Juvenile Nasopharyngeal Angiofibroma - Our Experience in a Tertiary Hospital
*Corresponding Author(s):
L Sudarshan ReddyDepartment Of Ent, Osmania Medical College, Hyderabad , India
Tel:+91 9885229954,
Email:drlsudarshanreddy9@gmail.com
Abstract
Nasopharyngeal angiofibroma is a highly vascular tumor seen typically in young adolescent males. It is an uncommon tumor accounting for less than 0.5% of all head and neck tumors. This peculiar tumor is thought to arise from the area of the sphenopalatine foramen and it classically presents with progressive nasal obstruction and epistaxis. This is a retrospective study of angiofibroma treated at our hospital over 3 years. The study discusses about clinical features, management options and the role of embolization in reducing the intra-operative blood loss.
Keywords
INTRODUCTION
MATERIALS AND METHODS
DISCUSSION
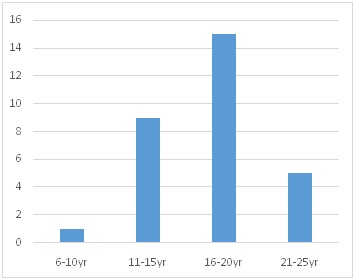
11-15 yrs [9]
16-20 yrs [15]
21-25 yrs [5]
| Symptoms/Signs | No. of Patients | Percentage (%) |
| (n=30) | ||
| Nasal obstruction | 22 | 73 |
| Epistaxis | 19 | 63 |
| Nasal discharge | 11 | 36 |
| Change in voice | 8 | 26 |
| Headache | 6 | 20 |
| Snoring | 5 | 16 |
| Face deformity | 3 | 10 |
| Cheek swelling | 2 | 6 |
| Intra-oral swelling | 2 | 6 |
The tumor was evaluated by clinical examination, diagnostic nasal endoscopy and imaging. CT scan of nose and paranasal sinuses was performed in all the cases. The extension of the tumor was studied. In our study we applied the Fisch staging system of the angiofibroma [2] (Table 2).
| Stage I | Tumor limited to nasopharyngealcavity, bone destruction negligible or limited to sphenopalatine foramen |
| Stage II | Tumor invading the pterygopalatine fossa or the maxillary, ethmoid or sphenoid sinus with bone destruction |
| Stage III | Tumor invading theinfratemporal fossa or orbital regiona) Without intracranial involvementb) With intracranial extradural involvement (parasellar) |
| Stage IV | Intracranial intradural involvementa) Without infiltration of the cavernous sinus, pituitary fossa or optic chiasmb) With infiltration of the cavernous sinus, pituitary fossa or optic chiasm |
The goal of tumor embolization is to occlude selectively the External Carotid Artery (ECA) feeders through intratumoral deposition of embolic material [3]. The embolic agents in common use are Polyvinyl Alcohol (PVA), Embospheres, liquid embolic agents (glue, Ethyl Vinyl Alcohol Copolymer [EVOH], or Onyx [ev3, Irvine, Calif]), gelatin sponge (Gelfoam; Phadia), and coils. The embolization is ideally performed 24 - 72 hours before surgical resection to allow maximal thrombosis of the occluded vessels and prevent recanalization of the occluded arteries or formation of collateral arterial channels (Figure 1).
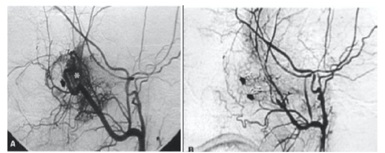
Figure 1: (a) Before embolization (tumor blush can be seen) (b) After embolization.
The most common complication of embolization is facial pain. Serious complications occur in less than two percent of patients. These are usually related to particle reflux, poor technique, or non-visualization of dangerous anastomoses resulting in blindness or irreversible neurologic deficits [4].
RESULTS
Among the 30 cases, pre-operative embolization with gelatin sponge (Gelfoam) was performed in 11 cases in our study. Embolization was performed by interventional radiologist 48 hrs prior to surgery in all the eleven patients. All the patients tolerated the procedure well, with 2 patients having facial pain for 2 days. Internal maxillary artery was the main feeding vessel in 9 patients and in 2 patients both internal maxillary and ascending pharyngeal artery.
Based on the clinical and imaging findings, surgery was planned for the all patients. Different approaches were used for the surgery depending on the extent of the tumor. Four main approaches used in this study are transpalatal, endoscopic, lateral rhinotomy and combined approach (Transpalatal and Endoscopic) [5]. Weber Ferguson approach was done in 2 patients.
Among the eleven embolized patients, the intraoperative blood loss in 4 patients was insignificant and in the rest of patients ranged from 250-700 ml with an average of 450 ml [3-5]. Embolization reduced intraoperative blood loss from an average of 950 ml in the non-embolized patients to 380 ml in the embolized patients. Lateral rhinotomy approach was performed in 6 patients, and this approach gives excellent exposure in infratemporal fossa involvement (Figure 2). Weber Ferguson approach was done in 2 patients with stage IIIa and IIIb.
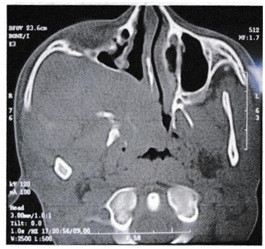
Figure 2: CT Scan showing extension of angiofibroma into the infratemporal fossa.
Endoscopic trans-nasal approach was performed in eleven patients [6,7]. This approach gives excellent exposure in stage I and stage II tumor (Figure 3).
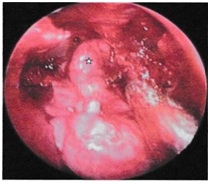
Figure 3: Endoscopic transnasal approach in a stage II tumor (embolized), intra-operative blood loss was insignificant.
All thirty patients were followed up for a period of two years (Table 3). Diagnostic nasal endoscopy was performed each month and a CT scans after one year. Recurrence was seen in three patients (case no. 3, 17 and 29) in the series. One patient who had been embolized and the other two, embolization was not performed. These patients presented with epistaxis after a mean duration of 4 months. All the three patients were later operated by endoscopic approach.
| Case No | Age | Pre-op stage | Embolization | Surgical approach |
| 1 | 24 | IIIa | No | Lateral Rhinotomy |
| 2 | 17 | II | No | Transpalatal |
| 3 | 16 | II | No | Endoscopic Transnasal |
| 4 | 21 | IIIb | No | Lateral Rhinotomy |
| 5 | 15 | II | No | Endoscopic Transnasal |
| 6 | 13 | II * | No | Lateral Rhinotomy |
| 7 | 16 | II | No | Endoscopic/Transpalatal |
| 8 | 18 | II | Yes | Endoscopic Transnasal |
| 9 | 20 | IIIa | Yes | Endoscopic/Transpalatal |
| 10 | 13 | II | Yes | Endoscopic |
| 11 | 20 | IIIa | Yes | Lateral Rhinotomy |
| 12 | 17 | I | No | Transpalatal |
| 13 | 1 | I | No | Transpalatal |
| 14 | 20 | II | Yes | Endoscopic Transnasal |
| 15 | 14 | IIIb | Yes | Weber-Ferguson |
| 16 | 20 | II | No | Endoscopic Transnasal |
| 17 | 16 | II | No | Transpalatal |
| 18 | 15 | IIIa | Yes | Endoscopic/Transpalatal |
| 19 | 13 | II | No | Endoscopic Transnasal |
| 20 | 15 | IIIa | No | Lateral Rhinotomy |
| 21 | 17 | I | No | Transpalatal |
| 22 | 17 | II | Yes | Endoscopic Transnasal |
| 23 | 19 | II | No | Endoscopic Transnasal |
| 24 | 22 | I | No | Endoscopic Transnasal |
| 25 | 18 | IIIa | Yes | Lateral Rhinotomy |
| 26 | 23 | II * | No | Transpalatal |
| 27 | 15 | II | No | Endoscopic Transnasal |
| 28 | 15 | IIIa | No | Weber-Ferguson |
| 29 | 9 | II | Yes | Endoscopic-Lateral Rhinotomy |
| 30 | 23 | II | Yes | Transpalatal |
Table 3: Summary of age group, pre-op stage, embolization and surgical approach in our study.
CONCLUSION
REFERENCES
- Panda NK, Gupta G, Sharma S, Gupta A (2012) Nasopharyngeal Angiofibroma-changing Trends in the Management. Indian J Otolaryngol Head Neck Surg 64: 233-239.
- Fisch U (1983) The infratemporal fossa approach for nasopharyngeal tumors. Laryngoscope 93: 36-44.
- Lv MM, Fan XD, Su LX, Chen D (2013) Preoperative direct puncture embolization of advanced juvenile nasopharyngeal angiofibroma in combination with transarterial embolization: an analysis of 22 consecutive patients. Cardiovasc Intervent Radiol 36: 111-117.
- Trivedi M, Desai RJ, Potdar NA, Shinde CA, Ukirde V, et al. (2015) Vision Loss due to Central Retinal Artery Occlusion Following Embolization in a Case of a Giant Juvenile Nasopharyngeal Angiofibroma. J Craniofac Surg 26: 51-53.
- Boghani Z, Husain Q, Kanumuri V, Khan MN, Sangvhi S, et al. (2013) Juvenile nasopharyngeal angiofibroma: a systematic review and comparison of endoscopic, endoscopic-assisted, and open resection in 1047 cases. Laryngoscope 123: 859-869.
- Andrade NA, Pinto JA, Nebrega MO (2007) Exclusively endoscopic surgery for juvenile nasopharyngeal angiofibroma. Otolaryngol Head Neck Surg 137: 492-496.
- Petruson K, Rodriguez-Catarino M, Petruson B, Finizia C (2002) Juvenile nasopharyngeal angiofibroma: long-term results in preoperative embolized and non-embolized patients. Acta Otolaryngol 122: 96-100.
Citation: Khan MM, Reddy LS, Reddy NV, Swamy DR (2017) Juvenile Nasopharyngeal Angiofibroma - Our Experience in a Tertiary Hospital. J Otolaryng Head Neck Surg 3: 008.
Copyright: © 2017 M Mujtaba Khan, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.