
Preparation and Thermoelectric Properties of Nickel Oxide added Strontium Ferrite Nano-Composite
*Corresponding Author(s):
Shin NishiyamaSchool Of Engineering, Chiba University, Japan
Email:shin@faculty.chiba-u.jp
Abstract
Keywords
Oxide Nano-Composite, Seebeck Coefficient, Thermal Conductivity, Thermoelectric Performance
INTRODUCTION
NiO was not so soluble in transition metals [6]. In this study, SrFeO3-δ bulk polycrystalline samples and NiO nano-particles composite samples were prepared by an ordinally method and effects of NiO addition on the thermoelectric properties were investigated. The electrical conductivities and Seebeck coefficients of these ceramics samples were measured and the thermal conductivities of the part of the samples were also measured.
MATERIALS AND METHODS
For conductivity measurements and Seebeck coefficient measurements, a rectangular sample of about 2 times 4 mm was polished by a sand paper. After a constant DC current was applied, the current versus the measured voltage, the cross-sectional area of the samples, and the distances between the two platinum wires were measured, and then the conductivities were calculated. The Seebeck coefficient was determined when the temperature of both ends of the samples became stable using an air pump. The temperature difference versus Seebeck voltage was plotted, and the slope was taken as the relative Seebeck coefficient. The absolute Seebeck coefficient of the sample was obtained by subtracting the absolute Seebeck coefficient of platinum from the relative Seebeck coefficient of the sample. For the thermal conductivity measurement, samples shaped and sintered so as to have a thickness of about 2 mm using a 15 mm × 15 mm square mold were used. The surface was polished in the same manner as the samples for electrical property measurement. From the measured thermal diffusivity, specific heat, and material density, the thermal conductivity was calculated.
RESULTS AND DISCUSSION
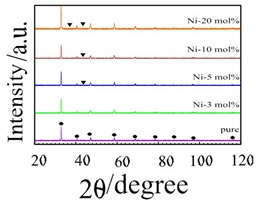 Figure 1: XRD patterns of s intered bodies of SrFeNixO3 (0 ≤ x ≤ 20). ? is SrFeO3 phase, ? is NiO phase.
Figure 1: XRD patterns of s intered bodies of SrFeNixO3 (0 ≤ x ≤ 20). ? is SrFeO3 phase, ? is NiO phase.
Figure 3 shows the temperature dependence of the conductivities of the samples. Those of all measured samples increased with temperature up to about 400°C, then reversed and decreased. This is thought to be due to the formation of oxygen deficiency of SrFeO3-δ at about 400°C. In the sample with the addition amount of 10 mol% or less, the conductivity showed almost the same value as that of the sample without addition, but in the sample added with 20 mol%, the conductivity was lower than that of the sample not added in the whole measured temperature range.
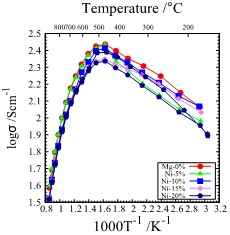
Figure 4 shows the temperature dependence of the Seebeck coefficient of the same sample. All specimens increased with temperature, and the tendency was remarkable over 400°C. Also, by adding NiO, the Seebeck coefficient over 400°C was greatly improved, and in the measured temperature range the higher the temperature, the more remarkable the tendency appeared. Furthermore, it was found that the addition of NiO lowers the thermal conductivity at room temperature.
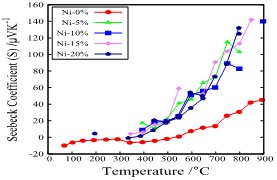
Table 1 shows the results of the thermal conductivity of a sample containing 5 mol% of NiO with different particle sizes. Samples obtained by pulverizing NiO with a planetary ball mill at 900 rpm for 2 h showed lower values than samples not pulverized. It is thought that the small NiO particles at grain boundaries exist as the scattering center of phonon transportation, and the effect was higher as the particle was finer. In addition, it can be considered that addition of NiO suppressed grain growth of the base material.
|
Specific heat(J/gK) |
Thermal diffusivity(cm2/sec) |
Density(g/cm3) |
Thermal conductivity(W/mK) |
|
|
Ni-5mol%(un milled) |
7.529×10−2 |
1.060×10−1 |
4.668 |
3.725 |
|
Ni-5mol%(900rpm, 2h) |
4.765×10−2 |
1.127×10−1 |
4.676 |
2.511 |
CONCLUSION
REFERENCES
- Dresselhouse MS, Chen G, Tang MY, Yang R, Lee H, et al. (2007) New directions for low?dimensional thermoelectric materials. Advanced Materials 19: 1043-1053.
- Hsu KF, Loo S, Guo F, Chen W, Dyck JS, et al. (2004) Cubic AgPb(m)SbTe(2+m): Bulk thermoelectric materials with high figure of merit. Science 303: 818-821.
- Boukai AI, Bunimovich Y, Tahir Kheli J, Yu JK, Goddard III WA, et al. (2008) Silicon nanowires as efficient thermoelectric materials. Nature 451: 168-171.
- Poudel B, Hao Q, Ma Y, Lan Y, Minnich A, et al. (2008) High-thermoelectric performance of nanostructured bismuth antimony telluride bulk alloys. Science 320: 634-638.
- Wang H, Hwang J, Snedaker ML, Kim IH, Kang C, et al. (2015) High thermoelectric performance of a heterogeneous PbTe nano-composite. Chem Mater 27: 944-949.
- Mandal SK, Das AK, Nath TK, Karmakar D (2006) Applied Physics Letters 89: 144105.
Citation: Nishiyama S, Yamada A (2019) Preparation and Thermoelectric Properties of Nickel Oxide added Strontium Ferrite Nano-Composite. J Mod Chem Sci 3: 004.
Copyright: © 2019 Shin Nishiyama, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

