Non-Invasive Diagnostic Methods: How Have They Modified the Therapeutical Indication?
*Corresponding Author(s):
Pier Luigi AntignaniVascular Centre, Nuova Villa Claudia, Rome, Italy
Tel:+39335318430,
Email:antignanipl@gmail.com
Abstract
The new tools to carry out researches into the vascular diseases modified the approach to define precise diagnosis and consequently, the therapy [1-5].
The duplex scanning is useful for the decisions of medical, endovascular or surgical treatment on young and old patients, easy to repeat non-invasive, if performed by an expert operator and a good instrument [5]. We will consider some of the most frequent pathologies:
Keywords
EDITORIAL
The duplex scanning is useful for the decisions of medical, endovascular or surgical treatment on young and old patients, easy to repeat non-invasive, if performed by an expert operator and a good instrument [5]. We will consider some of the most frequent pathologies:
1. Carotid stenosis
2. Abdominal aortic aneurysm
3. Venous thrombosis
4. Chronic venous insufficiency
CAROTID STENOSIS
One of the most frequent uses is the evaluation of the carotid arteries in patients presenting risk factors like; age, diabetes, heart disease, hypertension, other vascular diseases and autoimmune diseases. The evaluation for the treatment and the follow up in symptomatic or asymptomatic patients can be done exclusively on the report of the duplex scanning; only few cases need more investigation. The classification of the risk for stroke, connected to the plaque presence is calculated in conformity to the degree of the stenosis, morphology and echogenicity of the plaque.
The degree of the stenosis can be calculated in 3 ways; in size of diameter, in cross-sectional area and by the flow velocity; the last one is important especially in case of kinking or presence of stent. We have to report the method that we use, NASCET or ECST [4,5].
The morphological characteristics are (Figures 1-5):
• Thickness in mm
• Echogenicity of the plaque: hypoechoic or hyperechoic; it depends on the percentage of calcium inside the plaque
• Structure: homogeneous or heterogeneous; It shows the organization of the molecules inside the plaque, intraplaque haemorrhage
• Surface: regular, irregular, ulcerated: the last one is more dangerous because of the possibility of embolization
• Fibrous cup
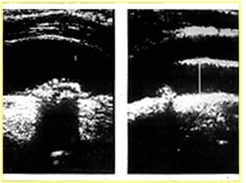
Figure 1: Echolucent plaque in carotid artery; Heavy calcification.
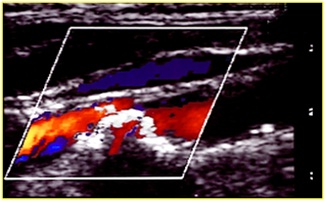
Figure 2: Echo color duplex: Echolucent plaque in carotid artery.
Figure 3: Uniform echolucent plaque in carotid artery.
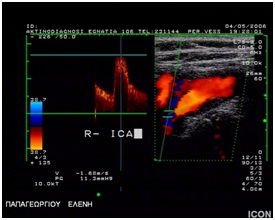
Figure 4: Carotid stenosis 50%.

Figure 5: GSM 17.
Regarding the echogenity, the plaque presents five possibilities:
• Type I (uniformly echolucent)
• Type II (predominately echolucent)
• Type III (predominately echogenic)
• Type. IV (uniformly echogenic)
• Type V (heavy calcification)
Guidelines of Italian Society for Vascular Investigation [5]: The color flow duplex scanning represents the investigation of choice for the diagnosis, measurement, vulnerability of carotid stenosis; surface ulceration, low GSM (<25), heterogeneous appearance of the plaque and the juxta-luminal location of the echolucent area after image normalization, are ultrasonographical indicators of plaque vulnerability and should be considered in the selection of appropriate therapy and the frequency of follow up.
Duplex scanning allows evaluating the follow up: surgical outcome, medical efficacy treatment, new stenosis. We should consider that after stent application, velocity values must be interpreted differently.
Sometimes, when the stroke is unexplained by the carotid’s plaque, hearth pathologies or other risk factors, we have to individuate other risk factors using the angio RM/TC, transcranial echo duplex or the more invasive angiography method to evaluate the intracranial vessel anatomy and presence of lesions and coexisting aortic arch lesions.
The Diffusion-Weighted magnetic resonance Imaging (DWI) represents the less invasive and fast method for the evaluation of the ischemic lesions, but sometimes we have to use the angio RM/TC because it shows better the calcification of the vessels.
The transcranial echo duplex is less used because finding experienced operators is not easy, which allows us to acquire more details for surgical CE/CAS decisions (Figure 6):
• Coexisting lesions of intracranial vessels
• Circle of Willis efficiency
• Intracranial hemodynamic effects of extra-cranial reserve
• Cerebrovascular reserve
• Micro-embolic events due to ulcerated plaques
• Cross clamping risk and indication for shutting
• Indication for surgery in asymptomatic subjects or in patients with bilateral carotid lesions
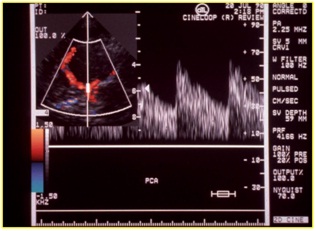
Figure 6: Evaluation with echo color duplex of the Willis’ circle. The flow is in cerebral media artery.
According to the results of non invasive methods, we can put the therapeutical indications and, in conclusion, surgical CE could be better in patients with these conditions:
• Long multifocal lesions
• Echolucent plaque
• Severe ulceration
• Heavy circumferential calcifications of carotid bifurcation
• Severe tortuosities
• Extensive aortic or brachiocephalic trunk lesions
• If a clot is suspected
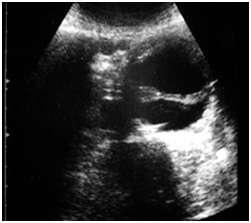
ABDOMINAL AORTIC ANEURYSM [1,3] (FIGURE 7)
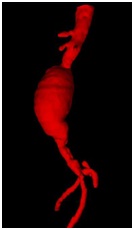
It is ideal for the follow up of the growth rate that is the criteria for the therapy. A growth rate > 0.7 cm per six months or 1 cm per years has been suggested as a threshold for proceeding to surgery, irrespective of size.
The evaluation with the color flow duplex imaging has to consider both morphology and hemodynamic looks.
Morphology: The endovascular wall thrombus, ulceration and calcification, true vessel lumen, size and longitudinal extension (Figures 8&9).
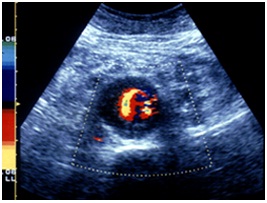
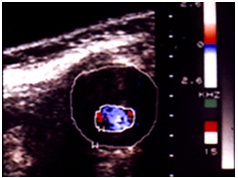
Hemodynamics: decrease of flow velocity, color mosaic imaging (Figure 10).
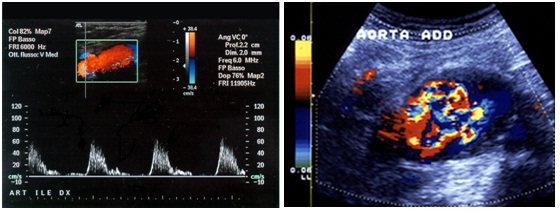
If we are in front an acute condition, we must look for the following aspects in figures 11,12 & 13:
• Wall dissection
• Wall rupture
• Rapid growth
• Acute thrombosis
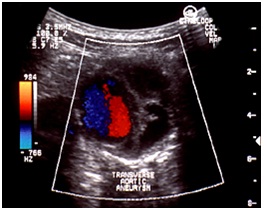
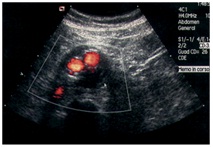
During the controls after the endovascular treatment, we have to evaluate these parameters (Figures 14, 15 &16):
• Position and patency of endograft (it can immigrate from the just place)
• Diameters and pulsatility of aneurysm
• Endoleak (sensitivity 81-100%, specificity 74-99%) we have 3 types of endoleaks, with or without necessity for treatment
• Patency of other vessels (renal artery)
• Infections
• 6 fistulas (intestinal fistulas)
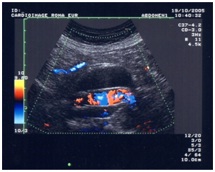
DEEP VENOUS THROMBOSIS (DVT)
The anamnesis of the patient, extend of the thrombosis, adequacy of collateral vein, severity of associated vascular pathologies and inflammation are important for the diagnosis and therapy. Surely, the color flow duplex scanning is the best tool for recognizing DVT, new thrombosis or differential diagnosis, like lymphedema, oedema post fracture or SPF. We note that the clinical diagnosis is inadequate because it represents only 30% of accuracy.
Color flow duplex scanning (Figures 16-22) can provide both morphologic and hemodynamic findings and represents now a quick test that gives us the necessary criteria for:
• Initial patient management, providing information about the morphology of the clot and the possibility to arrive to the resolution of the pathology.
• Identify the patients with a potential high risk for post thrombotic syndrome.
• To compare and evaluate the results of different regimens of anticoagulation and fibrinolytic drug therapy on the long-term outcome of venous thrombosis.
• Identify a new thrombosis case.
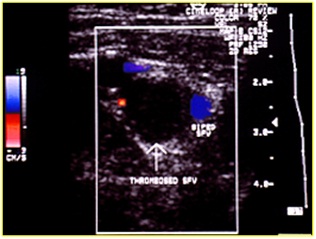
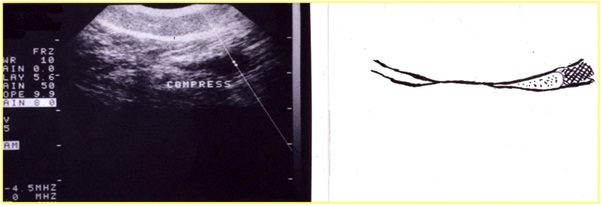
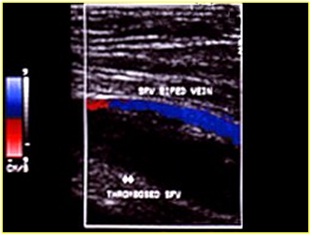
Figure 19: Recent vein thrombosis with hypoechoic thrombus.
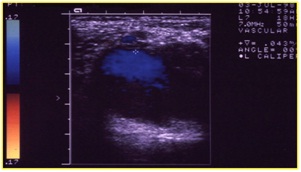
Figure 20: Anechoic thrombus; the color shows where the residual flow is.
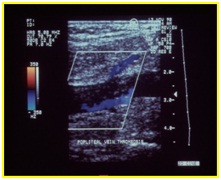
Figure 21: Recanalization of femoral vein thrombosis.
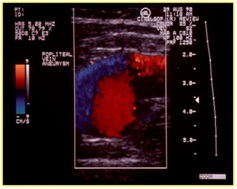
Figure 22: Reflux in post thrombotic syndrome.
The aspects that we have to look for are:
• The full or partial color inside the vein, mosaic color
• The compression manoeuvre, to identify the presence of any little part of thrombus, it has 100% accuracy
• It is important to know the morphologic characteristic and the localization of the thrombus to calculate the increase of the risk of EP
There are some coagulative factors that are more frequently associated to DVT [2]:
• Anti-phospholipid antibody (lupus anticoagulant or anti-cardiolipin antibody) is associated with a fold increase in risk of recurrent VTE
• Thrombophilic genetic disorders like alerted quality/quantity of AT III, protein C, protein S, factor V of Leidenn homozygous, elevated quantity or alteration of homocysteine, factor VIII coagulation (> 234 IU) caused with higher rates of DVT specially when associated risk factors are present (CA, pregnancy, long trips, obesity and diabetes)
• Heterozygous forms of factor V and G20210A prothrombin gene mutation confer relatively low increase risk for recurrent VTE
From our experience, 60% of prossimal deep veins pathology, develop post thrombotic syndrome within two years [1-3].
In 50% of the patients, stockings can reduce this complication. The importance of this behavior concerns avoiding recurrent thrombosis, better quality of life and less costs for the society. The pathophysiology that induces the development of PTS is not yet entirely understood. Probably, collateral factors like venous reflux, deep vein obstruction, calf muscle alteration, play a significant role in the presence and location of venous reflux and obstruction can be measured with ultrasound with high accuracy.
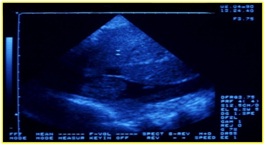
Comparative diagnosis to consider the presence of symptoms of DVT is the popliteal vein aneurysm and neoplasia of the cava vein (Figures 23 & 24).
SUPERFICIAL VEIN THROMBOSIS
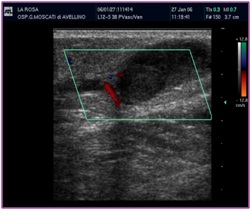
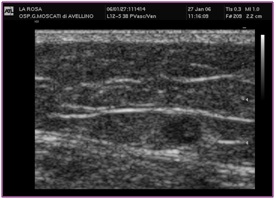
The ascending saphenous venous thrombosis represents a really dangerous situation because of the risk of extending serious evolution to SPF or EP. The criteria for deciding the therapy is the distance from the SF/J [5]. It is estimated like a thrombus at risk if its distance from SGF-J is 2 cm (Figure 27).
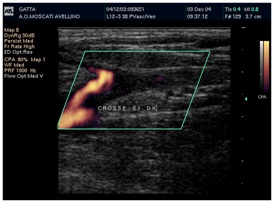
Duplex scanning showed 100% accuracy both in determining the presence of thrombosis and its extent. Another mandatory use of the duplex scanning represents the patients undergoing the new methods for the therapy of the lower limb vein, RFA, EVLT and Sclerotherapy, pre and after treatment to evaluate the outcome.
CHRONIC VENOUS INSUFFICIENCY
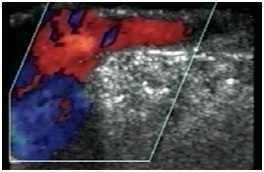
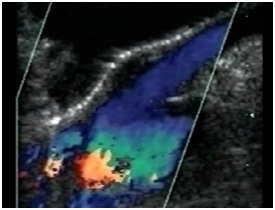
Figure 29: Sapheno-femoral junction without reflux during valsalva’s manoeuvre.
Goal to the treatment are:
• The varicosities must be permanently removed and underlying causes of venous hypertension treated
• The repair must be done in a cosmetic fashion as possible
• Complications must be minimized
The aim of varicose vein surgery is to relieve the symptoms, prevent and treat any complications while recognizing that the varicose disorder is likely to be progressive. Inadequate venous surgery is sometimes the main reason for post-surgical recurrences despite a good surgical technique. There are valid medical alternatives and sclerotherapy for collateral veins, which therefore do not necessarily call for a surgical approach. Actually, there are not defined the hemodynamic specific patters for each conservative surgical treatment. The decision should consider mini invasively and optimal long term results.
The new methods for management of the saphenous vein reflux Endovenous Ablation (EVA) with laser energy, radiofrequency VNUS closure or foam ultrasound guided sclerotherapy, need the preoperative, intra-operative and follow up valuation with duplex scanning flow.
The follow up after the procedure that was done, means to know well the basic diagnosis and the type of procedure done and a clinical and instrumental control after 1 week, then after 1 month, sequent after 6 month and after 1 year must be done.
Other new applications of non invasive diagnostic methods that change completely our therapeutic approach, are the evaluation of indication and follow up of venous stenting in the acute deep venous obstruction in the ilio-caval localization and in the chronic iliac obstruction such as the May-Turner syndrome and the non-invasive evaluation of morphologic and hemodynamic alteration of cerebral venous system CCSVI (Chronic Cerebrospinal Venous Insufficiency).
In conclusion, the most important factor in determining a good treatment outcome is making an accurate diagnosis. Recognizing common clinical patterns of vascular diseases is important, but with Color Flow Duplex Scanning now is readily available to many providers, direct visualization and mapping of vascular pathways is possible. This will ensure not only complete treatment of all of the abnormal vascular segments But preservation of normal ones.
REFERENCES
- Allegra C, Antignani PL, Kalodiki (2014) News in Angiology (1stedn). Edizioni Minerva Medica Torino, Italy.
- Allegra C, Antignani PL, Kalodiki E (2013) News in Phlebology (1stedn). Edizioni Minerva Medica, Torino, Italy.
- Allegra C, Antignani PL, Kalodiki E (2016) Tips and Tricks in Angiology (1stedn). Edizioni Minerva Medica, Torino, Italy.
- Antignani PL, Gossetti B, Righi D, Amato A, Annoni F, et al. (2004) Vascular diagnostics laboratory. Workloads and Indications. Minerva Cardioangiol 52: 447-464.
- Antignani PL, Gossetti B, Righi D, Amato A, Annoni F, et al. (2005) Accreditation process for the performance of ultrasound vascular diagnostics procedures. Minerva Cardioangiol 53: 485-506.
- Antignani PL, Benedetti-Valentini F, Aluigi L, Baroncelli TA, Camporese G, et al. (2012) Diagnosis of vascular diseases. Ultrasound investigations--guidelines; edited by Italian Society for Vascular Investigation. Int Angiol 31: 1-77.
Citation: Antignani PL, Schachter I (2016) Non-Invasive Diagnostic Methods: How Have They Modified the Therapeutical Indication? J Non Invasive Vasc Invest 1: 004.
Copyright: © 2016 Pier Luigi Antignani, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.