
A Functional Beverage Containing Lactobacillus helveticus and Mosla chinensis Maxim Supports Respiratory Health In Vitro
*Corresponding Author(s):
Chi-Fu ChiangResearch & Design Center, TCI GENE, Taipei, Taiwan
Tel:+886 287977811,
Email:Jimmy.Chiang@tci-bio.com
Abstract
Respiratory health has gained increasing attention in the post-pandemic era, particularly with respect to epithelial repair, inflammatory regulation, and fibrosis-associated risk following infection or environmental stress. Functional beverages combining microbiota-modulating components with botanical ingredients may provide supportive strategies for maintaining pulmonary homeostasis. This study investigated the lung-protective potential of a commercially available functional beverage containing Lactobacillus helveticus–associated functional components, Mosla chinensis Maxim., and additional medicinal and edible plant ingredients using in vitro cell-based models. Human Bronchial Epithelial Cells (BEAS-2B) were used to evaluate epithelial repair capacity via a wound healing assay, while human lung fibroblasts (WI-38) were employed to assess inflammatory and pro-fibrotic cytokine responses under Lipopolysaccharide (LPS)-induced inflammatory conditions. The results showed that the functional beverage significantly enhanced epithelial wound repair, with the repair rate reaching 161.6% compared with the control group. Under LPS stimulation, treatment with the composite beverage markedly attenuated the secretion of the pro-inflammatory cytokine interleukin-8 (IL-8) and significantly reduced levels of transforming growth factor-β1 (TGF-β1), a key regulator of pulmonary fibrosis. Notably, suppression of TGF-β1 secretion was also observed under basal conditions without inflammatory stimulation. Overall, these findings demonstrated that the functional beverage supported lung-protective functions in vitro by enhancing respiratory epithelial repair while modulating inflammatory and pro-fibrotic cytokine responses, providing mechanistic support for its potential application as a functional nutritional strategy for respiratory health maintenance.
Keywords
Lactobacillus helveticus; Lung Protection; Mosla chinensis Maxim; Respiratory Epithelial Repair
Introduction
The global outbreak of Coronavirus Disease 2019 (COVID-19) has profoundly reshaped public awareness of respiratory health. Beyond the acute phase of infection, a substantial proportion of individuals continue to experience prolonged respiratory discomfort, impaired airway repair, persistent inflammation, or reduced pulmonary function [1]. These post-infection sequelae highlight that maintaining respiratory resilience, tissue repair capacity, and immune homeostasis has become a critical health concern in the post-pandemic era [2]. Consequently, there is growing interest in preventive and supportive strategies that promote long-term respiratory health rather than solely addressing acute symptoms.
Conventional Western medical approaches to respiratory disorders primarily rely on anti-inflammatory agents, bronchodilators, and immunomodulatory drugs. While these treatments are effective in controlling acute symptoms and disease exacerbations, their long-term use may be associated with limitations, including drug tolerance, systemic side effects, and insufficient support for tissue repair and functional recovery [3]. In contrast, traditional Chinese medicine and botanical-based interventions have historically been used to support respiratory function through milder and more holistic approaches [4]. Plant-derived ingredients are often characterized by multi-target biological activities and are considered suitable for long-term use, with the potential to support immune balance and tissue homeostasis [5]. As a result, botanical extracts have gained increasing attention in the field of functional foods and integrative health research [6]. In parallel, advances in microbiome science have led to the recognition of the gut–lung axis as an important regulatory pathway linking intestinal microbial homeostasis to respiratory health [7]. Accumulating evidence suggests that gut microbiota can influence pulmonary immune responses, inflammatory signaling, and epithelial barrier function through microbial metabolites and immune-mediated mechanisms [8]. In this context, microbiota-modulating strategies, including the use of prebiotic or prebiotic-like components (also referred to as synbiotic-supporting matrices or “next-generation microbiota modulators”), have emerged as promising approaches to indirectly regulate respiratory health [9]. Lactobacillus helveticus–associated functional matrices and fermentation-derived components have been reported to exhibit immunomodulatory properties, including the regulation of inflammatory cytokine production and enhancement of epithelial cell function [10]. These observations suggest that microbiota-oriented nutritional strategies may contribute to respiratory health maintenance via gut–lung axis–mediated immune modulation.
In addition to microbiota-modulating components, medicinal herbs traditionally used for respiratory support have attracted scientific interest. Mosla chinensis Maxim., commonly known as Elsholtzia in traditional Chinese medicine, has long been used for the management of external pathogenic factors and respiratory discomfort [11]. Contemporary pharmacological studies indicate that Mosla chinensis Maxim. possesses anti-inflammatory, antioxidant, and bioactive properties relevant to respiratory health [12]. These characteristics suggest that this botanical ingredient may play a supportive role in maintaining pulmonary homeostasis and mitigating inflammation-related respiratory stress.
Based on these considerations, the present study investigated a commercially available microbiota-modulating botanical functional beverage, which contains Lactobacillus helveticus–associated functional components, Mosla chinensis Maxim., and thirteen additional medicinal and edible plant ingredients. Using an in vitro cell-based model, this study aimed to evaluate the potential lung-protective effects of this composite formulation. By examining cellular responses related to pulmonary protection, this research seeks to provide mechanistic evidence supporting the application of prebiotic-oriented, herbal functional beverages.
Materials And Methods
- Cell Culture
Human bronchial epithelial cells (BEAS-2B) and human lung fibroblasts (WI-38) were used in this study to evaluate the cellular effects of the tested formulation on airway epithelial function and fibroblast-related responses. The BEAS-2B cell line and WI-38 human embryonic lung fibroblasts were obtained from the American Type Culture Collection (ATCC). BEAS-2B cells were cultured in Dulbecco’s Modified Eagle Medium (DMEM) supplemented with 10% fetal bovine serum (FBS) and 1% penicillin–streptomycin. WI-38 cells were maintained in Minimum Essential Medium (MEM) supplemented with 10% fetal bovine serum and 1% penicillin–streptomycin. All cells were incubated at 37 °C in a humidified atmosphere containing 5% CO2.
- Test sample
The test sample evaluated in this study was a commercially available functional beverage, Probiotics Elsholtzia Compound Drink (SYRINX®, China). The formulation contains Lactobacillus helveticus TCI357 fermented, Mosla chinensis Maxim., and thirteen medicinal and edible botanical ingredients, including Mentha haplocalyx Briq., Morus alba L. leaf, Houttuynia cordata Thunb. extract, Platycodon grandiflorus (Jacq.) A. DC., Chrysanthemum morifolium Ramat., Poria cocos (Schw.) Wolf, Lilium brownii var. viridulum, Siraitia grosvenorii (Swingle) C. Jeffrey, Prunus mume Siebold & Zucc., Hippophae rhamnoides L., Glycyrrhiza uralensis Fisch., Dendrobium officinale Kimura & Migo flower, and Perilla frutescens (L.) Britton seed powder, and additional food-grade excipients, including citric acid, steviol glycosides, sucralose, plum-flavored food-grade flavoring, erythritol, apple juice concentrate, and purified water.
- Wound healing assay
Cellular repair capacity was evaluated using a culture-insert–based wound healing assay. Briefly, human bronchial epithelial BEAS-2B cells were seeded into culture inserts placed in 6-well plates, creating a defined cell-free gap between two cell populations. Cells were cultured under standard conditions until firm attachment and monolayer formation were achieved. After cell attachment, the culture inserts were carefully removed to generate a uniform wound area. The wells were gently washed with phosphate-buffered saline (PBS) to remove non-adherent cells, followed by the addition of fresh culture medium containing the test samples at the indicated concentrations. Cells cultured in medium alone served as the control group. Wound closure was monitored by phase-contrast microscopy. Images were captured immediately after insert removal (0 h) and after 6 h of incubation. The extent of wound healing was quantified by measuring the remaining wound area using ImageJ software.
- Inflammatory Cytokine Assay
WI-38 cells were seeded into culture plates and cultured under standard conditions until reaching approximately 70–80% confluence. To induce an inflammatory and pro-fibrotic response, cells were stimulated with Lipopolysaccharide (LPS) at a final concentration of 4 μg/mL. Simultaneously, cells were treated with the test samples at the indicated concentrations. Following incubation for the designated period, culture supernatants were collected and centrifuged to remove cellular debris. The levels of transforming growth factor-β (TGF-β) an IL-8 in the supernatants were quantified using a commercially available enzyme-linked immunosorbent assay (ELISA) kit, according to the manufacturer’s instructions.
- Statistical Analysis
All results are expressed as mean ± Standard Deviation (SD). Group differences were analyzed using Student’s t-test (Microsoft Excel, USA). A p value < 0.05 was considered statistically significant.
Results
- Functional beverage enhanced respiratory epithelial cell repair
The cellular results demonstrated that, compared with the control group, cells treated with the functional beverage exhibited more pronounced cell migration and coverage within the scratch area, indicating an enhanced epithelial repair process. Microscopic observations revealed that cells in the functional beverage–treated group showed accelerated movement toward the wound region and more effective closure of the scratched area, whereas the control group displayed relatively limited cell migration and delayed wound closure. Further quantitative analysis confirmed these observations, showing that the respiratory epithelial cell repair rate in the functional beverage–treated group was significantly increased, reaching 161.6% of that observed in the control group. This enhancement was statistically significant, demonstrating that treatment with the functional beverage markedly promoted epithelial wound healing capacity in vitro (Figure 1).
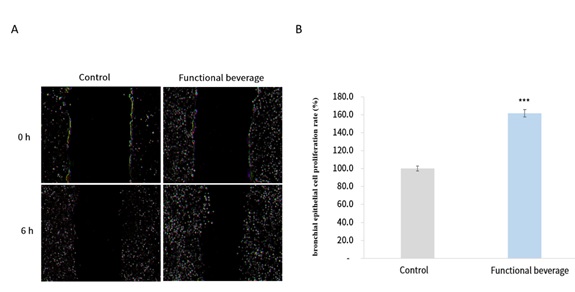 Figure 1: Wound healing assay for evaluating respiratory epithelial cell repair.
Figure 1: Wound healing assay for evaluating respiratory epithelial cell repair.
A scratch wound healing assay was conducted using human bronchial epithelial BEAS-2B cells to assess epithelial repair capacity following treatment with the functional beverage. Representative phase-contrast microscopic images show the wound area immediately after scratch induction and after incubation under control or functional beverage–treated conditions. Quantification of epithelial cell repair was performed by measuring wound closure using image analysis software. Data are expressed as mean ± standard deviation (SD). Statistical significance was determined by comparison with the control group.
- Functional beverage reduced inflammatory cytokines
Under inflammatory conditions induced by simulated bacterial infection (LPS stimulation), the secretion of transforming growth factor-β1 (TGF-β1), a key regulator of pulmonary fibrosis, was significantly increased, reflecting activation of pro-fibrotic signaling pathways. Compared with the LPS-only group, co-treatment with the functional beverage significantly reduced TGF-β1 secretion by 55.2%, indicating that the functional beverage effectively attenuated the excessive induction of pro-fibrotic mediators under inflammatory conditions (Figure 2A). Importantly, the regulatory effect of the functional beverage on TGF-β1 secretion was not limited to inflammatory stimulation. Under normal culture conditions without LPS exposure, treatment with the functional beverage also resulted in a significant reduction in TGF-β1 secretion, reaching 53.4% of the control level. These findings suggest that the functional beverage is capable of modulating pulmonary fibrosis–related factors not only during inflammatory stress but also under basal physiological conditions (Figure 2B). In parallel with the observed changes in pro-fibrotic signaling, inflammatory cytokine responses were also evaluated. Under LPS-induced inflammatory conditions, the secretion of the pro-inflammatory cytokine interleukin-8 (IL-8) was significantly elevated compared with the control group. Notably, co-treatment with the functional beverage markedly attenuated IL-8 secretion, reducing 77.6% relative to the LPS-only group. This reduction indicates that the functional beverage effectively suppressed infection-induced inflammatory responses at the cytokine level (Figure 2C).
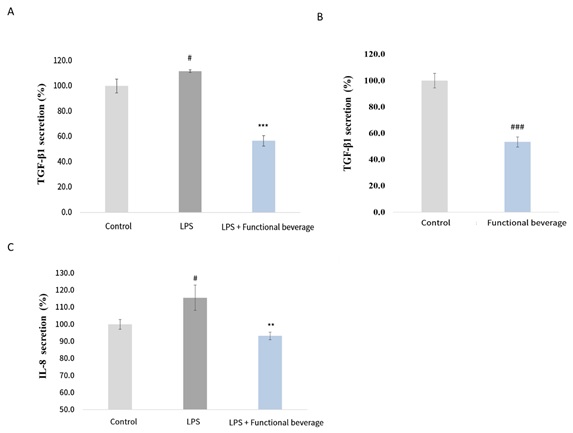 Figure 2: Evaluation of cytokine secretion under inflammatory and basal conditions.
Figure 2: Evaluation of cytokine secretion under inflammatory and basal conditions.
(A) Human lung fibroblasts (WI-38) were stimulated with lipopolysaccharide (LPS) to induce an inflammatory condition and treated with the functional beverage. The secretion of transforming growth factor-β1 (TGF-β1) in culture supernatants was measured by enzyme-linked immunosorbent assay (ELISA).
(B) WI-38 cells were maintained under basal culture conditions without LPS stimulation and treated with the functional beverage. TGF-β1 levels in the culture supernatants were quantified by ELISA. (C) Under LPS-induced inflammatory conditions, WI-38 cells were treated with the functional beverage, and the secretion of interleukin-8 (IL-8) in the culture supernatants was measured by ELISA. Compared with the control group: # p < 0.05, ### p < 0.001; compared with the LPS group: ** p < 0.01, *** p < 0.001.
Discussion
The present study investigated the lung-protective potential of a commercially available functional beverage containing Lactobacillus helveticus–associated functional components, Mosla chinensis Maxim., and additional medicinal and edible botanical ingredients using in vitro cell-based models. By evaluating respiratory epithelial repair capacity, inflammatory cytokine responses, and fibrosis-related signaling markers, this study aimed to provide mechanistic insights into how a microbiota-modulating botanical composite formulation may support respiratory health. The results demonstrated that the composite beverage enhanced epithelial wound repair, attenuated infection-induced inflammatory responses, and reduced pro-fibrotic cytokine secretion under both inflammatory and basal conditions. Collectively, these findings suggest that the tested formulation exerts multi-level regulatory effects relevant to pulmonary protection.
One of the most notable observations in this study was the concurrent modulation of inflammatory and pro-fibrotic mediators, particularly interleukin-8 (IL-8) and transforming growth factor-β1 (TGF-β1). IL-8 is a key chemokine involved in neutrophil recruitment and amplification of inflammatory cascades within the respiratory tract. Excessive or sustained IL-8 production has been implicated in acute lung injury, chronic airway inflammation, and post-infectious respiratory dysfunction [13]. In the present study, Lipopolysaccharide (LPS) stimulation markedly increased IL-8 secretion, reflecting activation of infection-associated inflammatory signaling. Importantly, co-treatment with the functional beverage significantly attenuated this response, indicating that the formulation may intervene at an early stage of inflammatory activation [14]. In parallel with the regulation of IL-8, the functional beverage significantly reduced TGF-β1 secretion, a central regulator of pulmonary fibrosis and tissue remodeling [15]. TGF-β1 plays a critical role in fibroblast activation, extracellular matrix deposition, and structural remodeling following lung injury [16]. Persistent elevation of TGF-β1 is commonly associated with progressive fibrotic changes and impaired pulmonary function. In this study, suppression of TGF-β1 secretion was observed not only under LPS-induced inflammatory conditions but also under basal culture conditions, suggesting that the regulatory capacity of the functional beverage is not limited to acute inflammatory stress [17]. The simultaneous attenuation of IL-8 and TGF-β1 supports the concept that modulation of early inflammatory responses may contribute to limiting downstream fibrosis-associated signaling and maintaining pulmonary homeostasis [18].
The observed biological effects may be partially attributed to the functional properties of Lactobacillus helveticus–associated components present in the functional beverage [19]. Although microbial colonization and gut–lung axis interactions were not directly examined in this in vitro study, fermentation-derived metabolites and postbiotic components from L. helveticus have been reported to exhibit immunomodulatory activities, including regulation of cytokine production, enhancement of epithelial barrier function, and modulation of immune cell responses [20]. These properties are consistent with the enhanced epithelial repair capacity and reduced cytokine secretion observed in the present models, suggesting that microbiota-oriented functional matrices may indirectly support respiratory health.
In addition to microbiota-associated components, Mosla chinensis Maxim. represents a traditional medicinal herb with a long history of use for respiratory discomfort in East Asian medicine [21]. Contemporary pharmacological studies have reported that M. chinensis contains bioactive compounds with anti-inflammatory and antioxidant properties, which may contribute to the suppression of inflammatory mediator release and mitigation of oxidative stress [22]. These characteristics are particularly relevant in the context of infection-induced or environmentally triggered respiratory inflammation. The inclusion of M. chinensis in the functional beverage may therefore complement microbiota-modulating effects by directly supporting inflammatory regulation at the cellular level [23]. Beyond these two key components, the functional beverage incorporates thirteen additional medicinal and edible plant ingredients traditionally associated with respiratory comfort, immune balance, or mucosal support. Botanical ingredients such as Platycodon grandiflorus, Houttuynia cordata, Glycyrrhiza uralensis, and Perilla frutescens have been reported in previous studies to exhibit anti-inflammatory, antioxidant, or immunomodulatory activities [24]. Rather than acting through a single dominant pathway, these botanicals may collectively form a functional matrix that supports immune homeostasis and epithelial integrity through mild, multi-target regulation. Such a profile is characteristic of functional food formulations designed for long-term supportive use rather than acute therapeutic intervention [25].
From an application perspective, the present findings support the potential use of the functional beverage as a microbiota-modulating botanical nutritional strategy for maintaining respiratory health. By enhancing epithelial repair capacity while concurrently attenuating inflammatory and pro-fibrotic signaling, such formulations may help support lung resilience during periods of environmental stress, post-infectious recovery, or chronic low-grade inflammation. Nevertheless, several limitations should be acknowledged. This study was conducted exclusively in vitro, and the molecular signaling pathways underlying the observed effects were not directly investigated. In addition, the individual contributions of specific ingredients were not isolated. Future studies incorporating in vivo models, clinical evaluation, and microbiome-related endpoints are warranted to further validate and extend these findings.
Conflicts of Interest
All authors declare no conflict of interest.
References
- Lee Y, Jang TS, Kim JK (2024) Effects of Coronavirus Disease 2019 on Prevalence of Acute Respiratory Viruses: Changes during the Pandemic. J Glob Infect Dis 16: 27-32.
- Xie K, Zhang P, Li Y, Xia L (2025) The post-COVID-19 pulmonary sequelae: manifestations, mechanisms and treatment strategies. J Thorac Dis 17: 7414-7429.
- Bakakos A, Sotiropoulou Z, Anagnostopoulos N, Vontetsianos A, Cholidou K, et al. (2025) Anti-inflammatory agents for the management of COPD - Quo Vadis? Respir Med 248: 108396.
- Wu D, Liu W, Wang J, Chen W, Shi M, et al. (2024) Clinical Application Guideline of Combination With Traditional Chinese Medicine and Western Medicine in the Prevention and Treatment of Chronic Obstructive Pulmonary Disease. J Evid Based Med 18: 70024.
- Sotto AD, Vitalone A, Giacomo SD (2020) Plant-Derived Nutraceuticals and Immune System Modulation: An Evidence-Based Overview. Vaccines 8: 468.
- Arshad Z, Shahid S, Hasnain A, Yaseen E, Rahimi M (2025) Functional Foods Enriched With Bioactive Compounds: Therapeutic Potential and Technological Innovations. Food Sci Nutr 13: 71024.
- Özçam M, Lynch SV (2024) The gut-airway microbiome axis in health and respiratory diseases. Nat Rev Microbiol 22: 492-506.
- Chen N, Li L, Han Y, Chen Z (2025) The Role of Gut Microbiota in the Modulation of Pulmonary Immune Response to Viral Infection Through the Gut-Lung Axis. J Inflamm Res 18: 11755-11781.
- Pires L, González-Paramás AM, Heleno SA, Calhelha RC (2024) Exploring Therapeutic Advances: A Comprehensive Review of Intestinal Microbiota Modulators. Antibiotics (Basel) 13: 720.
- Taverniti V, Minuzzo M, Arioli S, Junttila I, Hämäläinen S, et al. (2012) In vitro functional and immunomodulatory properties of the Lactobacillus helveticus MIMLh5-Streptococcus salivarius ST3 association that are relevant to the development of a pharyngeal probiotic product. Appl Environ Microbiol 78: 4209-4216.
- Yu JS, Ho CH, Hsu YC, Wang JJ, Hsieh CL (2014) Traditional Chinese medicine treatments for upper respiratory tract infections/common colds in Taiwan. Eur J Integr Med 6: 538-544.
- Ravipati AS, Zhang L, Koyyalamudi SR, Jeong SC, Reddy N, et al. (2012) Antioxidant and anti-inflammatory activities of selected Chinese medicinal plants and their relation with antioxidant content. BMC Complement Altern Med 12: 173.
- Cambier S, Gouwy M, Proost P (2023)The chemokines CXCL8 and CXCL12: molecular and functional properties, role in disease and efforts towards pharmacological intervention. Cell Mol Immunol 20: 217-251.
- Angrisano T, Pero R, Peluso S, Keller S, Sacchetti S, et al. (2010) LPS-induced IL-8 activation in human intestinal epithelial cells is accompanied by specific histone H3 acetylation and methylation changes. BMC Microbiol 10: 172.
- Yang L, Xia H, Gilbertsen A, Smith K, Racila E, et al. (2023) IL-8 concurrently promotes idiopathic pulmonary fibrosis mesenchymal progenitor cell senescence and PD-L1 expression enabling escape from immune cell surveillance. Am J Physiol Lung Cell Mol Physiol 324: 849-862.
- Yue X, Shan B, Lasky JA (2010) TGF-beta: Titan of Lung Fibrogenesis. Curr Enzym Inhib 6: 10067.
- Ask K, Bonniaud P, Maass K, Eickelberg O, Margetts PJ, et al. (2008) Progressive pulmonary fibrosis is mediated by TGF-beta isoform 1 but not TGF-beta3. Int J Biochem Cell Biol 40: 484-495.
- Zheng H, Zhang L, Wang C, Wang Y, Zeng C (2005) Metabolic dysregulation in pulmonary fibrosis: insights into amino acid contributions and therapeutic potential. Cell Death Discov 11: 411.
- Taverniti V, Guglielmetti S (2012) Guglielmetti, Health-Promoting Properties of Lactobacillus helveticus. Front Microbiol 3: 392.
- Jach ME, Sajnaga E, Bumbul M, Serefko A, Borowicz KK, et al. (2025) The Role of Probiotics and Their Postbiotic Metabolites in Post-COVID-19 Syndrome. Molecules 30: 4130.
- Duan ZY, Sun YP, Wang ZB, Kuang HX (2024) Moslae Herba: Botany, Traditional Uses, Phytochemistry, and Pharmacology. Molecules 29: 1716.
- Khafaji AM, Khafaji SS (2025) The protective effects of the combination of Simmondsia chinensis extract and vitamin E on nephrotoxicity and hepatotoxicity in rats exposed to cisplatin: An anti-inflammatory and antioxidant study. Open Vet J 15: 3486-3504.
- Zhang Q, Su T, Pan Y, Wang X, Zhang C, et al. (2025) Malus hupehensis leaves: a functional beverage for alleviating hepatic inflammation and modulating gut microbiota in diabetic mice. Food Funct 16: 2972-2990.
- Ho TJ, Ahmed T, Shibu MA, Lin YJ, Shih CY, et al. (2024) A prospective review of the health-promoting potential of Jing Si Herbal Tea. Tzu Chi Med J 36: 1-22.
- Hong CE, Lyu SY (2025) Formulation Strategies for Immunomodulatory Natural Products in 3D Tumor Spheroids and Organoids: Current Challenges and Emerging Solutions. Pharmaceutics 17: 1258.
Citation: Lin Y-K, Lin Y-H, Chan S-T, Chiang C-F (2026) A Functional Beverage Containing Lactobacillus helveticus and Mosla chinensis Maxim Supports Respiratory Health In Vitro. HSOA J Altern Complement Integr Med 12: 672.
Copyright: © 2026 Yung-Kai Lin, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

