Non Invasive Evaluation of Pelvic Congestion Syndrome
*Corresponding Author(s):
Pier Luigi AntignaniVascular Centre, Nuova Villa Claudia, Rome, Italy
Tel:+39335318430,
Email:antignanipl@gmail.com
Abstract
Pelvic Congestion Syndrome (PCS) is one of possible cause of chronic pelvic pain in women. PCS is described as chronic pelvic pain arising from dilated and refluxing incompetent pelvic veins. The diagnosis is based on patient-reported symptoms, clinical examination, anatomical features, duplex scanner ultrasound and, sometimes, magnetic resonance examinations and venographic findings [1].
Keywords
EDITORIAL
The identification of incompetent pelvic veins is essential for the diagnosis of PCS. Non-invasive methods such as ultrasound and magnetic resonance imaging are the first line of investigation, with assessment of the blood velocity and flow pattern. The main symptom of Pelvic Congestion Syndrome (PCS) is pelvic pain [2]. Women typically have a dull, throbbing, and achy pain in the vulvar region, which often worsens during or after intercourse, just before the onset of menstruation, and as the day progresses, especially in women who stand or sit for long periods. Symptoms must be present for at least 6 months before a diagnosis of PCS can be considered. Pelvic pain and refluxing pelvic veins are often present in premenopausal women; however, their presence does not always establish a cause and effect relationship. In patients who have clinical symptoms, signs, and imaging findings compatible with PCS, the diagnosis can be made only after other causes of abdominal and pelvic pain have been excluded [1,2]. The differential diagnoses for pelvic pain are vast, further adding to the complexity of the disorder. These diagnoses include endometriosis, chronic pelvic inflammatory disease, leiomyoma, adenomyosis, Nutcracker syndrome, diverticulitis, diverticulosis, Meckel’s diverticulum, interstitial cystitis, abnormal bladder function, chronic urethritis, fasciitis, nerve entrapment syndrome, hernia, scoliosis, spondylolisthesis, osteitis pubis, somatization, psychosexual dysfunction, and depression. The diagnosis of pelvic congestion syndrome can only be made by excluding other causes of chronic pelvic pain, and managing this complex condition can be a challenge for the primary-care provider.
The physical examination should evaluate the varicose vein network, which may be present on the perineal, vulval, gluteal, or posterior thigh areas. According to Monedero et al., [3,4] varicosities develop due to reflux in the left and right ovarian and pudendal-obturator veins in the case of pelvic floor insufficiency; whereas, perineal, gluteal, and posterior thigh varicosities are mostly due to reflux in the internal iliac vein. Diagnostic techniques can be noninvasive (duplex ultrasound) or minimally invasive (magnetic resonance, CT and phlebography) [5]. Duplex ultrasound is regarded as a first-line investigation for PCS because it is noninvasive, easily accessible, and inexpensive. This procedure provides essential hemodynamic information, and when combining transvaginal and transabdominal approaches, it could determine the presence of venous dilations, reflux, and compressions. Cross-sectional imaging techniques, such as CT or magnetic resonance scans, are often ordered for abdominal pain, which shows venous varices in the area of the uterus or the pelvis. CT can exclude other pelvic pathologies [2,6,7].
In comparison with CT, magnetic resonance venography is superior due to the absence of radiation. Catheter-directed retrograde selective venography or phlebography is the “gold-standard” method for the diagnosis of pelvic venous pathology, in all the variations according to the need. Phlebography confirms the diagnosis and assesses the venous anatomy, especially the collateral venous systems, and it is useful when planning embolization and coil selection [8,9]. In addition, this procedure is usually performed at the same time as the treatment, with consequent benefits for both patients and physicians. The correct diagnosis of PCS is essential when choosing a treatment option, especially due to the high number of under diagnosed cases. A vascular explorations are an essential element in the diagnosis and follow-up of venous disease through non-invasive methods. These non-invasive techniques provide highly reliable and sensitive data on the degree of venous stasis and the permeability and degree of valvular sufficiency in superficial and deep venous systems. These techniques can be used to determine the existence of venous disease, where it is located, and the level of hemodynamic compromise, to diagnose the disease in the early stages in asymptomatic patients of high-risk populations and to identify subclinical venous disorders and monitor and evaluate surgery, sclerotherapy, embolization, or medical treatment (Figures 1 and 2) [9-11].
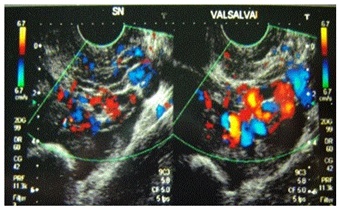
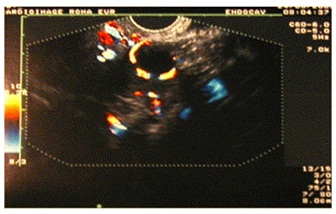
Duplex ultrasound scans provide essential hemodynamic information. Duplex ultrasound can determine the direction and speed of blood flow, and it can assess possible obstructions with in a vessel. The main problem of ultrasound investigations is that it is completely dependent on the observer; therefore, it is necessary to have specific training to determine all the pelvic venous structures. The most appropriate way to investigate pelvic veins by ultrasound is to combine both transvaginal and transabdominal approaches [11-13]. Transvaginal duplex ultrasound is performed on female patients in a gynecological position. It is used to obtain anatomical information and a hemodynamic diagnosis. The sonographic appearance of a normal pelvic venous plexus includes one or two straight tubular structures with a diameter of < 5 mm. However, the sonographic appearance of ovarian and pelvic varicoceles is multiple dilated veins (so-called “venous lakes”) around the ovary and uterus with a venous doppler signal of varying amplitude. The presence of circular or linear venous structures with a diameter > 5 mm is indicative of pelvic varicosities. The distribution (predominantly anterior or posterior) and side (left, right, or bilateral) should be described. With transvaginal duplex ultrasound, it is also possible to evaluate the presence of reflux, dilatation in gonadal veins, and the existence of uterine-ovarian varicose veins. In addition, a transvaginal duplex ultrasound provides important information for the diagnosis of a possible compression in the iliac vena cavasector or the aortic-mesenteric fork, identifying the presence of centrifugal (reflux), centripetal (derivative), or mixed flow [1,3,5,7,11].
The diagnosis of a compressive syndrome that causes PCS is performed using a transparietal duplex ultrasound, which is essential in morphological studies of the left renal vein and its hemodynamic behavior across the aortic mesenteric fork. In PCS, transparietal duplex ultrasound can detect pelvic varices and the presence of reflux during a valsalva maneuver. It can show a possible increase in the flow rate or the reversion of that flow through the left gonadal vein. The flow for an insufficiency pattern is of slow velocity, frequently with dilatation, venous stasis at rest, reflux (lasts more than 1 second), and an increase in velocity with the valsalva maneuver. It is produced in cases of valvular in competence of ovarian or internal iliac veins, and it is frequently seen in women with three or more pregnancies [6,12,13]. The flow for a derivative pattern is of high velocity at rest (usually ≥ 5 cm/s), with morphologic dilatation, no visiblere flux, and no reduction in flow velocity with the valsalva maneuver. It is seen in venous obstruction cases (as occurs in May-Thurner or Nutcracker syndrome and thrombosis or anomalies of the iliac or the cava vein). To rule out a compressive syndrome of the left renal vein, it is important to measure the ratio of the anteroposterior diameter between the hilar and the aorto mesenteric portion of the left renal vein (an anteroposterior ratio > 5 is considered abnormal) and the ratio of the peak velocity of the left renal vein between the aorto mesenteric and the hilar portion (a velocity ratio > 5 is considered significant) [11,13].
Meanwhile, decreases in flow in the left common iliac veinor reversion through the left internal iliac vein are frequent findings in the compressive May-Thurner Syndrome (MTS). For detecting left common iliac vein compressive syndrome, it is useful to measure the ratio of the peak velocity between the left common iliac vein (in the site of intersection with the right iliac artery) and the left external iliac vein. A velocity ratio > 5 is indicative of significant stenosis. It is also important to visualize the flow direction of the left internal iliac vein (normal or reversed) [14,15].
There are no generally accepted, well-defined clinical criteria for the diagnosis of PCS. We suggest a simple score system to identify the clinical symptoms and signs and the non invasive diagnostic (transvaginal ultrasound and magnetic resonance) data. The maximum clinical score is 27 (9 signs and symptoms with values from 0 to 3: chronic pelvic pain, dispareunya, vulval/perineal varices, atypical leg varices, dysmenorrhea, vulval congestion, haemorrhoids, premestrual and mestrual varices pain, sudden urination).

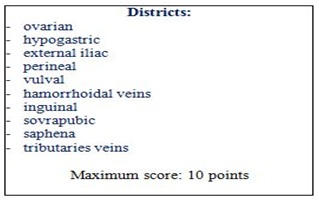
The instrumental score is 10 (1 point for each 10 venous district). The most common ultrasound criteria are: tortuous pelvic veins with a diameter of greater than 6 mm. Mean diameter in PCS ~ 8 mm. Slow blood flow < 3 cm/second or reversed caudal flow in the left ovarian vein demonstrated by Doppler waveforms. Dilated arcuate veins in the myometrium that communicate between the bilateral pelvic varicose veins.
In conclusion, the associated symptomatology with PCS can become extremely debilitating due to chronic lower abdomen pain, dyspareunia, dysmenorrhea, swollen vulva, lumbosacral neuropathy, and urinary urgency. An accurate diagnostic protocol requires the involvement of different specialists and diagnostic techniques. In addition to the ovarian and pelvic plexus engorgement the PCS definition also includes lower limb varicose veins fed by pelvic escape points. According to clinical experience, a clear distinction should be made between PCS and lower limb chronic venous disease of pelvic origin. An anatomical and pathophysiological link exists between the two conditions, but two distinct clinical scenarios characterize these different venous impairments.
REFERENCES
- Allegra C, Antignani PL, Kalodiki E (2013) News in Phlebology. Edizioni Minerva Medica, Torino, Italy.
- Antignani PL, Geroulakos G, Bokuchava M (2016) Clinical aspects of pelvic congestion syndrome. Phlebolymphology 23: 127-129.
- Monedero JL (1997) Insuficiencia Venosa crónica de la Pelvis y de los Miembros Inferiors. University City Tegucigalpa, MDC Honduras, Honduras.
- Monedero JL, Ezpeleta SZ, Perrin M (2012) Pelvic congestion syndrome can be treated operatively with good long-term results. Phlebology 27: 65-73.
- Jin KN, Lee W, Jae HJ, Yin YH, Chung JW, et al. (2009) Venous reflux from the pelvis and vulvoperineal region as a possible cause of lower extremity varicose veins: diagnosis with computed tomographic and ultrasonographic findings. J Comput Assist Tomogr 33: 763-769.
- Ezpeleta SZ, Monedero JL, Elkashishi AT (2016) Instrumental diagnosis of pelvic congestion syndrome. Phlebolymphology 23: 130-134.
- Park SJ, Lim JW, Ko YT, Lee DH, Yoon Y, et al. (2004) Diagnosis of pelvic congestion syndrome using trans abdominal and trans vaginal sonography. AJR Am J Roentgenol 182: 683-688.
- Lazarashvili Z, Antignani PL, Monedero JL (2016) Pelvic congestion syndrome: prevalence and quality of life. Phlebolymphology 23: 123-126.
- Dos Santos SJ, Holdstock JM, Harrison CC, Lopez AJ, Whiteley MS (2015) Ovarian Vein Diameter Cannot Be Used as an Indicator of Ovarian Venous Reflux. Eur J Vasc Endovasc Surg 49: 90-94.
- Greiner M, Dadon M, Lemasle P, Cluzel P (2012) How does the patho-physiology influence the treatment of pelvic congestion syndrome and is the result long-lasting? Phlebology 27: 58-64.
- Lazarashvili Z, Antignani PL, Monedero JL, Ezpeleta SZ (2016) Pelvic congestion syndrome: How to correctly manage it today. Acta Phlebologica 17: 23-26.
- Steenbeek MP, van der Vleuten CJM, Schultze Kool LJ, Nieboer TE (2018) Noninvasive diagnostic tools for pelvic congestion syndrome: a systematic review. Acta Obstet Gyenecol Scand 97: 776-786.
- Whiteley MS, Dos Santos SJ, Harrison CC, Holdstock JM, Lopez AJ (2015) Transvaginal duplex ultrasonography appears to be the gold standard investigation for the haemodynamic evaluation of pelvic venous reflux in the ovarian and internal iliac veins in women. Phlebology 30: 706-713.
- Gianesini S, Antignani PL, Tessari L (2016) Pelvic congestion syndrome: Does one name fit all? Phlebolymphology 23: 142-145.
- Mahmoud O, Vikatmaa P, Aho P, Halmesmäki K, Albäck A, et al. (2016) Efficacy of endovascular treatment for pelvic congestion syndrome. J Vasc Surg Venous Lymphat Disord 4: 355-370.
Citation: Antignani PL (2019) Non Invasive Evaluation of Pelvic Congestion Syndrome. J Non Invasive Vasc Invest 4: 011.
Copyright: © 2019 Pier Luigi Antignani, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.