
Single Blind Randomized Controlled Trial of ‘Azathioprine Versus Corticosteroids in Parthenium Hysterophorus Induced Contact Dermatitis
*Corresponding Author(s):
Smitha PrabhuDepartment Of Dermatology And Venereology, Kasturba Medical College, Manipal University, Karnataka, India
Abstract
Keywords
INTRODUCTION
So far, corticosteroids along with other measures like protection from antigen, photoprotection, frequent washing of exposed area and removing the plant from area of residence, has been the mainstay of treatment. Prolonged corticosteroid intake in a chronic though, benign disease may lead to various serious and even life-threatening systemic side effects. Azathioprine is a 6-mercaptopurine derivative which inhibits purine synthesis and acts as an immunosuppressive and a powerful anti-inflammatory agent [2]. It inhibits T helper cells which are primarily responsible for the dermatitis in ABCD and has been successfully tried in Chronic Actinic Dermatitis (CAD) [3]. Effectiveness of Azathioprine in the treatment of ABCD from sesquiterpene lactones has been reported [4]. In earlier preliminary studies we have found that 50-100 mg oral daily dose of Azathioprine induces a clinical remission without significant clinical or biochemical side effects in patients with ABCD to Parthenium [5,6].
This study is undertaken to compare the efficacy of azathioprine with that of corticosteroids, and to evaluate the side effects.
Aims
- To study the effectiveness of azathioprine and corticosteroids in patients with Parthenium hysterophorus induced airborne contact dermatitis.
- To monitor the side effects of the drugs both clinically and biochemically.
Materials and methods
Inclusion criteria
- All patients with Parthenium induced air-borne contact dermatitis, diagnosed clinically and proven by positive patch tests with Parthenium.
- Age group 20 to 80 years.
Exclusion criteria
- Women in childbearing age group who have not completed their family.
- Pregnancy and lactation.
- Patients with gastrointestinal problems like gastritis, peptic ulceration, hiatus hernia, liver or renal impairment.
- Patients whose baseline investigations revealed hematological abnormalities of liver or renal function.
- Patch test positivity to allergens in the Indian Standard Series (ISS).
- Intake of other drugs likely to affect the disease, e.g., medicines of alternate system.
Randomization procedure
Procedure: All clinically diagnosed patients were patch tested with Parthenium antigen, 0.5% and 1% in petrolatum. The antigen was prepared from freshly collected Parthenium plant in the Department of Pharmacognosy, College of Pharmaceutical Sciences (COPS), Manipal, Karnataka and India. The plant identification and confirmation was done by experienced taxonomist. Solid-liquid extraction technique by Soxhlet extraction method was used to make the patch test antigen. Dilutions of 0.5% and 1% of the extract in petrolatum were prepared.
Treatment procedure: Thirty four severely affected patients with features of Parthenium dermatitis, patch tested positive for Parthenium (and other allergens excluded by patch test) and satisfying the inclusion criteria were taken for the study. Detailed history was noted on the proforma after an informed content. Basic investigations included complete haemogram, urine and stool routine examination, fasting and postprandial blood sugars, renal and liver function tests, serum electrolytes, chest radiograph and ECG.
After randomization, Group A received Azathioprine 50 mg twice daily for 6 months and group B, prednisolone in tapering doses of 30 mg for 2 weeks, 20 mg for 2 weeks, and 10 mg for 4 weeks, thereafter oral placebo for next 4 months. The side effects were monitored at monthly intervals. After 6 months, the patients were followed up for the next 6 months. Antihistaminics (pheniramine maleate 25 mg twice daily) were given based on need.
- The patients on steroids were given antacids. All patients were given topical betamethasone valerate once daily.
- Clinical assessment was undertaken monthly for 12 months.
- Disease activity was evaluated using Dermatitis Assessment Severity Index (DASI).
- Itch was scored on a Visual Analogue Scale (VAS) 0-100 mm recorded graphically.
- Sleep loss was recorded in percentage graphically.
General management: In addition, patients in both groups were advised use of protective clothing and avoidance of sun; frequent washing of exposed areas and application of topical steroids and oral administration of antihistaminics if itching was severe.
Patients who developed side effects of the drug were given standard out or in patient care as appropriate. Patients who developed an exacerbation of their disease while on study were withdrawn and given other appropriate therapy to control the disease.
Statistical analysis: was done using chi-square test, paired samples test, independent samples test and nonparametric tests.
RESULTS
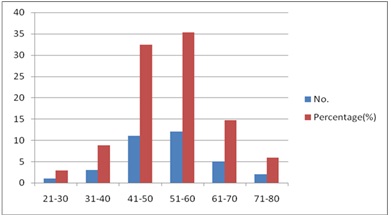
Chasrt 1: Age distribution.
Agriculturists were the maximum affected (64.7%), followed by manual laborers and house wives. All the patients were from various districts of Karnataka state, most of them from Davangere (12 patients, i.e., 35.3%), where Parthenium is rampant. 16 patients (47%) complained of aggravation in summer, followed by 5 patients (14.7%) who had aggravation in winter.
Twenty-eight patients (82.4%) gave history of direct contact with the plant. Thirty-two (94.1%) had history of exacerbations and remissions. A very significant number of patients (32, i.e., 94.1%) gave history of photo aggravation. Two patients (5.9%) had atopic background in the form of allergic rhinitis and family history of asthma. The most common associated disorders observed were diabetes mellitus in 6 (17.65%) and hypertension in 5 (14.7%). There were 12 smokers, 8 people who consumed alcohol and 4 betel nut chewers, but none attributed their aggravation to their habits. Previous treatment modalities: Majority were treated with topical steroids. (28 patients, i.e., 82.4%), antihistamines (67.7%) and systemic steroids (41.2%). Other modalities included emollients, azathioprine, ayurvedic and homeopathic medicines, sunscreens and hydroxychloroquine.
The most common pattern of presentation in this study was that of ABCD type in 31 patients (91.2%) involving photoexposed areas plus eyelids, ear creases, naso-labial folds, retro-auricular area, cubital and popliteal fossae.
Other presentations included photo dermatitis in 3 patients, Actinic reticuloid in 2 and hand dermatitis in one (Table 1).
| Pattern | Number (n=34) | Percentage (%) |
| ABCD | 31 | 91.2 |
| Photodermatitis | 3 | 8.8 |
| ABCD with hand dermatitis | 1 | 2.9 |
| ABCD with lichenoid pattern | 1 | 2.9 |
| CAD/Actinic Reticuloid pattern | 2 |
5.9 |
Table 1: Pattern of dermatitis.
Extent of involvement
Treatment
| Group | Completed | Dropped out |
| Azathioprine (n=17) | 7 (41.2%) | 10 (58.8%) |
| Prednisolone (n=17) | 13 (76.5%) | 4 (23.5%) |
In the Azathioprine group, one patient (14.3%) had complete remission at the end of six months, 5 patients (71.4%) had significant improvement and one patient (14.3%) initially improved, later worsened. Six out of 7 patients who completed treatment showed more than 50% improvement after 3 months of treatment (Table 3).The patient with complete remission did not relapse even after 6 months of follow up. One patient relapsed 18 months after stopping treatment and another relapsed within 2 months. An average of 2-18 months of relapse free period after stopping Azathioprine treatment was observed in this study. Clinical response to Azathioprine was visible by 2 months in all patients. All experienced mild worsening of dermatitis on exposure to sunlight and outdoor work. In the prednisolone group, out of 13 patients who completed the treatment, 8 had significant improvement (Table 3).
| Percentage of reduction in DASI | Azathioprine group | Prednisolone group | ||
| No. (n=7) | Percentage (%) | No. (n=13) | Percentage (%) | |
| Complete remission (100%) | 1 | 14.3 | 3 | 23.07 |
| 80-90% | 3 | 42.9 | 3 | 23.07 |
| 50-80% | 2 | 28.6 | 2 | 15.4 |
| Initial improvement followed by worsening | 1 | 14.3 | 5 | 38.5 |
Table 3: Percentage of reduction in DASI after treatment in Azathioprine group.
Side effects
Statistical analysis
DISCUSSION
Parthenium dermatitis is now rampant in India, especially in pockets in the north and southwest. It usually affects adult males, earlier studies showing a male: female incidence of 4.55:1 [7]. Our study showed a ratio of 7.5:1, probably because women tend to remain indoors more often, reducing the risk for exposure and sensitization. The influence of age in the study was striking, the maximum number of affected cases being in the group of 41-60 years, unlike other studies which observed 30-39 years[7]. The duration of the disease in this study ranged from 6 months to 20 years which shows the chronic nature of Parthenium dermatitis. Majority of the affected patients were invariably exposed to the allergen most of the times and the severity of the dermatitis depends not only on irritant or allergic potential of the plant, but also on degree of exposure and individual sensitivity. The principle source of exposure was direct contact with the living plant. The plant hairs (trichromes) contain surplus of dermatitis producing sesquiterpene lactones. The airborne pollen also contains the oleoresin leading to ABCD. The plants crumble during summer season to a finely scattered dust, which virtually fills the surrounding atmosphere and forms another source of ABCD [8]. Lack of sun protection, working and hygienic habits may also contribute to chronicity of the disease. Clothes dried outside in sun may also act as a potential trap for airborne Parthenium allergens and on sweating the allergens leach out on to the skin to produce a chronic recurrent dermatitis. One of our patients had onset of dermatitis following wearing a shirt which was kept to dry in open air on a Parthenium plant.
Most complain exacerbation in summer, may be due to sweating as well as photosensitive nature of the plant. Photo contact dermatitis to Parthenium has been reported earlier [9], but we were unable to prove it as the Parthenium antigen showed equal reactions on irradiated as well as non-irradiated sites on photo patch testing (unpublished observation). Exposure to airborne plant allergens may first cause ABCD which mimics photo dermatitis, but subsequently develop true photo dermatitis like Chronic Actinic Dermatitis (CAD) or Actinic Reticuloid (AR) syndrome.
Earlier studies have shown that reduction of MED to UVB is a definite indicator of photosensitivity in Parthenium dermatitis [10]. It is possible that retention of photo allergen at site of repetitive photoallergic reactions can cause classic photo allergic reactions with photo antigen trapped in the skin; the reactions lasting for months to years. Majority of our patients were middle to old aged, hence explaining the long term influences of the allergen.
Major aggravating factors in this study were the 3 ‘S’s - sunlight, summer and sweating. 82.2% had direct contact with the plant during their work, and the rest may have developed the disease due to the air borne nature of Parthenium dermatitis.
The various patterns of dermatitis we observed were airborne contact dermatitis, photo dermatitis, hand dermatitis, lichenoid and chronic actinic reticuloid pattern.
Frequent concurrence of atopic dermatitis with Compositae dermatitis has been reported in some earlier studies [11], but not in our study. Increased incidence of Parthenium dermatitis in atopics may be due to the chronically irritated and damaged skin which makes them susceptible to sensitization of any nature.
The efficacy of Azathioprine in Parthenium dermatitis has been shown earlier, so also its relapsing tendency on stopping medication [6,12]. Azathioprine is a 6-Mercaptopurine analogue which acts by interfering with DNA and RNA synthesis and repair. It has a greater effect on T cells than B cells, and also on Langerhans cells, NK cells and monocytes. Azathioprine inhibits the effector phase of immune response, but not the induction phase. The adverse effects include nausea, vomiting, diarrhea, abdominal pain, pancytopenia, hepatotoxicity, pancreatitis, polyneuropathy, drug rash and fever, as well as increased incidence of malignancies, especially in transplant patients on long term treatment.
In the present study, a large number of drop outs (58.8%) in the Azathioprine group may be due to the delay in onset of action and poor control of itch. We were unable to contact the dropouts and enquire the exact reason for the same. Though corticosteroids act faster, Azathioprine has a ‘steroid sparing effect’ leading to lower dosage of steroids and thus a lesser risk of serious steroid induced adverse effects while maintaining an adequate immunosuppressive effect.
In our study, prednisolone group generally had good initial improvement, but there was no long lasting remission whereas Azathioprine group had relatively better stable disease free period ranging from 2 to 15 months after stopping the drug. The non significance of p-value may be due to the higher number of dropouts in the Azathioprine group. The shorter duration of steroid schedule was safe enough to avoid serious side effects but not adequate enough for prolonged control of the dermatitis. Relapsed patients who were started on oral mini pulse steroid therapy (betamethasone 6 mg weekly once or twice) responded very well with longer remissions and least number of side effects.
The medical management of Parthenium dermatitis will be useless unless the patient is seriously motivated for proper preventive measures. The most effective treatment would be to fully remove the patient from Parthenium infested areas, but as this is impractical in most cases, we need to focus on other points like removal of plant from the patient’s environment as far as possible, frequent washing of exposed areas with soap and water before the antigen penetrates the skin, use of barrier cream, frequent changes and washing of used clothes, limiting sun exposure, drying of washed clothes indoors etc. We are also attempting to devise an acceptable mask to be worn by severely affected people when out in the open.
The statistical significance to confirm the superiority of either of the drugs in terms of improvement in DASI, itch and sleep was undermined by the large number of dropouts in the Azathioprine group, which clinically had an edge over the prednisolone group in terms of prolonged remission. Clinically the beneficial effects of Azathioprine were very obvious, with a progressively steady rise in improvement of scores in all assessment parameters, a stable post treatment period and a better patient compliance. Prednisolone, on the other hand, demonstrated a quicker onset of action, but a quicker relapse as well.
We would like to conclude that Azathioprine gives lasting results, a stable prolonged relapse free period and is suitable for a chronic problem like Parthenium dermatitis, but has to be used with caution with proper monitoring. It is imperative that the patients be counselled regarding the nature of their condition, the slow onset of action of azathioprine and the need for lifestyle changes and proper medication and photoprotection. Prednisolone is ideal for acute severe flare ups as a short course therapy, with an early withdrawal for avoiding the associated morbidities.
REFERENCES
- Lonkar A, Jog MK (1968) Dermatitis caused by a plant Parthenium Hysterophorus. A preliminary report. Ind J Dermatol Venereol Leprol 34: 194-196.
- Ahmed AR, Moy R (1981) Azathioprine. Int J Dermatol 20: 461-467.
- Leigh IM, Hawk JL (1984) Treatment of chronic actinic dermatitis with azathioprine. Br J Dermatol 110: 691-695.
- Roed-Petersen J, Thomsen K (1980) Azathioprin in the treatment of airborne contact dermatitis from compositae oleoresins and sensitivity to UVA. Acta Derm Venereol 60: 275-277.
- Verma KK, Pasricha JS (1996) Azathioprine as a corticosteroid sparing agent in air-borne contact dermatitis. Indian J Dermatol Venereol Leprol 62: 30-32.
- Srinivas CR, Balachandran C, Shenoi SD, Acharya S (1991) Azathioprine in the treatment of Parthenium dermatitis. Br J Dermatol 124: 394-395.
- Tiwari VD, Sohi AS, Chopra TR (1979) Allergic contact dermatitis due to Parthenium Hysterophorus. Ind J Dermatol Venereol Leprol 45: 392-400.
- Lonkar A, Mitchell JC, Calnan CD (1974) Contact dermatitis from Parthenium Hysterophorus. Trans St Johns Hosp Dermatol Soc 60: 43-53.
- Bhutani LK, Rao DS (1978) Photocontact dermatitis caused by Parthenium Hysterophorus. Dermatologica 157: 206-209.
- Srinivas CR, Shenoi DS (1994) Minimal erythema dose to ultra-violet light in Parthenium dermatitis. Ind J Dermatol Venereol Leprol 60: 149-150.
- Epstein S, Mohajerin AH (1964) Incidence of contact sensitivity in atopic dermatitis. Arch Dermatol 90: 284-287.
- Sharma VK, Chakrabarti A, Mahajan V (1998) Azathioprine in the treatment of Parthenium dermatitis. Int J Dermatol 37: 299-302.
Citation: Davis SV, Shenoi SD, Prabhu S, Pai S, Balachandran BC (2015) Single Blind Randomized Controlled Trial of ‘Azathioprine Versus Corticosteroids in Parthenium Hysterophorus Induced Contact Dermatitis’. J Clin Dermatol Ther 1: 004.
Copyright: © 2014 Suraj V Davis, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

