
The Effect of the Antioxidant Drug “U-74389g” on Uterus Congestion During Ischemia Reperfusion Injury in Rats
*Corresponding Author(s):
Constantinos TsomposDepartment Of Obstetrics And Gynecology, Messolonghi County Hospital, Nafpaktou Street, Mesologi 30200, Etoloakarnania, Greece
Tel:+30 2631360237; +30 6946674264,
Fax:+30 2106811215
Email:Constantinostsompos@yahoo.com
Abstract
Materials and methods: 40 rats of mean weight 231.875 g were used in the study. UC lesions were evaluated at 60 min of reperfusion (groups A and C) and at 120 min of reperfusion (groups B and D), A and B without but C and D with U-74389G administration.
Results: U-74389G administration significantly decreased the predicted UC scores by 0.31 [without lesions] [-0.47 - -0.16] (p=0.0004). Reperfusion time kept non-significantly increased the predicted UC scores by 0.10 [without lesions] [-0.21- 0.12] (p=0.2845). However, U-74389G administration and reperfusion time together produced a significant combined effect in decreasing the predicted UC scores by 0.17 [without lesions] [-0.27 - -0.08] (p=0.0005).
Conclusion: U-74389G administration whether it interacted or not with reperfusion time, significantly short-term kept the UC lesions scores unaltered.
Keywords
INTRODUCTION
| Variable | 1h rep | p-value | 1.5h rep | p-value | 2h rep | p-value | interaction of U-74389G and rep | p-value |
| RBC |
+1.39%+ 0.71% |
0.7161 |
+0.64%+ 0.32% |
0.8106 |
-0.10%+ 0.05% |
0.9762 |
+1.05%+ 0.53% |
0.4911 |
| Hemoglobin |
+5.2%+ 2.8% |
0.0925 |
+3.9%+ 2.1% |
0.0604 |
+2.7%+ 3.2% |
0.3544 |
+2.5%+ 1.3% |
0.0423 |
| Mean corpuscular hemoglobin |
+1.77%+ 0.96% |
0.0663 |
+2.40%+ 0.57% |
0.0001 |
+3.03%+ 0.71% |
0.0003 |
1.33%+ 0.36% |
0.0005 |
| Platelet-crit |
+3.80%+ 9.87% |
0.6373 |
+9.23%+ 6.29% |
0.1064 |
+14.66%+ 9.03% |
0.0833 |
+6.72%+ 3.73% |
0.0712 |
| PDW |
+1.1%+ 0.88% |
0.2368 |
+1.79%+ 0.76% |
0.0314 |
+2.49%+ 1.33% |
0.0807 | +0.96%+0.46% | 0.0396 |
| Glucose |
-6.41%+ 3.50% |
0.0663 | -8.57%+2.06% | 0.0001 |
-10.74%+ 2.52% |
0.0003 |
-4.76%+ 1.28% |
0.0005 |
| Total protein |
-5.48%+ 2.99% |
0.0663 |
-7.34%+ 1.76% |
0.0000 |
-9.20%+ 2.16% |
0.0000 |
-4.08%+ 1.10% |
0.0000 |
| Alkaline phosphatase |
+22.66%+ 12.37% |
0.0663 |
+31.91%+ 7.69% |
0.0001 |
+41.16%+ 9.65% |
0.0003 |
+17.75%+ 4.79% |
0.0005 |
| Creatine phosphokinas |
+54.32%+ 13.75% |
0.0012 | +35.34%+17.20% | 0.0260 |
+16.37%+ 30.24% |
0.495 |
+18.52%+ 9.44% |
0.0770 |
| Sodium |
+1.22%+ 0.66% |
0.0707 |
+0.17%+ 0.61% |
0.7714 |
-0.87%+ 1.03% |
0.3995 |
-0.32%+ 0.36% |
0.3693 |
| Chloride |
-0.58%+ 0.77% |
0.4533 |
-0.97%+ 0.53% |
0.0879 |
-1.36%+ 0.76% |
0.1113 |
-0.75%+ 0.38% |
0.0159 |
| Calcium |
0%+ 1.75% |
1 |
-0.14%+ 1.10% |
0.8782 |
-0.28%+ 1.54% |
0.8492 |
+0.14%+ 0.64% |
0.8245 |
| Phosphorus |
-2.23%+ 5.51% |
0.7966 |
-1.61%+ 3.32% |
0.5789 |
-1%+ 4.48% |
0.8129 |
-1.09%+ 2% |
0.5771 |
| Magnesium |
+1.33%+ 3.59% |
0.7033 |
-0.28%+ 2.75% |
0.9171 |
-1.90%+ 5.28% |
0.7161 |
+0.36%+ 4.58% |
0.8228 |
| Mean |
+5.57%+ 15.58% |
0.3552 |
+4.74%+ 12.98% |
0.3049 |
+3.92%+ 12.98% |
0.3485 |
+2.73%+ 7.06% |
0.2380 |
The aim of this experimental study was to examine the effect of U-74389G on rat model and particularly in as uterus IR protocol. The beneficial effect or non-effectiveness of that molecule was studied by evaluating Uterus Congestion (UC) lesions.
MATERIALS AND METHODS
Animal preparation
The detailed preceded prenarcotic and general anesthesiologic techniques are described in related reference [3]. Oxygen supply, electrocardiogram and acidometry were continuously provided during whole experiment performance.
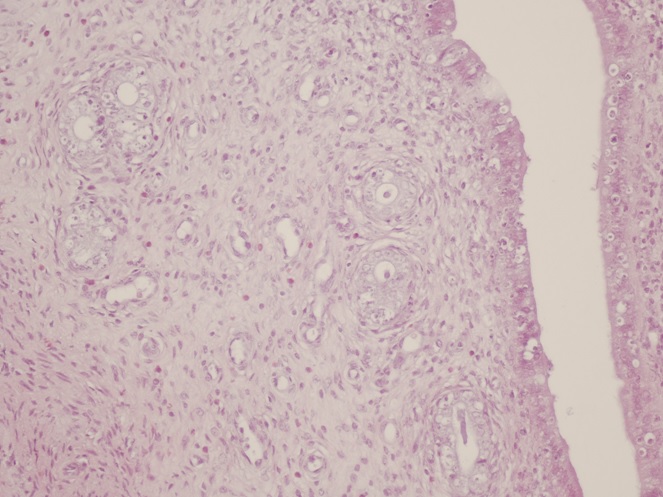
The protocol of IR was followed. Ischemia was caused by laparotomic forceps clamping inferior aorta over renal arteries for 45 min. Reperfusion was induced by removing the clamp and reestablishment of inferior aorta patency. The molecules were administered at the time of reperfusion, through catheterized inferior vena cava. The UC lesions evaluations were performed at 60 min of reperfusion (for groups A and C) and at 120 min of reperfusion (for groups B and D). Forty (40) female Wistar albino rats were used of mean weight 231.875 g [Std. Dev: 36.59 g], with min weight ≥ 165 g and max weight < 320 g. Rats’ weight could be potentially a confusing factor, e.g., the more obese rats to have higher UC scores. This suspicion was investigated. Also, detailed pathological study [4] and grading of UC findings was performed by scores, this is: 0 lesions were not found, 1 mild lesion was found, 2 moderate lesions were found and 3 serious lesions were found. The previous grading is transformed as follows: (0-0.499) without lesions, (0.5-1.499) mild lesions, (1.5-2.499) moderate lesions and (2.5-3) serious lesions damage, because the study concerns score ranges rather than point scores.
Model of ischemia reperfusion injury
Group A: Reperfusion which lasted 60 min concerned 10 control rats of mean weight 243 g [Std. Dev: 45.77 g] and mean moderate mild UC score 1.4 [Std. Dev: 0.51] (Table 2).
| Groups | Variable | Mean | Std. Dev |
| Α | Weight | 243 g | 45.77 g |
| UC | mild 1.4 | 0.51 | |
| B | Weight | 262 g | 31.10 g |
| UC | mild 1.1 | 0.31 | |
| C | Weight | 212.5 g | 17.83 g |
| UC | without lesions 0.3 | 0.48 | |
| D | Weight | 210 g | 18.10 g |
| UC | without lesions 0.4 | 0.51 |
Group B: Reperfusion which lasted 120 min concerned 10 control rats of mean weight 262 g [Std. Dev: 31.10 g] and mean mild UC score 1.1 [Std. Dev: 0.31] (Table 2).
Lazaroid (L) group: 20 rats of mean weight 211.25 g [Std. Dev: 17.53 g] experienced ischemia for 45 min followed by reperfusion in the beginning of which 10 mg U-74389G /kg body weight were IV administered.
Group C: Reperfusion which lasted 60 min concerned 10 L rats of mean weight 212.5 g [Std. Dev: 17.83 g] and mean without lesions UC score 0.3 [Std. Dev: 0.48] (Table 2).
Group D: Reperfusion which lasted 120 min concerned 10 L rats of mean weight 210 g [Std. Dev: 18.10 g] and mean without lesions UC score 0.4 [Std. Dev: 0.5163978] (Table 2).
Statistical analysis
| DG | Variable | Difference | p-value |
| Α-Β | Weight | -19 g | 0.2423 |
| UC | without lesions 0.3 | 0.0833 | |
| Α-C | Weight | 30.5 g | 0.0674 |
| UC | mild 1.1 | 0.0067 | |
| Α-D | Weight | 33 g | 0.0574 |
| UC | mild 1 | 0.0102 | |
| Β-C | Weight | 49.5 g | 0.0019 |
| UC | mild 0.8 | 0.0092 | |
| Β-D | Weight | 52 g | 0.0004 |
| UC | mild 0.7 | 0.0154 | |
| C-D | Weight | 2.5 g | 0.7043 |
| UC | without lesions -0.1 | 0.5637 |
| Groups | Mean | Std. Dev |
| Α | mild 0.88 | 0.51 |
| B | mild 1.03 | 0.31 |
| C | mild 0.64 | 0.48 |
| D | mild 0.63 | 0.51 |
| DG | Difference | p-value |
| Α-Β | without lesions -0.14 | 0.1843 |
| Α-C | without lesions 0.23 | 0.0745 |
| Α-D | without lesions 0.25 | 0.0743 |
| Β-C | without lesions 0.38 | 0.0093 |
| Β-D | without lesions 0.40 | 0.0069 |
| C-D | without lesions 0.01 | 0.9182 |
RESULTS
| Decrease | 95% c. in | Reperfusion time | Wilcoxon |
p-values glm |
| mild 1.1 | -1.56 - -0.63 | 1h | 0.0067 | 0.0001 |
| mild 0.9 | -1.19 - -0.60 | 1.5h | 0.0003 | 0.0000 |
| mild 0.7 | -1.10 - -0.29 | 2h | 0.0154 | 0.0018 |
| without lesions 0.1 | -0.51 - 0.31 | reperfusion time | 0.632 | |
| without lesions 0 | -0.33 - 0.33 | reperfusion time | 1.0000 | |
| without lesions 0.47 | -0.67 - -0.27 | Interaction | 0.0000 |
| Decrease | 95% c. in | Reperfusion time | Wilcoxon |
p-values glm |
| without lesions 0.23 | -0.48 - 0.01 | 1h | 0.0745 | 0.0653 |
| without lesions 0.31 | -0.47 - -0.16 | 1.5h | 0.0008 | 0.0001 |
| without lesions 0.40 | -0.58 - -0.21 | 2h | 0.0069 | 0.0002 |
| without lesions -0.06 | -0.11 - 0.24 | reperfusion time | 0.4831 | |
| without lesions -0.14 | -0.30 - 0.00 | reperfusion time | 0.0859 | |
| without lesions 0.17 | -0.27 - -0.08 | interaction | 0.0005 |
| Decrease | 95% c. in | Reperfusion time | p-values |
| without lesions 0.23 | -0.48 - 0.01 | 1h | 0.0699 |
| without lesions 0.31 | -0.47 - -0.16 | 1.5h | 0.0004 |
| without lesions 0.40 | -0.58 - -0.21 | 2h | 0.0035 |
| without lesions -0.10 | -0.21- 0.12 | -0.21- 0.12 | 0.2845 |
| without lesions 0.17 | without lesions 0.17 | interaction | 0.0005 |
DISCUSSION
The following situations show the association between ischemia and congestion in uterus. Salas SP postulated [6] that cerebral congestion, secondary to compression of the abdominal organs by the large uterus, diverts blood to the brain, causing eclamptic convulsions. Surcel VJ et al., showed that uterus fibroma has always been accompanied by pelvic congestion inducing [7] experimentally estrogen tumors in animals. Douglas BH observed liver and renal glomerular congestion both in pregnant and non-pregnant rats producing [8] hypertension, however, only in pregnant ones.
Lazaroids, a novel series of glucocorticoid compounds 21-aminosteroids have the properties of free radical scavenging. U-74389G is one of the 132 similar lazaroid compounds. It has a molecular weight of 726.90406 g/mol; it has a selective action on vascular endothelium with vitamin E-like properties. The most famous activity is that of neuroprotective and membrane-stabilizing properties. Although it accumulates in the cell membrane, thus protecting vascular endothelium from peroxidative damage hardly penetrates the blood-brain barrier. More specifically, Hori H et al., showed [9] its excellent effect on central nervous system trauma and ischemia. The degree of elevation of action potential thresholds and the rate of missing outer hair cells were significantly reduced demonstrating that U-74389G has a protective effect on cisplatin-induced ototoxicity without glucocorticoid action. Schmid-Elsaesser R et al., showed [10] significantly less neurologic deficits postoperatively and significantly reduced cortical infarct volumes by the neuroprotective microvascularly acting 21-aminosteroid U-74389G. Passaquin AC et al., elicited [11] a beneficial effect of glucocorticoids in Duchenne muscular dystrophy, attributing it to a reduction of the pathological increase in Ca++ influx via an effect on the sarcolemma of C2C12 skeletal muscle cells. van Klaveren RJ et al., supposed that direct inactivation of the membrane-bound γGT by hyperoxia is the most likely mechanism for the increased [12] γGT, SOD, and GSH levels in oxygen-exposed cells by U-74389G. Schmid-Elsaesser R et al., concluded [13] that antioxidative compounds which cross the blood-brain barrier are more effective in focal cerebral ischemia than agents which predominantly act on the endothelium of cerebral microvessels. Lehmann C et al., decreased [14] TNFα release during endotoxemia permitting treatment of septic states. As immunosuppressant it may act through activation of T-cells or by inhibiting the activation of helper cells. While immunosuppression primarily prevents rejection of transplanted organs, new applications involving mediation of the effects of interleukins and other cytokines are emerging. Lehmann C et al., attenuated [15] leukocyte adherence and their rolling behavior in intestinal venules which is found increased during endotoxemia. Horáková L et al., [16] calculated the preventive effect concerning lipid peroxidation at 160 IC50 μmol/l by U 74389G in oxidative stress. Heim C et al., totally prevented [17] the learning impairments, suggesting that lipid peroxidation may be responsible for the late learning deficiencies. Vlkolinský R revealed [18] protective activity on Synaptic Transmission (ST) recovery and on t1/2 during hypoxia; a protective potency of U-74389G on Population Spikes (PoS) recovery and a possibility to delay the early ST decay during hypoxia, which might indicate improved energetic state of neurons in the treated tissue. Durmaz R et al., [19] showed antiproliferative properties on cancer cells calculating an IC50 value at 91 m mM. Kondziolka D et al., prevented [20] regional edema favoring radiosurgery, surrounding brain protection without reducing the desired therapeutic effect.
Uterine congestion can be met at many clinical situations. Smith CC et al., assessed [21] intravascular growth in 20% of 41 leiomyoma patients median aged 46 years old but this finding is apparently devoid of clinical significance due to limited data. Typical presence included menorrhagia; average tumor size 15.4 cm; most exophytic, usually contiguous with the intramural dissecting leiomyomatous components but typically isointense to myometrium, multinodular, occasionally cystic masses with a congested, spleen or placenta-like color, protruding over the uterine serosa and variably, the broad ligaments and adjacent organs. Hu LQ et al., displayed [22] loosened cervical tissue with significantly lessened, swollen, convoluted and ruptured collagen fiber, showing sparse disorderly lined-up reticular status in rats after pitocin medication. Degradation of collagen fiber, vascular dilatation and congestion with massive amount of inflammatory cells infiltration, increased matrix components, many leucocytes, fibroblast in the stroma and higher cervical score were also displayed in treated group than placebo (P<0.05). Gaspard UJ et al., noted [23] in decreasing order of frequency mastalgia, vaginal discharge, nausea, abdominal and leg cramps, headaches, weight increase, spotting, breakthrough and withdrawal bleeding. 20% of women dropped out of the study essentially for breast tenderness, weight increase, spotting and nausea during continued 10 cycles use of a new triphasic oral contraceptive (WL-49(50) ‘Trinordiol’) containing the lowest quantity of steroids in 75 healthy teenager young women. Singh ND et al., recorded [24] vascular changes and hemosiderosis in uterus associated with lymphocytic depletion and apoptosis in the lymphoid organs in induced toxic Wistar rats.
CONCLUSION
ACKNOWLEDGMENT
REFERENCES
- https://www.caymanchem.com/app/template/Product.vm/catalog/75860
- Shi F, Cavitt J, Audus KL (1995) 21-aminosteroid and 2-(aminomethyl)chromans inhibition of arachidonic acid-induced lipid peroxidation and permeability enhancement in bovine brain microvessel endothelial cell monolayers. Free Radic Biol Med 19: 349-357.
- Tsompos C, Panoulis C, Toutouzas K, Zografos G, Papalois A (2015) The Acute Effect of the Antioxidant Drug “U-74389g” on Platelet Distribution Width During Hypoxia Reoxygenation Injury in Rats. J Neurol Stroke 3: 00111.
- Osmana?ao?lu MA, Kesim M, Yulu? E, Mente?e A, Karahan SC (2012) Ovarian-protective effects of clotrimazole on ovarian ischemia/reperfusion injury in a rat ovarian-torsion model. Gynecol Obstet Invest 74: 125-130.
- Boon NA, Davidson S, (2006) In: Boon NA (ed.). Davidson’s Principles & Practice of Medicine. Elsevier/Churchill Livingstone. Pg no: 544.
- Salas SP (1999) What causes pre-eclampsia? Baillieres Best Pract Res Clin Obstet Gynaecol 13: 41-57.
- Surcel VJ, Rotaru O, Toader S (1982) [Experimental induction of uterus tumours by modification of localised vascularization]. Zentralbl Gynakol 104: 669-677.
- Douglas BH (1976) The rat as a model for preeclampsia. Perspect Nephrol Hypertens 5: 411-419.
- Hori H, Kanno H (1999) [An experimental study of the protective effect of lazaroid (U-74389G) on cisplatin-induced toxicity]. Nihon Jibiinkoka Gakkai Kaiho 102: 8-18.
- Schmid-Elsaesser R, Hungerhuber E, Zausinger S, Baethmann A, Reulen HJ (1999) Neuroprotective efficacy of combination therapy with two different antioxidants in rats subjected to transient focal ischemia. Brain Res 816: 471-479.
- Passaquin AC, Lhote P, Rüegg UT (1998) Calcium influx inhibition by steroids and analogs in C2C12 skeletal muscle cells. Br J Pharmacol 124: 1751-1759.
- van Klaveren RJ, Pype JL, Demedts M, Nemery B (1997) Decrease in gamma-glutamyltransferase activity in rat type II cells exposed in vitro to hyperoxia: effects of the 21-aminosteroid U-74389G. Exp Lung Res 23: 347-359.
- Schmid-Elsaesser R, Zausinger S, Hungerhaber E, Baethmann A, Reulen HJ (1997) Neuroprotective properties of a novel antioxidant (U-101033E) with improved blood-brain barrier permeability in focal cerebral ischemia. Acta Neurochir Suppl 70: 176-178.
- Lehmann C, Egerer K, Georgiew A, Weber M, Grune T, et al. (1999) Inhibition of tumor necrosis factor-alpha release in rat experimental endotoxemia by treatment with the 21-aminosteroid U-74389G. Crit Care Med 27: 1164-1167.
- Lehmann C, Georgiew A, Weber M, Birnbaum J, Kox WJ (2001) Reduction in intestinal leukocyte adherence in rat experimental endotoxemia by treatment with the 21-aminosteroid U-74389G. Intensive Care Med 27: 258-263.
- Horáková L, Ondrejicková O, Bachratá K, Vajdová M (2000) Preventive effect of several antioxidants after oxidative stress on rat brain homogenates. Gen Physiol Biophys 19: 195-205.
- Heim C, Kolasiewicz W, Sontag KH (2000) The effects of the 21-aminosteroid U-74389G on spatial orientation in rats after a cerebral oligemic episode and iron-induced oxidative stress. J Neural Transm (Vienna) 107: 95-104.
- Vlkolinský R, Stolc S (1999) Effects of stobadine, melatonin, and other antioxidants on hypoxia/reoxygenation-induced synaptic transmission failure in rat hippocampal slices. Brain Res 850: 118-126.
- Durmaz R, Deliorman S, Isiksoy S, Uyar R, Erol K, et al. (1999) Antiproliferative properties of the lazaroids U-83836E and U-74389G on glioma cells in vitro. Pathol Oncol Res 5: 223-228.
- Kondziolka D, Somaza S, Martinez AJ, Jacobsohn J, Maitz A, et al. (1997) Radioprotective effects of the 21-aminosteroid U-74389G for stereotactic radiosurgery. Neurosurgery 41: 203-208.
- Smith CC, Gold MA, Wile G, Fadare O (2012) Cotyledonoid dissecting leiomyoma of the uterus: a review of clinical, pathological, and radiological features. Int J Surg Pathol 20: 330-341.
- Hu LQ, Cai LL (2008) [Clinical and experimental study on effect of cuichan zhusheng decoction on the structure and tension of pregnant cervix uteri]. Zhongguo Zhong Xi Yi Jie He Za Zhi 28: 513-517.
- Gaspard UJ, Deville JL, Dubois M (1983) Clinical experience with a triphasic oral contraceptive (‘Trinordiol’) in young women. Curr Med Res Opin 8: 395-404.
- Singh ND, Sharma AK, Dwivedi P, Patil RD, Kumar M (2007) Citrinin and endosulfan induced maternal toxicity in pregnant Wistar rats: pathomorphological study. J Appl Toxicol 27: 589-601.
Citation: Tsompos C, Panoulis C, Toutouzas K, Triantafyllou A, Zografos G, et al., (2016) The Effect of the Antioxidant Drug “U-74389g” on Uterus Congestion During Ischemia Reperfusion Injury in Rats. J Reprod Med Gynecol Obstet 1: 001.
Copyright: © 2016 Constantinos Tsompos, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

