
Antioxidant and Antihypertensive Effect of Azadirachta Excelsa Leaf Extract in Spontaneously Hypertensive Rat (SHR) Model
*Corresponding Author(s):
Hafandi ADepartment Of Veterinary Preclinical Sciences, Faculty Of Veterinary Medicine, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor Darul Ehsan, Malaysia
Tel:+60 386093416,
Email:hafandi@upm.edu.my
Abstract
A research was carried out to evaluate the antioxidant activities of Azadirachta Excelsa and its antihypertensive effect in Spontaneously Hypertensive Rat (SHR). The Total Phenolic Content (TPC) and Total Flavonoid Content (TFC) was quantified and IC50 level of A. excelsa was determined. For the antihypertensive effect, the rats were randomly assigned into four treatment groups as followed: Group I (normotensive control from Wistar-Kyoto rats), Group II (hypertensive control from SHR), Group III (SHR receiving 250 mg/kg of A. excelsa extract), and Group IV (SHR receiving 40 mg/kg of captopril). The Systolic Blood Pressure (SBP) of these animals was performed by tail-cuff method. The average of TPC and TFC was 202 + 0.42 mg Gallic Acid Equivalent (GAE)/g extract and 198+0.67 mg rutin equivalent/g extract, respectively. Meanwhile, the IC50 value of free radical scavenging activity was about 308 μg/ml. The systolic blood pressure level of the SHR treated with A.excelsa significantly reduced (153 mmHg; P <0.05) compared to the untreated SHR control (187 mm Hg; Group II). In conclusion, we found that A. excelsa extract at a dose of 250 mg/kg possesses phenolic properties that can be used as a potential treatment for hypertension due to its high antioxidant activities.
Keywords
INTRODUCTION
Azadirachta Excelsa or locally known as “sentang” from Meliaceae family, where it belongs to a plant family that is known to have various benefits associated to medical properties. Various parts of A. excelsatree have been used for traditional medicine due to its biological activity such as antiseptic, anti-inflammatory, antimicrobial and antifeedant [8]. Subsequently, there was a finding reported that this plant had a great potential as medicinal plants due to the presence of phenolic and flavonoid as well as antioxidant compounds [9] although the compound have not been characterized yet. However, there were only a few findings about the effect of A. excelsa extract on mammals.
Therefore this study was conducted to evaluate the antioxidant activity by quantifying the total phenolic and flavonoid contents, followed by the determination of free radical scavenging activity of A. excelsaethanolic extract. The quantitative determination of total antioxidant capacity was conducted to investigate the relationship between dietary antioxidants and pathologies induced by the oxidative stress [10]. In addition, the evaluation of blood pressure on experimental hypertension rats model was measured to investigate the potential antihypertensive effects of this plant.
MATERIALS AND METHODS
Preparation of A.excelsa leaves extract
Determination of total phenolic, total flavonoid content and antioxidant activity by 2,2-Diphenyl-1-Picryl Hydrazyl (DPPH) assay
Free radical scavenging activity (%) = [(A0 - A1)/ A0] × 100
Where A0 was the absorbance of the blank and A1 was the absorbance of the sample. A curve of percent scavenging effect against samples concentrations was plotted and the concentration of the sample required from 50% inhibition was determined as IC50. Ascorbic acid and α-tocopherol were used as the standard references antioxidants.
Preparation of animal model and experimental design on the antihypertensive effect of A.excelsa extract in SHR
Measurement of Systolic Blood Pressure (SBP)
STATISTICAL ANALYSIS
RESULTS
Phenolic, flavonoid content and antioxidant activity
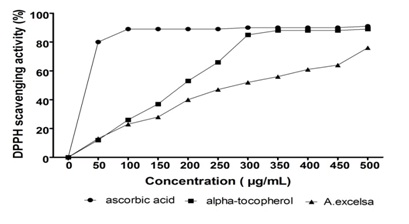 Figure 1: DPPH free radical scavenging activity of A. excelsa ethanolic extract and reference antioxidants as concentration increase.
Figure 1: DPPH free radical scavenging activity of A. excelsa ethanolic extract and reference antioxidants as concentration increase.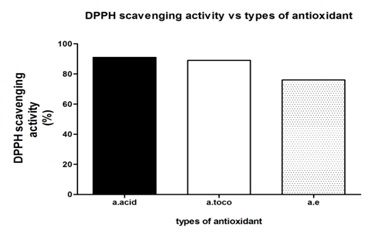 Figure 2: Scavenging activities (%) of different types of antioxidant at 500 μg/ml.
Figure 2: Scavenging activities (%) of different types of antioxidant at 500 μg/ml.| Ethanolic leaf extract of A.excelsa | |
| Total Phenolic Content | 202 + 0.42 mg gallic acid/g extract |
| Total Flavonoid Content | 198 + 0.67 mg rutin /g extract |
| IC50 | 308 μg/ml |
Effect of A.excelsa extract on systolic blood pressure in SHR model
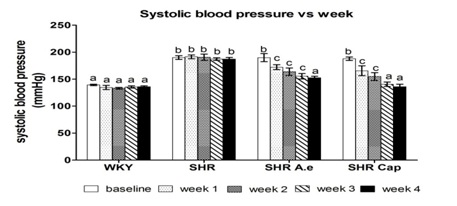 Figure 3: Values are expressed as mean ± SEM (n=6). a,b,c Values with different alphabets are significantly different at P<0.05 between group. WKY control: (WKY), SHR control: (SHR), SHR received 250 mg/kg of Azadirachta Excelsa: (SHR A.e) and SHR received 40 mg/kg of captopril: (SHR cap).
Figure 3: Values are expressed as mean ± SEM (n=6). a,b,c Values with different alphabets are significantly different at P<0.05 between group. WKY control: (WKY), SHR control: (SHR), SHR received 250 mg/kg of Azadirachta Excelsa: (SHR A.e) and SHR received 40 mg/kg of captopril: (SHR cap).DISCUSSION
The result on SBP of animals showed that oral administration of A.excelsa prevented the prolonged of hypertension in SHR model. It was established that the persistent elevated blood pressure in these genetic hypertension animal model was due to an excessive production of ROS in arterial wall where the major source is NADPH oxidase [15]. Hence, any compounds that can ameliorate the oxidative stress by inhibiting an excessive ROS production seem to be the therapeutic interest in lowering the blood pressure. An antioxidant activity of A.excelsa might promoted its antihypertensive effect in animal model similar to the antihypertensive drug captopril. However, captopril served a good ability in normalizing blood pressure better due to its antioxidant and cardioprotective effect [16]. In blood pressure regulation, captopril acts as an Angiotensin Converting Enzyme Inhibitor (ACEI) that inhibits the production of Angiotensin II, a potent vasoconstrictor in Renin-Angiotensin System (RAS). While the protective effect of phenolics and flavonoids not only as an antioxidant, but also as an antithrombotic, antiischemic and vasorelaxant in heart disease [17]. Thus, this plant has a great potential as medicinal plants due to the presence of phenolics and flavonoids as well as antioxidant compounds.
The present study showed that the quantification on total phenolic and flavonoid content present in A. excelsa ethanolic leaf extract. In addition, this study suggested that the antioxidant activity of A. excelsaalleviate hypertension in SHR model. To the best of our knowledge, this is the first study reporting of A. excelsa on its antioxidant and antihypertensive activity. Hence, further isolation, characterization and purification of the active constituents and experimentation would be necessary to elucidate the exact mechanism of A.excelsa as a plant with antioxidant and antihypertensive effect.
ACKNOWLEDGEMENT
REFERENCES
- Kotsis V, Stabouli S, Papakatsika S, Rizos Z, Parati G (2010) Mechanisms of obesity-induced hypertension. Hypertens Res 33: 386-393.
- Rodrigo R, González J, Paoletto F (2011) The role of oxidative stress in the pathophysiology of hypertension. Hypertens Res 34: 431-440.
- Hirooka Y (2011) Oxidative stress in the cardiovascular center has a pivotal role in the sympathetic activation in hypertension. Hypertens Res 34: 407-412.
- Fanelli C, Zatz R (2011) Linking oxidative stress, the renin-angiotensin system, and hypertension. Hypertension 57: 373-374.
- Khalil MI, Sulaiman SA (2010) The potential role of honey and its polyphenols in preventing heart diseases: a review. Afr J Tradit Complement Altern Med 7: 315-321.
- Halvorsen BL, Holte K, Myhrstad MC, Barikmo I, Hvattum E, et al. (2002) A systematic screening of total antioxidants in dietary plants. J Nutr 132: 461-471.
- Pisoschi AM, Negulescu GP (2011) Methods for total antioxidant activity determination: A review. Biochem & Anal Biochem 1: 106.
- Akhtar Y, Yeoung YR, Isman MB (2008). Comparative bioactivity of selected extracts from Meliaceaeand some commercial botanical insecticides against two noctuid caterpillars, Trichoplusia ni and Pseudaletia unipuncta. Phytochemical Reviews 7: 77-88.
- Shafie NI, Samsulrizal N, Sopian NF, Rajion MA, Meng GY, et al. (2015) Qualitative phytochemical screening and GC-MS profiling of Azadirachta Excelsa leaf extract. Malaysian Applied Biology 44: 87-92.
- Serafini M, Bellocco R, Wolk A, Ekström AM (2002) Total antioxidant potential of fruit and vegetables and risk of gastric cancer. Gastroenterology 123: 985-991.
- Chan KW, Khong NMH, Iqbal S, Mansor SM, Ismail M (2013). Defatted Kenaf Seed Meal (DKSM): Prospective edible flour from agricultural waste with high antioxidant activity. Food Science and Technology 53: 308-313.
- Kim H, Moon JY, Kim H, Lee DS, Cho M, et al. (2010) Antioxidant and antiproliferative activities of mango (Mangifera indica L) flesh and peel. Food Chemistry 121: 429-436.
- Sithisarn P, Carlsen CU, Andersen ML, Gritsanapan W, Skibsted LH (2007) Antioxidative effects of leaves from Azadirachta species of different provenience. Food Chemistry 104: 1539-1549.
- Maisuthisakul P, Suttajit M, Pongsawatmanit R (2007) Assessment of phenolic content and free radical-scavenging capacity of some Thai indigenous plants. Food Chemistry 100: 1409-1418.
- Suzuki H, Swei A, Zweifach BW, Schmid-Schönbein GW (1995) In vivo evidence for microvascular oxidative stress in spontaneously hypertensive rats. Hydroethidine microfluorography. Hypertension 25: 1083-1089.
- Bolterman RJ, Manriquez MC, Ortiz Ruiz MC, Juncos LA, Romero JC (2005) Effects of captopril on the renin angiotensin system, oxidative stress, and endothelin in normal and hypertensive rats. Hypertension 46: 943-947.
- Jendekova L, Kojsova S, Andriantsitohaina R, Pechanova O (2006) The time-dependent effect of Provinols on brain NO synthase activity in L-NAME-induced hypertension. Physiol Res 55: 31-37.
Citation: Nur Farhana AS, Nurul Izzati S, Nurdiana S, Rajion MA, Goh YM, et al. (2016) Antioxidant and Antihypertensive Effect of Azadirachta Excelsa Leaf Extract in Spontaneously Hypertensive Rat (SHR) Model. J Altern Complement Integr Med 2: 013.
Copyright: © 2016 Nur Farhana AS, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

