His labs revealed the following: haemoglobin of 7.7 gm/dL, white cell count of 8.9K/mcL, mcv of 63fL, platelet count of 58K/mcL, BUN of 12 mg/dL, creatinine of 0.80 mg/dL, serum sodium of 129mmol/L, potassium 4.6mmol/L, chloride 97mmol/L, bicarbonate 19mmol/L, glucose was elevated at 426mmol/L, haemoglobin A1c of 9.8mmol/mol total protein 7.8 gm/dL, albumin 3.7 gm/dL, alkaline phosphatase 234IU/L, AST 49IU/L, ALT 48IU/L, total bilirubin 1.4 gm/dL, amylase, lipase, EKG and cardiac enzymes were within normal limits. Urine toxicology screen was positive for cocaine. In summary, our patient had portal hypertensive gastropathy, hypersplenism and poorly controlled diabetes mellitus, resulting in the aforementioned lab abnormalities. During his hospital stay he developed as cites with increased abdominal swelling, fever and abdominal pain. However, ascitic fluid analysis was negative for spontaneous bacterial peritonitis. A CT scan of the abdomen and pelvis with oral and intravenous contrast revealed air within the liver tracking along the portal vein and extending into the superior mesenteric vein consistent with portal venous gas (Figure 1). A severely distended stomach was identified with air within the venous system of the stomach. There was no evidence of ischemic bowel and therefore a laparotomy was held off. An upper gastrointestinal endoscopy showed features of portal hypertensive gastropathy and a severely dilated stomach with large gastric bezoar due to solid food debris. The gastric bezoar occupied nearly two-thirds of the stomach, which was fragmented and suctioned off during the procedure, resulting in a decrease in bezoar size. A gastric emptying study with Tc99m Sulphur colloid mixed in a scrambled egg meal revealed significant gastroparesis. The patient was managed conservatively with small frequent meals and metoclopramide with improvement in symptoms. A repeat CT scan of the abdomen showed complete resolution of the portal venous gas and patient was discharged home.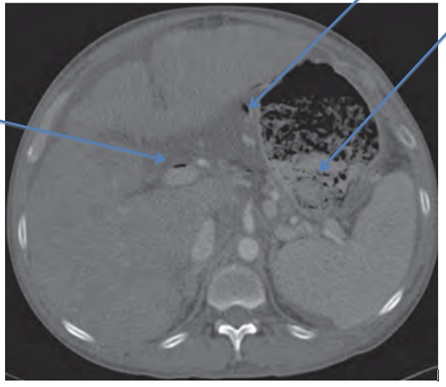 Figure 1: Gastric bezoar, portal and gastric venous gas.
Figure 1: Gastric bezoar, portal and gastric venous gas.
DISCUSSION
Since first described by Wolf and Evans in infants [2], HPVG has been reported in adults with increasing frequency. It is associated with a wide range of pathologies including mesenteric ischemia, post-procedural complications, Crohn’s disease, intra- abdominal abscess and bowel obstruction. Less common pathologies include colon cancer, gastric ulceration, acute pancreatitis, portal pyelophlebitis, infectious causes and sigmoid diverticulitis [1]. In a review of 64 cases by Liebman et al., HPVG was associated with necrotic bowel (72%), ulcerative colitis (8%), intra-abdominal abscess (6%), small bowel obstruction (3%) and gastric ulcer (3%) [3].
A significant mortality rate is associated with HPVG. A review of 182 cases by Kinoshita et al., showed a mortality rate of 39%. There was also a significantly higher mortality rate at 75% in patients who had underlying bowel necrosis. There was no mortality associated with inflammatory bowel disease, intra peritoneal tumour, cholangitis, pancreatitis and complications post endoscopic prociedures [4]. Recently, mortality rate in HPVG has been declining due to early diagnosis of these conditions via ultrasound, CT scan and by finding the underlying cause. HPVG must be distinguished from pneumobilia, as these two pathologies are often confused. HPVG extends within 2 centimeters of the liver capsule, while pneumobilia does not. HPVG appears peripherally in the liver, while air in the biliary tree appears centrally [5]. Gastric dilatation has been reported to be a cause for HPVG [6,7]. The pathogenesis of HPVG is not well understood. It has been proposed that mucosal integrity, intraluminal pressure and intestinal microflora contribute to the pathogenesis [8]. Portal venous gas might be related to an increase in intraluminal pressure, which forces intraluminal gas through a damaged or undamaged bowel wall, where it is absorbed into the portal circulation. This scenario has been reported in cases of ileus or gastric dilatation or after blunt abdominal trauma, endoscopy or barium enema examination [9]. Portal venous gas has a high carbon dioxide content [10]. It is expected to only briefly exist in the vascular system before being absorbed or removed by flow unless the production of gas persists [8].
Our patient had poorly controlled diabetes mellitus, a major underlying contributor to gastroparesis. This gastroparesis caused further complication of gastric bezoar, which resulted in severe gastric dilatation. We believe that the resultant increase in intra-luminal pressure was responsible for the portal venous gas noted on imaging. The patient responded well to conservative management. McElvanna et al., described one case acute pancreatitis and another of acute diverticulitis causing HPVG, which were treated conservatively. A third case of abdominal haematoma was managed surgically, highlighting the importance of clinical, laboratory and radiological findings to determine the appropriate course of treatment [11]. It is therefore prudent to arrive at the exact etiology of HPVG, so that appropriate surgical or conservative treatments can be applied [11].
In conclusion, though HPVG is usually associated with a grave prognosis, mortality is usually determined by the underlying etiology and may be conservatively managed if there is no evidence of bowel ischemia or necrosis. A physician’s treatment plan should be determined by clinical signs and symptomatology and not be based solely on imaging studies. Early diagnosis and appropriate treatment strategy have reduced the mortality rates of this rare disease.
REFERENCES
- Iannitti DA, Gregg SC, Mayo-Smith WW, Tomolonis RJ, Cioffi WG, et al. (2003) Portal venous gas detected by computed tomography: is surgery imperative?. Dig Surg 20: 306-315.
- Wolfe JN, Evans WA (1955) Gas in the portal veins of liver in infants; a roentgenographic demonstration with postmortem anatomical correlation. Am J Roentgenol Radium TherNucl Med 74: 486-488.
- Liebman PR, Patten MT, Manny J, Benfield JR, Hechtman HB (1978) Hepatic--portal venous gas in adults: Etiology, pathophysiology and clinical significance. Ann Surg 187: 281-287.
- . Kinoshita H, Shinozaki M, Tanimura H, Umemoto Y, Sakaguchi S, et al. (2001) Clinical features and management of hepatic portal venous gas: four case reports and cumulative review of the literature. Arch Surg 136: 1410-1414.
- Kesarwani V, Ghelani DR, Reece G (2009) Hepatic portal venous gas: A case report and review of literature. Indian J Crit Care Med 13: 99-102.
- Radin DR, Rosen RS, Halls JM (1987) Acute gastric dilatation: a rare cause of portal venous gas. Am J Roentgenol 148: 279-280.
- Benson MD (1985) Adult survival with intrahepatic portal venous gas secondary to acute gastric dilatation, with a review of portal venous gas. Clin Radiol 36: 441-443.
- St Peter SD, Abbas MA, Kelly KA (2003) The spectrum of pneumatosis intestinalis. Arch Surg 138: 68-75.
- Chevallier P, Peten E, Souci J, Chau Y, Padovani B, et al. (2002). Detection of portal venous gas on sonography, but not on CT. Eur Radiol 12: 1175-1178.
- Wiot JF, Felson B (1961) Gas in the Portal Venous System. Am J Roentgenol Radiat Ther Nuclear Med 86: 920-929.
- McElvanna K, Campbell A, Diamond T (2012) Hepatic portal venous gas - three non-fatal cases and review of the literature. Ulster Med J 81: 74-78.
Citation: Talluri S, Thimmapuram J, Srour J (2017) A Rare Case of Hepatic Portal and Mesenteric Venous Gas in a Patient with Gastric Dilatation Secondary to Gastroparesis and Gastric Bezoar. J Clin Stud Med Case Rep 4: 044.
Copyright: © 2017 Sriharsha Talluri, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

