A Case of IgG4-Related Mikulicz’s Disease with Chronic Rhinosinusitis and Elevated Levels of IgG4-and IgE-Immune Complexes
*Corresponding Author(s):
Yasushi OtaDepartment Of Otorhinolaryngology, Toho University, Tokyo, Japan
Tel:+81 434628811,
Email:yasushiota5610@yahoo.co.jp
Abstract
We report a case of male IgG4-related Mikulicz’s disease with chronic rhinosinusitis with high serum IgG4-and IgE-Immune Complex (IC). Untreated serum IgG4 was 3,110 mg/dL, while Polyethylene Glycol (PEG)-treated serum IgG4-IC was 1,460mg/dL. Both were markedly elevated (normal IgG4 range <105 mg/dL). Serum IgE and PEG-treated serum IgE-IC was also elevated (normal IgE range <170 IU/mL) at 376 IU/mL and 237 IU/mL, respectively. IgE PEG Precipitated (PP) index was higher than normal (<20%) at 63%. We speculate that serum IgG4 and IgE represented autoantibodies for a component of the paranasal sinus mucosa and submandibular gland tissue.
Keywords
INTRODUCTION
CASE REPORT
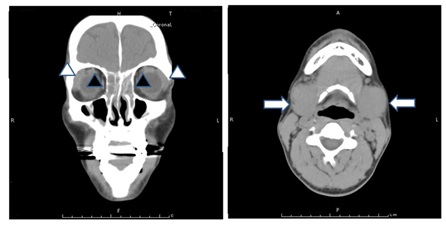
Figure 1: Computed Tomography (CT) of the paranasal sinuses and neck revealed bilateral sinusitis (black arrow heads), lacrimal gland swelling (white arrow heads) and bilateral submandibular glands (white arrows).
HISTOPATHOLOGICAL FINDINGS
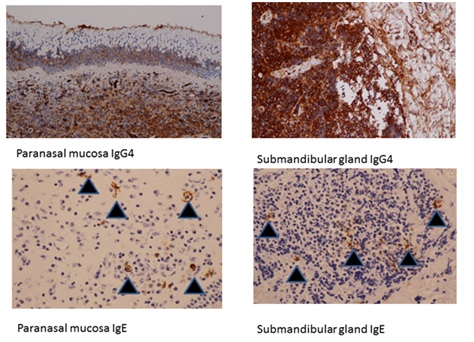
Figure 2: Histopathological findings revealed that most lymphocytes and plasma cells that had invaded the paranasal lamina propria, as well as epithelial cells in the parasinus mucosa and lymphocytes and plasma cells that had invaded the submandibular gland were strongly positive for anti-IgG4 antibody. Some mast cells and some of the stroma in the paranasal lamina propria and submandibular gland were positive for anti-IgE antibody (black arrows).
MEASUREMENT OF ANTIGEN-SPECIFIC IGG4 AND IGE ANTIBODIES IN THE SERUM
| sIgG4 (mg/dL) | sIgE (Iµ/mL) | |
| Total | 3110 | 420 |
| Allergen | ||
| Dermatophagoides pteronyssinus | 0.6282 | <0.10 |
| Dermatophagoide farinae | 0.8504 | <0.10 |
| House dust-Japan | 0.68 | <0.10 |
| Japanese Cedar | 0.4749 | 0.479 |
| Dog Dander | 0.4529 | <0.10 |
| Aspergillus fumigatus | 0.5092 | <0.10 |
| Mucor racemosus | 0.4332 | <0.10 |
| Candida albicans | 0.5558 | <0.10 |
| Alternaria tenuis | 0.4288 | <0.10 |
| Malassezia furfur (pityrosporum orbiculare) | 0.5514 | <0.10 |
| Enterotoxin A (S.aureus) | 0.8355 | <0.10 |
| Enterotoxin B (S.aureus) | 0.9488 | <0.10 |
| Cockroach | 0.8575 | <0.10 |
ASSESSMENT OF IMMUNE COMPLEXES IN THE SERUM
| IgG4 | IgE | PP Index | |||
| PEG-Treated | Untreated | PEG-Treated | Untreated | ||
| Serum mg/dL | Serum mg/dL | Serum mg/dL | Serum mg/dL | % | |
| Present case | 1,460 | 3,110 | 237 | 376 | 63 |
| Control 1 | 5.5 | 20.9 | 160 | 1880 | 8.5 |
| Control 2 | 7.2 | 52.6 | 122 | 1960 | 6.2 |
DISCUSSION
REFERENCES
- Morgan WS, Castleman BA (1953) A clinicopathologic study of Mikulicz’s disease. Am J Pathol 29: 471-503.
- Yamamoto M, Takahashi H, Sugai S, Imai K (2005) Clinical and pathological characteristics of Mikulicz’s disease (IgG4 related plasmacytic exocrinopathy). Autoimmun Rev 4: 195-200.
- Takano K, Yamamoto M, Takahashi H, Shinomura Y, Imai K, et al. (2010) Clinicopathologic similarities between Mikulicz disease and Küttner tumor. Am J Otolaryngol 31: 429-434.
- Himi T, Takano K, Yamamoto M, Naishiro Y, Takahashi H (2012) A novel concept of Mikulicz’s disease as IgG4-related disease. Auris Nasus Larynx 39: 9-17.
- Takano K, Abe A, Yajima R, Kakuki T, Jitsukawa S, et al. (2015) Clinical evaluation of sinonasal lesions in patients with immunoglobulin G4-related disease. Ann Otol Rhinol Laryngol 124: 965-971.
- Inoue A, Wada K, Matsuura K, Osafune H , Ida Y, et al. (2016) IgG4-related disease in the sinonasal cavity accompanied by intranasal structure loss. Auris Nasus Larynx 43: 100-104.
- Lee YS, Cho HJ, Yoo HS, Shin YS, Park HS (2014) A case of IgG4-related disease with bronchial asthma and chronic rhinosinusitis in Korea. J Korean Med Sci 29: 599-603.
- Ohno K, Matsuda Y, Arai T, Kimura Y (2015) Nasal manifestations of IgG4-related disease: A report of two cases. Auris Nasus Larynx 42: 483-487.
- Umehara H, Okazaki K, Masaki Y, Kawano M, Yamamoto M, et al. (2012) Anovel clinical entity, IgG4-Related Disease (IgG4RD): General concept and details. Mod Rheumatol 22: 1-14.
- Ota Y, Fujiwara M, Hirabayashi Y, Kumasaka T, Takemura T, et al. (2015) Eosinophilic granulomatosis with polyangiitis demonstrating IgE-immune complexes and the possible involvement of IgE autoantibodies. Intern Med 54: 2687-2691.
Citation: Ota Y, Fujiwara M, Yoji H, Ota R, Suzuki M (2018) A Case of IgG4-Related Mikulicz’s Disease with Chronic Rhinosinusitis and Elevated Levels of IgG4-and IgE-Immune Complexes. J Otolaryng Head Neck Surg 4: 019.
Copyright: © 2018 Yasushi Ota, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.