
Posterior Reversible Encephalopathy Syndrome: A Rare Complication of Peripartum Pre-eclampsia or Eclampsia- A Case report
*Corresponding Author(s):
Ram Sundar TwayanaDepartment Of Cardiology, Dhulikhel Hospital, Dhulikhel, Nepal
Tel:+977 9841492720,
Email:dr.ramsundar@gmail.com
Abstract
Background: Posterior Reversible Encephalopathy Syndrome (PRES) is a clinic-neuroradiological entity characterized by a headache, blurred vision, altered mental sensorium, vomiting and seizure associated with white matter changes in neurological imaging. The lesions in PRES are thought to be due to vasogenic oedema, predominantly in the posterior cerebral hemispheres, and are reversible with appropriate management.
Case report: We report a 28 years primipara lady who had high Blood Pressure (BP) during intrapartum period followed by a generalized tonic-clonic seizure. Her CT scan revealed bilateral parietal and occipital lobe hypodense areas. We managed her administering antihypertensive and antiepileptic drugs along with mechanical ventilator support. Her clinical condition improved without any neurological deficit and discharged.
Conclusion: PRES is an uncommon complication of peripartum pre-eclampsia or eclampsia. An early evaluation for the diagnosis and prompt management has the excellent prognosis without neurological damage.
Keywords
INTRODUCTION
Posterior Reversible Encephalopathy Syndrome (PRES) is a clinical-neuroradiological entity, initially described in 1996 by Hinchey and co-workers, as reversible posterior leukoencephalopathy syndrome. PRES is characterized by clinical symptoms of a headache, visual perception defects, altered mental status, and seizures in conjunction with radiological findings of posterior cerebral white matter edema or hypodensities [1,2].
Hypertensive encephalopathy is the most common cause of PRES, in which the rate of increase in blood pressure is an important role than the absolute blood pressure level [3]. Other causes of PRES in absence of hypertension includes immunosuppressant drugs, such as cyclosporine, tacrolimus, various antineoplastic agents, severe hypercalcaemia, thrombocytopenic syndromes, Henoch-Schonlein purpura, haemolytic uraemic syndrome, amyloid angiopathy, Systemic Lupus Erythematosus (SLE), renal failure, post-transplantation, infection, sepsis (gram positive organisms predominate) and shock [4-6]. The exact mechanism for PRES is still unknown, regardless the various triggering factor, it involves the development of vasogenic oedema in the affected area of the brain [7,8]. Hypertension with failed autoregulation and hyperperfusion remains a popular consideration for the developing brain edema [9,10]. Alternatively, endothelial dysfunction or injury, hypoperfusion, and vasoconstriction may lead to altered integrity of the blood-brain barrier [11].
CASE REPORT
A 28 years primipara lady, who had normal vaginal delivery attended at the general hospital, presented to Emergency Department with the history of abrupt rise in arterial blood pressure (200/110mmHg) during intrapartum period (third stage of labour) followed by multiple episodes of generalized tonic-clonic seizure without regaining consciousness. She delivered a baby weighing 2.6 kg with normal Apgar score and discharged. There is no history of fever, altered sensorium, headache, blurred vision prior to hospital presentation. Her antenatal period was uneventful without features of pre-eclampsia and eclampsia. She had no major illness in the past and during pregnancy. She was referred from general hospital for control of blood pressure and generalized tonic clonic seizure in our hospital after administering loading dose of magnesium sulphate.
At emergency department, her BP was 220/110 mmHg and on Status epilepticus, with poor GCS (7/15). Hence loading dose of sodium valproate was given which failed to control seizure and in addition loading dose of levetiracetam was administered. Concomitantly, endotracheal intubation was done under propofol and shifted to intensive care unit followed by support of mechanical ventilation. Her general examinations were unremarkable. Neurological examination revealed altered mental status (under sedation) with a bilateral normal sized pupil of sluggish reactivity. Fundoscopic examination showed bilateral papilloedema, deep tendon reflexes and plantar signs were bilaterally normal. There was no significant finding on systemic examination. Her routine blood investigations including a complete hemogram, coagulation profile, liver function test, renal function test, blood glucose, serum electrolytes, chest X-ray, ECG were normal. CT scan brain showed hypo dense lesions on gray matter mostly on bilateral parieto-occipital areas and diffuse poorly defined sulci and gyrus (Figures 1 & 2). She was administered parental labetalol and furosemide for initial control of arterial blood pressure and later given oral amlodipine as maintenance. She was extubated on 3rd day of ICU stay after all parameters became normal and transfer to ward.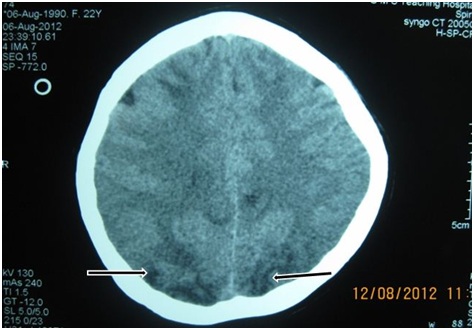 Figure 1: CT brain showing hypo dense lesions on gray matter of bilateral parieto-occipital areas.
Figure 1: CT brain showing hypo dense lesions on gray matter of bilateral parieto-occipital areas.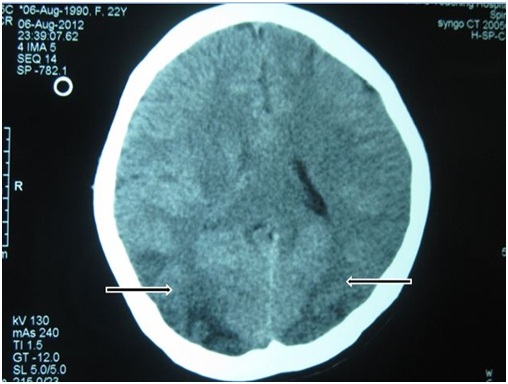 Figure 2: CT showing poorly defined sulci and gyri on bilateral parieto-occipital areas.
Figure 2: CT showing poorly defined sulci and gyri on bilateral parieto-occipital areas.
She was diagnosed as postpartum eclampsia complicated by PRES and treated with the maintenance dose of antiepileptics (levetiracetam), antihypertensive drugs and mechanical ventilator support. Her condition improved without any neurological sequelae and discharged home. We followed up her after 14 days of discharge and found normal without neurological deficit evidence by repeated CT scan screening. We advised her to continue antihypertensive and antiepileptic drugs for at least 6 months with further follow up at interval of 6 weeks time but we lost her follow up presumably she is doing well.
DISCUSSION
PRES is one of the rare complications of pre-eclampsia, eclampsia or hypertension during pregnancy that occurs either in an antepartum, intrapartum or postpartum period. With recent advancement in neuro-imaging facilities, it can be now detected early and treated promptly to prevent complications like intracranial haemorrhage or ischemia. Proper diagnosis requires careful attention to the clinical and radiographic presentation. Pathogenesis of PRES has been suggested that there is a temporary failure of autoregulatory capabilities of the cerebral vessels, leading to hyperperfusion, a breakdown of the blood-brain barrier and consequent vasogenic oedema [12]. Vasogenic oedema results from the combination of Hypertension (HTN) and endothelial injury [13]. Since cases of PRES without HTN have been reported, endothelial dysfunction may represent a common pathway in the pathogenesis of PRES, regardless of a etiology. There is an apparent predisposition for oedema to occur in the posterior CNS areas, particularly in the occipitoparietal areas [1]. This is thought to be secondary to the partial lack of sympathetic innervations of the vasculature that emerge from the basilar artery [3]; however, other sites of the brain and cerebellum may be affected even when occipitoparietal areas are not involved [8,14].
In PRES associated with pregnancy-induced HTN, it is thought that pregnancy itself predisposes the brain to oedema formation, particularly in late pregnancy. Pregnancy has direct effects on perivascular innervations by mechanisms not yet elucidated, particularly in pial vessels, in which nerves hypertrophy is produced [15]. This suggests that neurotransmitters may have a role in hypertensive intolerance that results in the genesis of PRES.
The characteristics features on MRI, T1-weighted images show hypointense and hyperintense areas on T2-weighted images, hyperintensity on FLAIR images in the parietooccipital and posterior frontal cortical and subcortical white matter, less commonly brain stem, basal ganglia, and cerebellum are involved. On CT scan, abnormalities are seen as symmetric bilateral areas of low attenuation of white matter [16]. Atypical imaging appearances include contrast enhancement, haemorrhage and restricted diffusion on MRI [17]. Follow-up MRI after appropriate therapy for the cause of PRES shows resolution of the lesions, unless the condition progresses to infarction or haemorrhage. PRES lesions involving the occipital lobe spare the calcarine and paramedian occipital lobe. This feature, along with the predominant involvement of white matter helps to distinguish this syndrome from bilateral infarctions of the posterior cerebral artery.
Treatment of acute PRES is the removal of triggering factors and responsible aetiology. Complete recovery occurs within two weeks of treatment and late sequelae are rare. The differential diagnosis of PRES includes other neurological events including ischemic stroke.
Various study showed different results, a study of 76 patients by Alexander M McKinney et al., showed that the incidence of regions involvement was parieto-occipital 98.7%, temporal 68.4%, thalamus 30.3%, cerebellum 34.2%, brain stem 18.4% and basal ganglia 11.8%. The incidence of less common manifestations was enhancement (37.7%), restricted diffusion (17.3%), haemorrhage (17.1%) and a newly described unilateral variant (2.6%) [16].
WS Bartynski et al., described vasogenic oedema in parietal or occipital regions (98%), frontal lobes (68%), inferior temporal lobes (40%), cerebellar hemispheres (30%), basal ganglia (14%), brainstem (13%), deep white matter (18%) and splenium (10%). Three major patterns of PRES were noted: the holohemispheric watershed (23%), superior frontal sulcal (27%), parieto-occipital (22%), with additional common partial or asymmetric expression of these primary PRES patterns (28%) [16].
CONCLUSION
PRES is one of the rare complications of peripartum preeclamsia or eclamsia. Clinical and neuro-radiological imaging study supports the diagnosis. This syndrome is reversible and early attention will enable physicians to avoid a delay in diagnosis and institute treatment promptly to avoid permanent neurological sequelae. Control of seizures and blood pressure, with proper counselling should be the goal for management of such cases.
REFERENCES
- Hinchey J, Chaves C, Appignani B, Breen J, Pao L, et al. (1996) Reversible posterior leucoencephalopathy syndrome. N England J Med 334: 494- 500.
- Finocchi V, Bozzao A, Bonamini M, Ferrante M, Romano A, et al. (2005) et al. Magnetic resonance imaging in posterior reversible encephalopathy syndrome: report of three cases and review of literature. Arch Gynecol Obstet 271: 79-85.
- Vaughan CJ, Delanty N (2000) Hypertensive emergencies. Lancet 356: 411-417.
- Bartynski WS, Boardman JF, Zeigler ZR, Shadduck RK, Lister J (2006) Posterior reversible encephalopathy syndrome in infection, sepsis, and shock. AJNR Am J Neuroradiol 27: 2179-2190.
- Ishikura K, Ikeda M, Hamasaki Y, Hataya H, Nishimura G, et al. (2008) Nephrotic state as a risk factor for developing posterior reversible encephalopathy syndrome in paediatric patients with nephrotic syndrome. Nephrol Dial Transplant 23: 2531-2536.
- El Karoui K, Le Quintrec M, Dekeyser E, Servais A, Hummel A, et al. (2007) Posterior reversible encephalopathy syndrome in systemic lupus erythematosus. Nephrol Dial Transplant 23: 757-763.
- Alehan F, Erol I, Agildere AM, Ozcay F, Baskin E, et al. (2007) Posterior leukoencephalopathy syndrome in children and adolescents. J Child Neurol 22: 406-413.
- McKinney AM, Short J, Truwit CL, McKinney ZJ, Kozak OS, et al. (2007) Posterior reversible encephalopathy syndrome: incidence of atypical regions of involvement and imaging findings. AJR Am J Roentgenol 189: 904-912.
- Schwartz RB, Bravo SM, Klufas RA, Hsu L, Barnes PD, et al. (1995) Cyclosporine neurotoxicity and its relationship to hypertensive encephalopathy: CT and MR findings in 16 cases. AJR Am J Roentgenol 165: 627-631.
- Trommer BL, Homer D, Mikhael MA (1988) Cerebral vasospasm and eclampsia. Stroke 19: 326-329.
- Eichler FS, Wang P, Wityk RJ, Beauchamp NJ Jr, Barker PB (2002) Diffuse metabolic abnormalities in reversible posterior leukoencephalopathy syndrome. AJNR Am J Neuroradiol 23: 833-837.
- Lamy C, Oppenheim C, Meder JF, Mas JL (2004) Neuroimaging in posterior reversible encephalopathy syndrome. J Neuroimaging 14: 89-96.
- Cipolla MJ (2007) Cerebrovascular function in pregnancy and eclampsia. Hypertension 50: 14-24.
- Doi Y, Kimura F, Fujiyama T, Fujimura C, Nishina T, et al. (2006) Hypertensive brainstem encephalopathy without parieto-occipital lesion--two case reports. Neurol Med Chir (Tokyo) 46: 75-79.
- Aukes AM, Vitullo L, Zeeman GG, Cipolla MJ (2007) Pregnancy prevents hypertensive remodeling and decreases myogenic reactivity in posterior cerebral arteries from Dahl salt-sensitive rats: a role in eclampsia? Am J Physiol Heart Circ Physiol 292: 1071-1076.
- Bartynski WS, Boardman JF (2007) Distinct imaging patterns and lesion distribution in posterior reversible encephalopathy syndrome. AJNR Am J Neuroradiol 28: 1320-1327.
- Schwartz RB, Mulkern RV, Gudbjartsson H, Jolesz F (1998) Diffusion-weighted MR imaging in hypertensive encephalopathy: clues to pathogenesis. AJNR Am J Neuroradiol 19: 859-862.
Citation: Twayana RS, Poudel R, Paniyar RK, Rana PS (2017) Posterior Reversible Encephalopathy Syndrome: A Rare Complication of Peripartum Pre-eclampsia or Eclampsia- A Case report. J Clin Stud Med Case Rep 4: 045
Copyright: © 2017 Ram Sundar Twayana, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

