ABSTRACT
Background
Following practices with the Lachman Test (subjective assessment) [1-7], we observe how rehabilitation and daily stress applied on the ACL after ACL surgery impacts the stiffness (or the compliance [8]) of the ligament graft over time and the repercussion it has on the knee (stability or clinical instability of the patient’s knee) but it was without any objective measurements and precise follow-up.
Hypothesis
The more the constraints applied on the graft are inadapted and harmful, the more the risk of functional instabilities is high when returning to pivot sport activities.
Study design
The study design was to analyze with more accuracy the evolution of graft’s stiffness during ligamentization time. We used a new compliancemeter and the first motorized device (GNRB arthrometer, Genourob company, France [9,10]), which can assess with accuracy the force/displacement curves of the ACL graft over time. In addition, we know the close link between compliance-metry and knee stability (Bercovy [8] with more than 1500 cases), which not the same as when only measuring laxity at one load applied.
Methods
We did an early follow-up (from day 0 to 2-3 Y) with the help of automated dynamic laximetry (or compliancemetry to be more specific). First study (hamstring tendon surgery) involved: 53 patients (15F, 38M). Second study (patellar tendon surgery) involved: 43 patients (8F, 32M). Many think that the evolution of the compliance (inverse of stiffness) of the graft only depends on biological factors (it only depends on the surgery technique we thought). Therefore, we want to understand if there are any other important aspects to consider. We want to know if, by applying biomechanical contraints on the knee, it would influence these grafts’ compliance after surgery. Additionally, does this apply when it is done when performing the same surgery technique by the same surgeon (using hamstring or patellar tendon techniques of surgery)?
Results
We obtained the laxity and compliance measurements (in mm & µm/N), which we compared to the clinical instability after one year (after returning to pivot sports). Results were given in µm/N and not in mm/N because it made it easier to read as numbers were round and without comas. Following the surgery, nobody showed divergent curves at the beginning (Day 0) (see Graph 2, “difference between healthy and operated knee curves”) but 15% of the patients (first study) had divergent curves at 1 month after surgery (same surgery, same surgeon, same technique but not the same rehab after surgery). We noticed that patients who underwent a more aggressive rehab with higher constraints applied on the graft (see on results of the survey) showed divergent curves (more compliance of the graft). However, if patients followed strict recommendations with less contraints applied on the graft, results after 1 Y were very good (parallel curves=good stiffness of the graft) with a high clinical stability. Nevertheless, it’s only possible during the first 3 months and not after 3 months to correct the compliance grafts (this is verified with the follow-up of more than 1000 ACL patient files).
Conclusion
This study has shown the influence of aggressive rehab and the influence of constraints applied outside rehab on the graft’s compliance. In addition, it has a big role on the patient’s knee instability following rehab. Also, the earlier you correct this (we show that it’s possible during the first 3 months), the more you decrease the risk of high instability in the future for patients (after 1 Y). That’s why we propose to do a compliance-metry test one month after surgery in order to allow a great follow-up of the ACL graft’s compliance during one year: 1 M, 3 M, 6 M and 1 Y post-op.
KEYWORDS
ACL; Automated dynamic laximetry; Laxity; Ligamentory compliance; Stiffness of ACL surgery
Introduction
What is Known
We know the biological evolution of ACL grafts, [11-16]. However, we know less concerning the impact of constraints applied on the ACL graft after surgery, more specifically regarding the compliance of the graft. During the eighties, Noyes F[6] has shown the interest of beginning rehabilitation early in the process after surgery. This was studied as back then, plasters were prescribed to avoid movements of the knee during the first 6 weeks. Today, it is the opposite with day clinic surgery. The patient indeed quickly recovers. Consequently, the rehab and patient recommandations must be done with caution.
What this Study Shows
The impact of constraints on the ACL graft’s compliance. We objectively measured it in this study. We show that during the first 3 months following the surgery, it is crucial to evaluate the ACL’s stiffness in order to detect any distension of the ACL graft, which would be problematic for the rest of ligamentization process. The sooner we detect distension; we indeed show that we can correct this distention, as long as it’s taken care of within the 3 months following the surgery (after 3 month it is difficult to alter the process).
Introduction
For a long time, we have known that the stiffness of the ACL (Anterior Cruciate Ligament) reconstructions evolved during time after the surgery [5,17-19]. It passes through a first stage during the per-surgery phase where it is very resistant [6,7,20]. The graft can indeed support up to twice as much loads as a native ACL (2000N). Then, this resistance decreases during the second stage, the necrosis stage (it has been evaluated that the ACL resistance during this stage is at about 40% compared to the native ACL=approximately 800N). After this stage, the ACL tendon graft transforms itself into a neoligament (6 months after the surgery, the resistance of this neoligament is at about 80% compared to a native ACL). The anchor points of the graft in the tunnels should also not be forgotten. These are indeed much less resistant compared to the graft just after the surgery [15,16].
The work of Bienvenutti [1], Markolf [3,4,21,22], Noyes [6,7], Butler [14,23] have shown that the Lachman test (Flexion of the knee between 15 and 25° as Torg has shown) has a high sensibility for detecting ACL ruptures because the ACL was the primary brake while applying anterior tibial translation (> 80%) in this articular range but this test lacked objectivity. The arrival of the first laximeters (Daniel [24], with KT 1000, Staubli [25] with Telos), now more than thirty years ago, allowed for the first time to objectify these tests done on ACL ruptures (in pre surgery) but analyzing the graft’s stiffness in particular during the post-surgery phase with accuracy was not possible. The KT 1000 and the Telos were pioneers in this field of study as they allowed analysis to be done with different forces applied on the tibia but they were either not precise enough or to invasive [25-45]. The GNRB® (Figure 1), which is a new motorized and computerized compliance-metry device created during the years 2000 (company Genourob®, France), see study of Robert [10], allowed measures to be much more precise regarding the compliance of the ACL following knee injuries (additionally to the evaluation of the differential of displacement at a certain force applied commonly known as laxity).
Nowadays, it is the only international study to have been done on the state of the resistance of the ACL reconstruction at day 0 (10 minutes after the surgery) until the full healing of it (more than two years after the surgery). During the tests, the data that is saved comprises several displacement measurements while applying different forces on the tibia: force/displacement allows the tracing of compliance curves (inverse of the stiffness or rigidity=1/R).
Maximal resistance of the graft and its anchor points during time were of course taken into account during the post-surgery phase. Thus, a GNRB test was done during the surgery (at the end of the surgical operation), the 1st month and the 2nd month with 100 N max applied (much less than the peak force that the ACL can sustain while walking, 350 N according to Nagura [42]) but enough to evaluate the state and behavior of the ACL graft while under stress.
During the surgical procedure, the graft has a resistance which is much higher than the normal ACL (around 4000N depending on the sample instead of 2000N for a normal native ACL) and the anchor points that are used today have resistances varying from 800 to more than 1000N. Therefore, the force applied during the GNRB test (100 N) is ten times lower than the lowest rupture thresholds, the ones that are associated to the anchor points! One month into the rehabilitation phase, the same force (100N) was applied by the GNRB but this time, the lowest rupture thresholds were rather located on the ACL graft itself (around 800N) and on the anchor points of the semitendinosus-gracilis (That is different when patellar tendon surgery is performed because of the presence of bone tissue at the two endpoint of the graft allowing healing in the femoral and tibial tunnels to be faster). Consequently, there is no way of modifying the ACL graft’s stiffness while doing GNRB tests. If that were the case, walking would be forbidden to the patient because the forces applied would be much higher (peak force > 300 N) than the GNRB tests at 100 N.
A study has been realized to follow the evolution of the resistance of ACL grafts over time. Tests were done per-surgery and gave objective results. The data collected allowed the creation of a database composed of the initial resistances that the ACL grafts provided just after the surgery (The patient were still under anesthesia=no muscular activity was therefore present and after the GNRB device can detect muscular activity during test [10]).
Another important fact is that this study has been realized by the same operator for each of the two surgical techniques (same surgeon that applied the same surgical procedure (semitendinosus-gracilis for one and patellar tendon technique for the other). All patients were diagnosed with isolated ACL rupture. This permitted concluding whether there were different final resistances with the same surgical procedure. The operator of the GNRB® was also the same experienced operator for both of the surgical techniques that were done (the tightening force used to maintain the patella against the femur were the same for both knees, likewise concerning the positioning of the patient’s knee). A recent study done at the Biomechanical department of the University of Lyon 1 has validated the high reproducibility of the GNRB® device with more than 10000 tests that were run whilst respecting different standards.
The starting point was to see if by applying different sollicitations after the surgery, the same stiffness or flexibility results were found on the graft one year later (with an initial resistance almost identical at day 0 like we will see later). After one year, the ACL graft has already strongly evolved generating a neoligament but we know that the ACL reconstruction process is not fully achieved. At this time, we can highlight the fact that the ACL reconstruction process is already very advanced and accordingly, at this state, the compliance does not modify a lot afterwards (see Table 1 and 5 Y after surgery).
The aim of this study was to demonstrate the evolution of the stiffness curves of these ACL reconstructions over time using none-invasive equipment in order to draw reliable conclusions from it. The interest we found in doing this was that it proved how the compliance curves of the ACL graft evolved over time after surgery (we supposed this but could not verify if it was true). Also, analyzing the curves can bring interesting information to the surgeon one month after the surgery. He/she can indeed see if the curves behave properly or not (biomechanic of the graft). We managed to prove that the evolution of these so-called compliance curves was different when patients were subjected to an aggressive rehab program. Finally, the surgeons can detect bad evolution of the graft’s compliance (divergent curves) and recommend the PTs to choose an adequate personalized rehabilitation program for the patient.
Methods
The first study was done on 53 individuals (15 women and 38 men) who were 33 years old in average [18-49]. See table 1 below. Ethics agency approval from XXX, France with n° 2013/07.
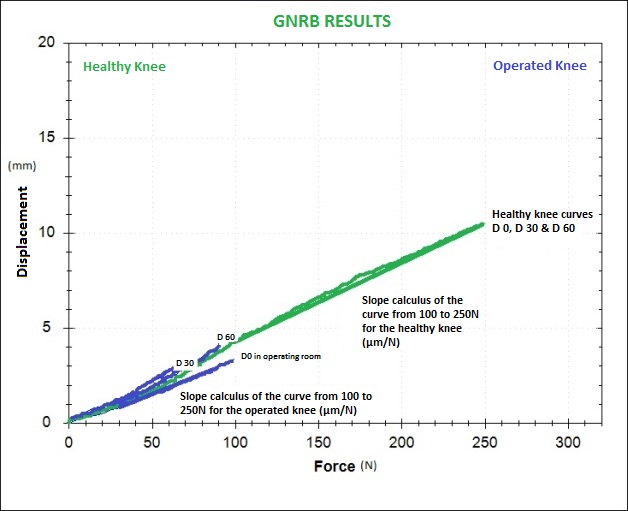
In the beginning, there were 63 patients but only 53 of them had isolated ACL tears. At the end of the surgery, GNRB tests were done on the surgery table (the patients were still under the effect of anesthesia). A series of tests were done with the GNRB® (Graph 1), product description [18], on the healthy knee (3 tests at 250N). Then, the pathological knee was also tested but only at 100N (3 tests). The reason we applied only 100N (and not 250N like on the healthy knee) was for safety of the ACL graft. However, it should be noted that when testing the patient at 250N of force, the displacement measurement is automatically saved at 100N of force. This consequently makes both assessments comparable. Muscular hamstring contraction could not falsify the tests since the patients were under anesthesia. Following the surgery, we could still detect hamstring muscular activity as electrodes were placed on the thigh and connected to the GNRB (with Biofeedback system).
Patients were seen again one month after the surgery (Graph 2) and additional tests were performed with the GNRB® (the same as the ones done at the end of the surgery). The same tests were also performed during the months following the surgery at 100N on the operated knee and 250N on the healthy knee. However, from 6 months after the surgery, both knees were tested at 250N. Tests were also run 1 year and 2 years after the surgery (at 250 N max for operated knees).
Results
Results were consequently analyzed by prioritizing the analysis of the curve’s parallelism (by analyzing the slopes of the curves and making sure they behave alike). This was of course done when the displacement differential between both legs was inferior than 2mm at 100N force applied on the tibia and when the differential of the slope of the curves (linear regression line) was less than 5µm/N. This slope differential matched the one done on the pathological leg because it is the same linear line. The notion of divergence (strong compliance) was characterized by a slope differential superior than 10µm/N for strong divergences (this has a strong link with knee instabilities according to Bercovy [8]).
Rehabilitation exercises only started 3 weeks after the surgery (The only instructions that were given to the patient was an auto-rehabilitation procedure when he exited the hospital).
At Day-0, 100% of the curves representing the operated knee were parallel to the ones representing the healthy knee, with more than 80% of them being underneath the curves of the healthy legs. This means that the ACL reconstructions were more tightened in the beginning. This is not very surprising because we know that this is intentionally done today according to the idea that the graft “loosens” during the months following surgery. It should be noted that the curves representing the operated knees did not show any divergence when compared with the curves of the healthy knee! This is an important fact because in the orthopaedic field, it is often believed that the surgical technique conditions at 100% the final result. Surgical techniques are nowadays pretty well handled. With this study, we show that the ACL graft can evolve favorably and unfavorably after the surgery depending on the sollicitations that are applied on it (with the same surgery and the same surgeon, we can observe different results one year after the surgery.
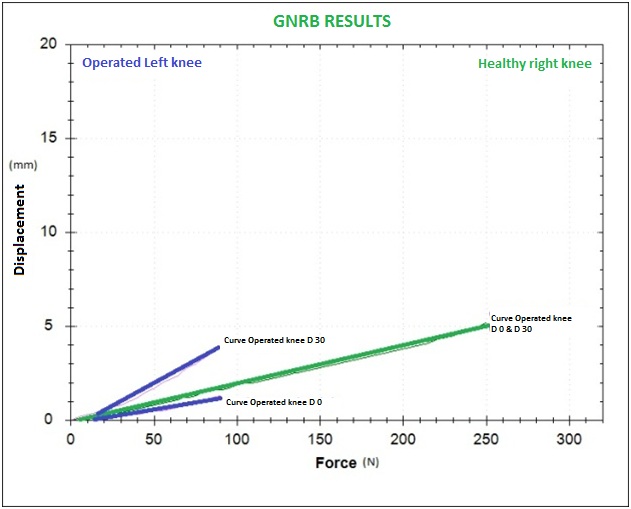
At D-30, we could observe that the grafts were less tense (more than 80% of the operated knee’s curve’s were above the curves calculated from the healthy knees. All those curves were parallel.) In 15% of the cases, we could see divergent curves (displacement differential > 2mm at 100N and slope differential > 10µm/N). We showed particular interest in those cases because we wanted to know if the curves would stay divergent over time if we did not apply any strict procedures during the rehabilitation.
A survey (Table 2) also permitted to note quickly all the daily practices to allow finding an explanation on the evolution towards divergent curves for some patients (Was it linked to biology or was it linked to biomechanical sollicitations?). Footwear (tightened shoes) or support stockings put in positions favoring the increasing of mechanical stresses on the graft have been identified. High mechanical stresses applied on the graft seem to be predominant (see results of the survey). Day-surgery has undoubtedly permitted rehabilitation to be much quicker for patients but it has also increased the possibility of applying too much mechanical stress on the graft. During the eighties, Noyes F has shown the importance of mobilizing the leg at an early stage after the surgery [7]. He indeed showed that by immobilizing the leg (with plasters), it decreased the resistance of ACL graft and made other problems like knee joint ankylosis surface. Following this, the rehabilitation was more active precociously. Today however, it is the opposite. We must recommand to slow down the quick rehab or lower the aggressive constraints applied on the ACL grafts after surgery [17,43-47]. However, the evolution was surprising concerning the patients who had strong follow-up and strict recommandations to decrease the constraints applied on the ACL grafts. 15% of the patients who showed divergent curves (first study) at one month were developing parallel curves month after month. We indeed witnessed the curves becoming parallel again over time whereas the curves of the patients that had not been followed (witness group) continued to diverge and kept their laxity differential. Nowadays, after having revised older folders belonging to the same surgeon using the same technique (> 1000 patients during 8 years), we can say that some patients who had ACL reconstruction surgery (With significant divergent curves > 6 months after surgery) have under went another surgery 2 years after the first surgery because clinical instabilities were difficult to live with for patients who wanted to do pivot sports again. Some patients got used to these loosened grafts but they also admitted to have low sport activity. In this case, we can talk about adaptation.
Finally, those patients consider themselves in good condition during every day life even though we could consider this as a therapeutical failure on a surgical scale. They also have a high risk on instability and of gonarthose in the long run (Meniscal surgery often happens next…) (Tables 3 and 4).
The second study focused on the analysis of patients that were operated using the patellar tendon technique. There were 41 patients (11F, 30 H with an average age of 26 years old [16,47]). See table 5 below.
In this study, we ran tests using the GNRB® one month after the surgery occurred. We almost did not have any cases of divergent curves (only 3%). However, we did notice some divergent curves making their apparition (7%) 6 months after the surgery, which means that occurred during the rehabilitation phase (without any measures that were taken like the ones that had been taken for the patients who underwent semitendinosus-gracilis surgery one month after the surgery). It should be noted that the divergences were not so high when comparing them to the ones of the first study (Slope differential < 10µm/N).
It seems that this patellar tendon technique allows fewer divergences to occur one month after surgery than with this hamstrings tendon technique (ANOVA p=0.003: there exists a significant difference between the data (Side to side differences for slopes and laxity at one month for both surgeries). Certainly, this must be linked with the rapid healing of the graft in the bone tunnels compared to the semitendinosus-gracilis and therefore, at this point, we can note a better endurance of graft sollicitations at this time. We must now take it into account in the rehabilitation programs.
This would explain why we prefer using the patellar tendon technique on professional athletes (the anchor points being more rapidly consolidated compared to semitendinosus-gracilis) because with the same sollicitations applied on the graft during the first and second month, we can conclude better results using the patellar tendon technique compared to semitendinosus-gracilis. If on the other hand we sollicitate differently applying less stress on the semitendinosus-gracilis graft (during the two first month post-surgery), we obtain an identical evolution than the one we get when using the patellar tendon technique: same results one year later with parallel curves and the curves are very close to each other (displacement differential < 2mm and slope differential < 5µm/N). Those patients are very stable on the field (while playing football for example).
Discussion
Laxity is different compared to compliance
Using the GNRB® and obtaining parallel curves (more than 1 or 2 years post-surgery) while having displacement differentials higher than 3mm at 134N (high laxity) is possible for a professional football player. Nevertheless, this player’s knee will be stable on the field even with high laxity as long as the compliance curves are parallel (operated/ healthy knees). In fact, we should extend the compliance curves to 1000N to understand the interest of measuring the slope because it would explain better why a player is stable or unstable on the field (this is not the only criteria but it is fundamental in this case). We recognize that other parameters play a role in knee instabilities like the neuro-muscular control but this state of ligament resistance is certainly predominant than laxity analysis (last threshold if the muscles are defective) to obtain a good stability in extreme situations like we find in pivot sports. The fact remains, however, that patients showing strong divergent curves 2 or 3 years later following surgery are often seen having another surgery or lowering their sport activity because of discomfort in every day life (Instability when working for example..): 100% cases (17) of 1145 patients in this study (with an average of 2% per year ACLR). The human body can adapt but still, it has its limits!
As quoted from the Bercovy studies [8] who analyzed « ACL Reconstruction Stiffness curves” (more than 1500 tests), a strong correlation was noted at the time (P 0.95 Value=0.0001) between clinical knee instabilities and high compliance (=divergent curves) of the ACL graft one year after surgery. This is not always true when analyzing just laxity differentials at a certain force applied.
Bercovy managed to take pictures using the Telos® (device that uses X-ray technology) while applying force at 0, 100, 150, 200, 250 and 300 N). Analyzing the compliance curves using this technique was the only one available back then. This was of course very invasive because of the X-ray repetitions but he at least got the merit of showing the interest of studying the compliance (=opposite of the stiffness) compared to the displacement differential at a given force (less precise and less discriminating on the state of resistance of the sollicited tissue). Dale Daniel has shown the same in creating the KT-2000 by showing stiffness curves on a computer at the end of the 80’s (The KT-1000 that preceded this device was sold in the USA in 1982). Consequently, during the 90’s, the idea of analyzing stiffness instead of the displacement differentials at a given force quickly emerged. However, 20 years later, nothing changed. We are still analyzing ACL laximetry at a given force and this method is used all over the world. A last study that was published in 2016 by the team of Pr Romain Seil [9] also pointed out this principle by calculating the two slope differentials (one before (P1) and one after 100N (P2)). Either way, displacement differential analysis at 134 or 200N (which would be more sensible and more specific than the displacement differential at 134N or at 250N using the GNRB®) associated with the analysis of ligamentory compliance (opposite of the stiffness) allows a better interpretation on the state and performance of ligamentous structures in terms of resistance. Quoting Bercovy: “The analysis of the displacement differential (laxity) at certain force is a numerical value without any biomechanical meaning”…
Finally, doing a test with the GNRB one month after surgery would allow controlling how the plasty is behaving according to plan just like we nowadays verify the muscle recovery 4 and 6 month after surgery with the help of isokinetic devices. Good understanding of biomechanics allows significant apprehending of which exercises are better compared to others. Concerning patients, it is the same thing: simplifying and popularizing what they have to do allow them to better understand why they have to do it this way whether than another way (see survey responses). Also, when seeing the GNRB curves, particularly when they are divergent, the patient tends to better understand (after a clear explanation) what they should and what they should not do. He therefore becomes much more attentive. Problem visualization, especially seeing divergent curves leads to a better understanding of what the patient should and should not do. This also allows a better follow-up of the strict rehabilitation recommendations and consequently leads to applying less solicitation on the plasty (better observance in these cases).
There is also no doubt that it would be interesting to be able to accumulate more data (with more samples) in order to analyze them more accurately.
Conclusion
Actually, this study allowed showing that even if the surgical procedure is well done; it will not prevent a bad evolution of the state and performance of the plasty one year later. All iterative and aggressive sollicitations that are endured by the plasty during the rehabilitation phase or after it, especially the ones that are made one month after surgery, will almost inevitably lead to a bad evolution of the resistance of the plasty. This most of the time has repercussions on knee stability and instabilities that are clinically validated concerning the knee of a patient one or two years after ACL surgery. Controlling the state and performance of the plasty one month after surgery using the GNRB® allows the surgeon to have a clear vision on the impact of rehabilitation or external sollicitations (patient). Concerning the physiotherapists, it allows them to modify or correct the rehabilitation process or the external sollicitations done by the patient (by insisting on lowering the sollicitations endured by the graft). This can be done knowing that at this stage, correction is still possible whereas 3 to 6 month after surgery it becomes too late. Also, rehabilitation must stick to the fact that it must not be too harsh and not too frequent in the beginning (with a strict follow-up of the recommandations) to let the healing take place (ligamentization) in the perfect conditions… It would be better to do it once a week during the first month (unless a problem is detected) and prolong it until resuming a pivot sport. This does not mean that nothing should be done in the beginning, in the contrary, exercises should be done but with the least sollicitations on the plasty (knowing that absence of constraints on the plasty is also harmful like Noyes has shown [6,7]) and objective data is needed to confirm this !
The first months are crucial concerning what Bercovy called “The survival curves of ACL grafts” and also to obtain excellent knee stability in the long term.
Figures
Figure 1: GNRB Motorized device: a new motorized and computerized laximeter.
Graph 1: Curves at D-0, D-30 and D-60) representing the operated leg are blue and curves (at D-0, D-30 and D-60) representing the healthy leg are green (This graph represents a good evolution of the ACL reconstruction).
Graph 2: Curves (D-0 and D-30) that are divergent at D-30 should undergo strict rehab procedures (it should be noted that at D-0, the curve of the operated knee was under the one of the healthy leg).