ABSTRACT
Objectives: Despite little data on efficacy, 5-Aminosalicylates (5-ASA) are commonly used in pediatric Crohn's Disease (CD). Our aim was to assess prevalence of 5-ASA utilization in children newly diagnosed with CD, as well as clinical outcomes among these patients.
Study design: Data were obtained from a large observational inception cohort from 2002-2014. First, we analyzed initial treatments received immediately following diagnosis. Then, clinical outcome and disease activity were measured using the "Physician Global Assessment" (PGA) scale. The primary outcome was a PGA of "inactive", without corticosteroids (CS), immunomodulators, biologics or surgery one year following diagnosis in patients receiving 5-ASA ± CS only as initial therapy following diagnosis.
Results: 440/1297 subjects with CD (34%) received 5-ASA ± CS only as initial therapy, and were the focus of this study. No baseline differences were observed between the 5-ASA + CS (n=263) vs. 5-ASA - CS (n=177) treatment groups for age, gender, disease distribution or disease behavior. Baseline moderate/severe PGA was more common in the 5-ASA + CS group compared with the 5-ASA alone group (70% vs. 38%, p<0.001). The primary outcome was achieved by 34% of those treated with 5-ASA alone vs. 18% of those treated with 5-ASA + CS (p<0.001). In multivariate models, achieving the primary outcome was significantly associated with initially mild disease severity and no initial CS use.
Conclusion: The prevalence of 5-ASA utilization for pediatric CD is high despite a low likelihood of achieving clinical remission on 5-ASA therapy, although somewhat more favorable for select children with mild disease who do not receive CS at diagnosis.
Keywords: Aminosalicylates; Children; Crohn's disease
Introduction
While efficacy supporting 5-aminosalicylate (5-ASA) use for the induction and maintenance of remission for Ulcerative Colitis (UC) has been well established in adult as well as several pediatric studies [1-6], evidence regarding 5-ASA use in Crohn's Disease (CD) for induction or maintenance of remission, or to prevent relapse after surgically induced remission, is conflicting and weak [7-12]. Despite the absence of strong supporting data, 5-ASA compounds are commonly prescribed for CD in general clinical practice. A 2014 Swiss Cohort study including adults and children found that among 1420 patients with CD 59% of patients had been treated with 5-ASA at some time [13].From our large prospective, inception cohort of children newly diagnosed with CD which included patients from 30 pediatric centers in North America managed by independently practicing pediatric gastroenterologists we aimed to:(1) Describe the prevalence of 5-ASA utilization (2) Describe clinical outcomes at 1-3 years following therapy with 5-ASA ± corticosteroids (CS) (3) Identify clinical and demographic factors associated with clinical remission
Methods and Study Population
This was a prospective, multicenter, inception cohort study of a sub-group of children with CD enrolled in the Pediatric Inflammatory Bowel Disease Collaborative Research Group Registry. The Registry was an ongoing, observational research program conducted at 30 pediatric gastroenterology centers in North America for which patients were enrolled between 2002 - 2014. Children under the age of 16 years newly diagnosed with inflammatory bowel disease (CD, UC or indeterminate colitis), were eligible for enrollment in the Registry through one of the participating sites. Medical data for each enrolled patient were collected and submitted to a centralized data management center. Data were submitted at the time of diagnosis, 30 days after diagnosis, and every 3 months thereafter. Demographic data were recorded as were standardized laboratory tests (serum albumin, hemoglobin, erythrocyte sedimentation rate) when available. Disease location and behavior were classified by the Paris system [14]. All patients were managed by individual physician dictate and not by standardized protocols.Per our first aim, we collected data on initial treatment prescribed following diagnosis. For this we analyzed treatments listed at 30 days following diagnosis which was the first data collection point following diagnosis. We looked at the utilization of 5-ASA alone as well as in combination with other treatments. We also collected information on the utilization of other treatments regimens that did not include 5-ASA.Next, we focused on the group of patients who received 5-ASA as the only maintenance therapy given initially following diagnosis in order to study outcomes among these patients. For this analysis we included patients also given corticosteroids (CS) initially for induction of remission. In addition, we did not exclude the small number of patients who also received rectal therapies. However, we excluded all patients receiving any other CD-specific oral or parenteral maintenance treatment at 30 days following diagnosis. Approximately 97% of patients receiving 5-ASA received mesalamine and only 3% received sulfasalazine. In order to simplify our statistical analysis, we combined results and refer to both compounds collectively as 5-ASA. Dose was recorded when available and expressed in mg/kg/day.
Disease activity and outcome measures for 5-ASA use
Disease activity was classified by the Physician Global Assessment (PGA), which is a validated index that categorizes disease as "inactive", "mild" or "moderate/severe" [15,16]. This tool takes into account clinical characteristics of Crohn's disease severity including abdominal pain, diarrhea, bleeding, weight loss and linear growth as well as other elements of the physical examination. In most cases PGA is determined prior to knowledge of laboratory studies. Previous work has shown very good correlation between PGA and the Pediatric Crohn's Disease Activity Index (PCDAI) [15,16].The primary clinical outcome for our analysis was remission at 1 year or "inactive disease" by PGA designation, off CS, and without escalation of therapy to immunomodulators (IM), anti-Tumor Necrosis Factor (TNF) therapy, or resectional surgery. A secondary outcome measure was "response" or "mild disease" by PGA designation with similar constraints. Additional secondary outcomes included disease activity by PGA at two years and three years following diagnosis, as well as by Pediatric Crohn's Disease Activity Index (PCDAI) when data for calculation were available [16].
Statistical analysis
Groups were compared using t-tests for continuously distributed variables and chi-square and exact tests for categorical variables. Multivariate logistic regression was used to analyze possible predictors of our primary outcome. Kaplan-Meier analysis and log-rank tests were used to compare differences in duration of remission between the study groups. A significance level of 0.05 was used for all tests.
Institutional Review Board (IRB)
The IRB at each participating Center approved the protocol. Informed consent and, when appropriate, assent was obtained from all parents/caregivers and patients enrolled in the Registry in accordance with local regulations.
Results
Deriving the study population and utilization of 5-ASA
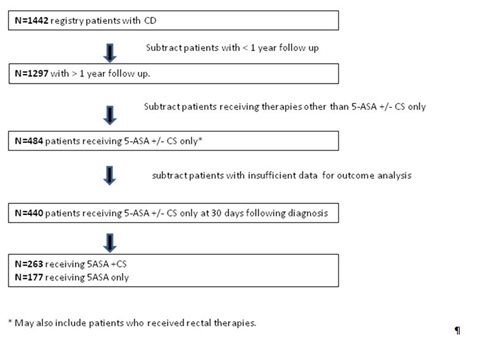
During the study period there were 2116 registry patients enrolled, of whom 1442 were diagnosed with CD (Figure 1). 1297 of these patients had at least 1 year of follow up. Within this subgroup of 1297 patients there were 31distinct treatment combinations prescribed by 30 days following diagnosis that included one or more of the following treatments: 5-ASA, IM, anti-TNF therapy, exclusive enteral nutrition therapy, rectal therapy (our database does not specify dose or type of rectal therapy used), and surgery. Table 1A - 1B presents the highlights of our analysis. Of note, the most frequent treatment prescribed at 30 days following diagnosis was 5-ASA with 53% of the total population receiving this alone or in combination. IM were used in 40%, Biologics (infliximab or adalimumab in all cases) were used in 7%, enteral nutrition therapy was used in 5%, rectal therapies in 3%, surgery in <1%, and calcineurin inhibitor (CI) were used in <<1% of all patients. Of note, when we looked at treatments recorded at 30 days following diagnosis during the first half of our observation period compared to the second half we noted a higher rate of 5-ASA utilization in the first half (57% vs. 46%), a lower rate of anti-TNF therapy in the first half (5% vs. 10%), and a constant utilization of IM throughout the study period (40%). Table 1B shows the most frequent treatment combinations prescribed at 30 days. Of 31 distinct combinations of therapy prescribed, 9 accounted for 88% of all patients and are shown in the figure. 5-ASA + CS were the most frequently prescribed combination accounting for 22% of the total. 5-ASA alone accounts for 16% of the total.
In order to analyze 1 year outcomes data following 5-ASA use, we identified the subset of individuals for whom 5-ASA ± CS was prescribed as the only therapy by 30 days followed diagnosis (n=484). Forty-four of these patients were excluded from further analysis because of incomplete data, leaving 440 subjects (34% of all patients with CD in this study with >1 year of follow up) who form our study group. Of these patients, 177 received 5-ASA without CS and 263 received 5-ASA with CS at 30 days following diagnosis Figure 1.
Demographic and clinical characteristics at diagnosis of study population
Clinical and demographic characteristics of the study population are shown in table 2. No baseline differences were observed between 5-ASA - CS vs. 5-ASA + CS groups for gender, age, disease distribution, presence/absence of perianal disease, baseline behavior (Paris classification), hemoglobin level, presence/absence of growth failure (height z-score <-1.65), or dose of 5-ASA used. The mean 5-ASA dose was 50 mg/kg/day. Baseline PGA was significantly different between the two groups with a higher percentage of moderate to severe disease, higher PCDAI scores, higher ESR (mm/hr), lower albumin (g/dL), and higher platelet count in those subjects who received 5-ASA + CS, likely reflecting a greater burden of disease.
Clinical outcomes at 1 year
For the 440 patients receiving 5-ASA ± CS within the first 30 days following diagnosis, 25% (n=108) had an inactive PGA and were corticosteroid free without escalation of therapy or surgery by one year (Table 3A). An additional 8% had a mild PGA while meeting similar criteria. We further delineated 1 year outcomes as a function of initial CS use and/or initial PGA. Depending on the subgroup, a positive outcome (response or remission) after 1 year ranged between 18% - 58%. Not-unexpectedly, patients who were PGA mild at diagnosis and only received 5-ASA without CS were most likely to have CS-free remission at 1 year following diagnosis without step up therapy. Patients with moderate/severe PGA at diagnosis and who received CS were least likely to have a positive outcome. Of the total, 67% (n=295) did not achieve primary or secondary outcome. Of these, 92 had at least 1 hospitalization, 11 underwent surgery, 265 received step up therapy with IM +/- biologic therapy, 65 required CS and 48 were classified as moderate/severe PGA at the one-year data collection point.
Clinical outcomes at 2 - 3 years
Data at 2 years following diagnosis was available for 365 patients initially treated with 5-ASA ± CS (Tables 3B-3C). The likelihood of a favorable outcome diminished after 2 years. Of these patients 18% and 4% achieved the primary and secondary outcome, respectively (CS free, no step-up therapy, and PGA inactive or mild). For the remaining 78% 122 had at least 1 hospitalization, 20 underwent surgery 264 escalated to IM +/- biologic therapy, 36 were classified as moderate/severe PGA and 38 required CS.Data at 3 years following diagnosis were available for 289 patients initially treated with 5-ASA ± CS. We observed that the likelihood of a favorable outcome diminished further. 13% achieved primary outcome and 3% achieved secondary outcome at that point. For the remaining 84% 113 had at least 1 hospitalization, 22 underwent surgery 236 escalated to IM +/- biologic therapy, 15 were classified as moderate/severe PGA and 25 required CS. Kaplan-Meier analysis table 3D shows the probability of being in remission (± standard error) at 1, 2 and 3 years to be 0.34 ± 0.04, 0.24 ± 0.03, and 0.20 ± 0.03 for the 5-ASA only patients compared to 0.18 ± 0.02, 0.12 ± 0.02 and 0.07 ± 0.02 for the 5-ASA + corticosteroid group, respectively (p<0.001). Results by baseline PGA and for treatment/PGA subgroups are also presented in the table.
PCDAI
Data points needed for PCDAI (Pediatric Crohn's Disease Activity Index), or weighted - PCDAI were not available in many cases. Since PGA data were widely available and yielded the most robust and complete data set and has been shown to correlate well with PCDAI [16], it was primarily used for this study. We performed a sub-analysis looking at 1, 2 and 3 year outcomes data for the patients for whom PCDAI data were available. PCDAI data were analyzed irrespective of whether step up therapies including CS were used. We defined inactive disease/remission as PCDAI
Laboratory studies
We examined laboratory studies in those patients achieving PGA inactive at one year in an attempt to determine whether surrogate markers of inflammation may have normalized as well. For the 60 patients treated with 5-ASA only who achieved remission at 1 year, for whom we had laboratory data available, the percent with normal labs (ESR<20 mm/hour, albumin >3.4 g/dL, Hemoglobin ≥11 g/dL) at baseline versus 1 year were 32% versus 85% for ESR, 66% versus 84% for albumin, and 68% versus 94% for hemoglobin. For the 48 patients treated with 5-ASA + CS who achieved remission at 1 year, for whom we had laboratory data available, the percent with normal labs at baseline versus 1 year were 47% vs. 85% for ESR, 59% vs. 96% for albumin and 70% vs. 100% for hemoglobin. Overall, a trend of lab normalization was seen in many but not all patients who achieved remission.
Growth
Growth delay is an important consideration in the treatment of pediatric CD. We found that 11% of the study subjects had low height z scores at diagnosis (<-1.65). This percentage is lower than that found previously [17,18]. For patients receiving 5-ASA+/- CS initially following a CD diagnosis, 43/346 (12%) had a low height z score at 1-year. At 2 and 3 years following diagnosis 29/267 (11%) and 20/210 (10%) had a low height z score, respectively. There was no statistically significant difference in the incidence of growth failure at 1, 2 or 3 years following a CD diagnosis between patients achieving remission and those that did not achieve remission. It is likely that children presenting with growth failure were less likely to be treated with 5-ASA ± CS without additional initial therapy such as IM or biologics and therefore would not have been included in our study group.
Predictors of response
We attempted to identify relevant predictors of one-year clinical remission including age, gender, PGA at diagnosis, location of disease on presentation and whether or not CS were utilized within the first 30 days following diagnosis. Table 4 shows the results of logistic regression analysis. In the multivariate models both mild disease severity at diagnosis and absence of CS use within the first 30 days after diagnosis were significantly associated with the desired outcome. Normal laboratory studies including hemoglobin, ESR, and albumin were not independently associated with PGA inactive without CS or additional therapy at one year after adjusting for initial PGA and CS use.
Summary and Discussion
For most children with CD who present with moderate to severe symptoms, and often with growth delay, current therapy frequently includes the early use of immunomodulators and biologics [19,20] which indeed, has been associated with improved clinical outcomes at one year following diagnosis of very ill children [21]. More difficult, however, is choosing initial therapy in those children who present with mild disease for whom there is parental and sometimes clinician reluctance to use therapies associated with potentially more significant side effects. 5-ASA therapy, which is perceived to have a more favorable side effect profile, is often used per anecdotal evidence as well as sparse published data [13] for patients with milder disease despite minimal data in the pediatric (or adult) Crohn's disease literature to support this practice.In this prospective multicenter observational registry study, we aimed to provide data on 5-ASA utilization among children newly diagnosed with CD. We also aimed to describe clinical outcomes in patients following 5-ASA treatment. At 30 days following a CD diagnosis which is the first data collection point in our study design, 5-ASA appears to be the most frequent initial treatment prescribed with over half (53%) of all patients receiving 5-ASA either alone or in combination. Furthermore, over one-third (37%) of patients received only 5-ASA ± CS in the first 30 days following diagnosis. Outcomes data show that 25% of all study subjects who started on only 5-ASA +/- CS had clinically inactive disease without the need for CS or step up therapy at 1 year following diagnosis. When stratified by PGA at diagnosis and/or need for CS at 30 days, 1 year outcomes varied considerably. CS-free remission or response at 1 year without step up therapy was as high as 58% for the subgroup of patients with mild PGA at diagnosis whom were not given CS initially. Among patients with moderate/severe PGA at diagnosis who were given initial CS (undoubtedly reflecting a sicker population), only 18% achieved primary or secondary outcome. We did not find that disease location, age, or gender influenced outcomes. When looking at outcomes data after 2-3 years, favorable outcomes diminished considerably. Patients started on only 5-ASA +/- CS at diagnosis whom had clinically inactive disease without the need for CS or step up therapy at 2 and 3 years, were 22% and 16%, respectively. The subset who were most likely to have a favorable outcome at 2 and 3 years were those receiving 5-ASA only without CS and who had mild PGA at diagnosis. For this subset, remission or response was noted to be 46% and 34% at 2 and 3 years, respectively.As this was not a controlled study with a single preparation of 5-ASA we are only able to give dosing data in aggregate. The mean 5-ASA dose was about 50 mg/kg/day. Given that efficacy in adult patients with ulcerative colitis may be improved with higher dosing schedules [22] as well as some adult CD studies that provide data on dose [12]. It is possible that dosing greater than 50 mg/kg/day might be more effective.The main strengths of our study are the size of the population studied and the breadth of an experience from multiple institutions reflecting true real world experience. However, our study is not a randomized, case-control clinical trial. All treatment choices were dictated by physician discretion and were not protocol based including doses and specific formulations of 5-ASA compounds, use of rectal therapies, and decision to start or move to alternative therapies. Therefore, placebo effect and/or selection bias should be considered. While placebo effect has been demonstrated in adult clinical trials of inflammatory bowel disease [23,24], similar studies have not been conducted in pediatrics and therefore, we have no data on the potential size of this effect in our population. We did note, however, that standard laboratory measures of disease activity normalized in many patients who achieved an inactive PGA suggesting improvement of inflammation.In addition, adherence was not systematically monitored and can be a significant confounder. And other markers that may potentially correlate with clinical outcomes that have gained more uniform use were not systematically collected throughout the study period and were not included in our analysis. Such markers include C-reactive protein, fecal calprotectin, X-ray computed tomography and/or magnetic resonance enterography and repeat endoscopic examinations to document mucosal healing. The use of PGA as an outcome variable is not ideal though reasonable correlation with PCDAI has been demonstrated [16]. Since PGA was all that was available for many of the study subjects, it was therefore utilized for our study analysis. We presented PCDAI data when available.Given that over half of children newly diagnosed with CD are receiving 5-ASA medications as part of their initial therapy, it is important to develop evidence on efficacy or futility. It is highly unlikely that future placebo controlled trials of 5-ASA will be conducted in children and therefore observational registry data may be all that is available. pediatric literature and clinical experience, most pediatric practitioners would agree that use of 5-ASA monotherapy is not appropriate for pediatric patients with CD who demonstrate moderate to severe phenotypical features including extensive small bowel disease, stricturing or penetrating disease, or disease that impacts on growth or pubertal development [19-21]. Our data suggest the possibility of clinical utility in select patients with initially mild disease, though further exploration of specific compounds as well as dosing schedules would be helpful. For patients with more significant disease activity, however, our outcomes data (1-3 years following diagnosis) suggests that the use of 5-ASA as primary therapy does not appear to be efficacious. The development of newer risk stratification tools including serology, genetics and microbiome analysis may help identify patients at lower risk for progressive disease [25].
Acknowledgements/Sources of Funding and Conflict of Interest Statement
Early support for the present study came from an unrestricted grant provided by Janssen Ortho-Biotech. There was no external funding from 2012 to the close of the Registry. We would like to acknowledge participants of the "Pediatric Inflammatory Bowel Disease Collaborative Research Group Registry" for enrolling patients, collecting and submitting data to the centralized data management center.Bella Zeisler, Trudy Lerer, report no conflicts of interest. Jeffrey Hyams reports the following disclosures: Janssen Ortho Biotech-research support, advisory board. Consultant Abbvie, Celgene, Takeda, Lilly, Boerhinger Ingelheim, Astra Zeneca. Shire- research support.
Roles in the Submitted Work
Bella Zeisler - Design of work, interpretation of data, drafting and revising work, final approval and accountability.Jeffrey Hyams - Acquisition of data, design of work, interpretation of data, drafting and revising work, final approval and accountability.Trudy Lerer - Design of work, interpretation of data, statistical analysis drafting and revising work, final approval and accountability.