
Association between Clinical Parameters and Hemodynamics in Patients with Refractory Cardiomyopathy
*Corresponding Author(s):
Macedo FDivision Of Cardiology, Department Of Internal Medicine, Palmetto General Hospital, Hialeah, FL, United States
Tel:+1 7863023739,
Email:fmaccedo@gmail.com
Abstract
Objective: We sought to determine whether clinical outcomes are linked in patients who do not exhibit hemodynamic improvement in refractory cardiomyopathy.
Methods: Patients with a right atrial pressure decrease and a pulmonary wedge capillary pressure decrease were considered refractory to standard therapy. Baseline characteristics were compared using t-tests and chi-squared tests. Mortality was assessed by Cox hazard models and Kaplan Meier curves.
Results: In our study 55 patients (38%) were refractory to treatment with no improvement in Left Ventricular (LV) pressure and 88 (61%) responded to treatment with a corresponding improvement in LV pressure. The refractory group was more likely to be caucasian (p=0.01) exhibiting a lower baseline systolic blood pressure, supine heart rate and hemoglobin (p≤0.05). From baseline to discharge, the refractory group had a worsening in creatinine versus improvement in creatinine in those who responded to therapy (p=0.008), smaller reduction in weight (p=0.007), and smaller improvement in cardiac output (p<0.0001) and a larger decrease in left atrial volume (p=0.03) and LV end-diastolic volume (p=0.03). Rates for combined outcome of death and Heart Failure (HF) rehospitalization, HF rehospitalizations, or cardiovascular deaths did not differ between the groups.
Conclusion: Patients who failed to respond to therapy by lack of changes in hemodynamics exhibited worsening of creatinine, less improvement in weight, and cardiac output despite greater decreases in left atrial and LV end-diastolic volumes, compared to patients who had improvement in Pulmonary Capillary Wedge Pressure (PCWP) and Right Atrial Pressure (RAP).
INTRODUCTION
Despite recent advances in the treatment of acute decompensated heart failure, this condition remains a major public health problem. It exerts a substantial impact on morbidity, mortality, and costs, and therefore, represents a major burden for hospitals, patients, and society [1]. There are currently no adequate surrogate measures to indicate whether treatment of this entity has been successful, and despite their widespread use, symptoms and signs have limited usefulness and can be misleading.
In patients with acute decompensated heart failure, inotropes, vasopressors, and diuretics remain the cornerstone of therapy, but even with appropriate treatment, approximately 40% of patients are discharged from the hospital with unresolved congestion, which can lead to increased rehospitalization and mortality rates [2-4]. Fluid retention and congestion are responsible for 90% of HF hospitalizations and greater severity of congestion is associated with worse outcomes.
The clinical response to standard therapy can vary from patient to patient, and the widespread use of pulmonary artery catheters to monitor intra cardiac and left ventricular filling pressures has been advocated for years, despite the lack of strong evidence that they are beneficial for patients with decompensated heart failure. Current American Heart Association/American College of Cardiology guidelines designate the use of PACs to guide therapy as a level IIa recommendation.
Higher mortality rates have been reported for critically ill patients who required the use of a pulmonary artery catheter, although no excess risk was noted for those patients with heart failure. Based on these findings and the need to more definitively determine the benefits and harms of pulmonary artery catheters use in heart failure, the Evaluation Study of Congestive Heart Failure and Pulmonary Artery Catheterization Effectiveness trial was conducted. The results of this trial demonstrated no improvement in survival with pulmonary artery catheter-guided therapy compared to standard clinical assessment-guided therapy. However, it has been demonstrated that survival in acute decompensated heart failure is not related to an initial elevated filling pressure but it is best predicted by the pulmonary capillary wedge pressure equal or less than 16 mmHg during therapy and an elevated central venous pressure [5], as a surrogate of right atrial pressure is independently related to worsening renal function and mortality in cardiovascular patients [6].
The ability to reduce ventricles filling pressures and improve Cardiac Output (CO) with adequate heart failure treatment may identify patients with an irreversible or non-modifiable HF condition, with a potentially worse and short long-term prognosis where advanced heart failure therapies might be beneficial if applied at an earlier state. The purpose of the present study was to identify whether patients enrolled in the trial who did not exhibited improved hemodynamics with acute decompensated heart failure therapy exhibited different baseline characteristics and clinical outcomes compared to those who respond to therapy.
METHODS
The ESCAPE trial was a prospective, randomized trial of Pulmonary Artery Catheter (PAC) guided Heart Failure (HF) therapy in patients admitted to the hospital with severe symptomatic HF with a Left Ventricular Ejection Fraction (LVEF) ≤ 30%, systolic Blood Pressure (BP) ≤ 125 mm Hg, 3 months of symptoms despite Angiotensin Converting Enzyme (ACE) inhibitor and diuretic therapy, and at least one sign and one symptom of congestion. A total of 433 patients were randomized to either PAC-guided or clinical assessment-guided HF therapy, with 209 and 212 patients in each arm, respectively. The trial was conducted in the United States and Canada between 2000 and 2003 at 26 sites, and the primary endpoint was days alive out of the hospital within the first 6 months after randomization. As reported by the trial investigators, the primary endpoint was not significantly different between patients in the PAC-guided treatment group and those in the clinical assessment-guided treatment group [7].
In the present study, only those patients (n=143) who had paired right heart catheterization values at baseline and discharge were included in the analysis. We excluded 66 patients who did not have both baseline and final hemodynamic measurements. This analysis was conducted with the public release of the trial database.
The IRB for this project was reviewed and waived as exempt according to 45 CFR 46.104 code through Integ Review IRB and approved by the NIH National Heart, Lung and Blood Institute Committee before releasing us the data.
RATIONALE FOR THIS STUDY
Based on the difference between the final and baseline hemodynamic findings of patients enrolled in the PAC arm group of the trial, we defined refractory as those exhibiting a decrease in the Right Atrial (RA) pressure < 4 mmHg or < 28% and a decrease in the Pulmonary Wedge Capillary Pressure (PCWP) < 8 mm Hg or < 32%. Those who did not meet the definition of refractory to treatment were defined as non-refractory. The mean values observed in the trial were used to determine the group assignments.
STATISTICAL ANALYSIS
Data are presented as mean ± standard deviation for continuous variables and percentage for categorical variables, unless otherwise specified. Baseline demographic and hemodynamic characteristics were compared between responder and non-responder groups, with Chi-squared tests (or Fisher’s exact tests) used for categorical variables and t-tests used for continuous variables after performing tests for normality. Paired t-tests were used to detect differences in demographics and hemodynamics between baseline and hospital discharge. The combined outcome was death or HF rehospitalization at 6 months after randomization. Logistic regression analysis was performed to compare the impact of demographics and hemodynamic predictors on the combined outcome when adjusting for age, New York Heart Association (NYHA) functional class, systolic BP, supine Heart Rate (HR), serum creatinine, and LVEF at baseline. Survival analyses were performed with the Cox proportional hazard model and Kaplan Meier curves. STAT software for Mac (STAT Inc., College Station, TX, USA) was used for the analyses, and p values < 0.05 were considered statistically significant.
RESULTS
Clinical and hemodynamic parameters at baseline
55 (38%) patients were refractory and 88 (61%) non-refractory. Patients in both groups had a similar age (58 ± 12.2; 56.2 ± 13.7) and evidence of advanced HF with NYHA class III (11%,; 12%), NYHA IV (89%; 88%), peak VO2 (10.4 mL/kg/min; 9.4 mL/kg/min), LVEF (20%; 18.8%) of refractory and non refractory, respectively (p > 0.05) as seen in table 1. Refractory patients were more likely to be Caucasian than non-refractories (p = 0.01). Systolic BP (99.8 ± 13 vs. 110 ± 16 mm Hg), supine HR (79 ± 12 vs. 81 ± 15 beats/min), weight (83.6 ± 24 vs. 84.4 ±19 kg), and hemoglobin (12 ± 1.6 vs. 13.4 ±19 mg/dL) were lower in refractories then non-refractories (p ≤ 0.05).
|
Baseline Characteristics |
Refractory to treatment |
Non refractoryto treatment |
p value |
|
Age |
57.9 ± 12.2 |
56.2 ± 13.7 |
0.46 |
|
Male (%)/Female (%) |
39 (30)/ 16 (29) |
63 (72) / 25 (28) |
1 |
|
White (%)/Non White (%) |
39 (71) / 16 (29) |
43 (49) / 45 (51) |
0.01 |
|
NYHA Functional Classification |
|||
|
Class III (%) |
6 (38) |
10 (63) |
1 |
|
Class IV (%) |
49 (39) |
78 (61) |
1 |
|
Diabetes (%) |
12 (23) |
30 (34) |
0.18 |
|
HTN (%) |
31 (56) |
40 (45) |
0.23 |
|
Ischemia (%) |
31 (56) |
46 (52) |
0.73 |
|
EF (n) |
20.4 ± 6.6 |
18.8 ± 7.1 |
0.2 |
|
VO2 |
10.4 ± 5.0 |
9.5 ± 3.4 |
0.51 |
|
Systolic BP |
99.8 ± 13.2 |
110.0 ± 16.9 |
0.0002 |
|
HR supine |
79.7 ± 12.0 |
81.6 ± 15.4 |
0.44 |
|
BNP |
1247.9 ± 1103.8 |
1274.8 ± 1236.2 |
0.94 |
|
BUN |
35.4 ± 21.9 |
33.3 ± 19.8 |
0.55 |
|
Cr |
1.5 ± 0.6 |
1.5 ± 0.6 |
0.84 |
|
Weight (kg) |
83.7 ± 24.9 |
84.5± 19.5 |
0.82 |
|
Na |
136.3 ± 4.0 |
136.6 ± 4.8 |
0.65 |
|
Hb |
12.6 ± 1.7 |
13.3 ± 7.6 |
0.42 |
|
Hct |
38.1 ± 5.1 |
37.6 ± 4.9 |
0.56 |
|
Albumin |
3.7 ± 0.5 |
3.5 ± 0.5 |
0.04 |
|
MLHF score |
76.4 ± 17.7 |
72.7 ± 17.8 |
0.23 |
|
JVP |
2.4 ± 1.0 |
2.8 ± 1.0 |
0.049 |
|
Per Edema |
1.15 ± 1.2 |
1.4 ± 1.1 |
0.2 |
|
Global |
45.4 ± 23.0 |
44.0 ± 20.9 |
0.72 |
|
Orthopnea |
3.1 ± 1.1 |
3.4 ± 1.1 |
0.05 |
|
CI |
2.2 ± 0.5 |
1.85 ± 0.5 |
0.0009 |
|
CO |
4.19 ± 1.2 |
3.6 ± 1.13 |
0.009 |
Table 1: Baseline characteristics of refractories and non-refractories.
Clinical and hemodynamic changes from baseline to PAC discontinuation or discharge
From baseline to discharge, patients in the non-refractory group exhibited a greater reduction in weight (p = 0.007) and creatinine level (p = 0.008) than those in the refractory group (Table 2). Indeed, the creatinine increased slightly from baseline in the refractory group. CO increased more in the non-refractory group than in the refractory group (1.19 ± 2.6 vs. 0.49 ± 1.12 L/min, p < 0.0001). The Left Atrium (LA) and Left Ventricular End Diastolic Volume (LVEDV) decreased more between baseline and discharge in the refractory group than the non-refractory group (−0.15 ± 0.4 and −28.35 ± 81 mm vs. − 0.08 ± 0.6 and 0.3 ± 51 mm, respectively, p < 0.05).
|
Study End Points |
Refractory |
Non refractory |
p value |
|
Δ Weight |
−2.9 ± 3.5 |
− 4.5 ± 5.1 |
0.04 |
|
Δ Global |
22.2 ± 26 |
22.9 ± 23 |
0.91 |
|
Δ BNP |
−212 ± 887 |
− 846 ± 1059 |
0.1 |
|
Δ BUN |
1.1 ± 16 |
1.9 ± 18 |
0.79 |
|
Δ Cr |
0.12 ± 0.66 |
-0.04 ± 0.5 |
0.12 |
|
Δ Na |
-0.8 ± 3.5 |
− 1.7 ± 4 |
0.17 |
|
Δ Albumin |
− 0.13 ± 0.3 |
− 0.09 ± 0.4 |
0.68 |
|
Δ Peripheral edema |
− 1 ± 0.97 |
− 0.94 ± 1.1 |
0.14 |
|
Δ JVP |
−1.4 ± 1 |
− 0.96 ± 0.92 |
0.01 |
|
Δ CI |
0.2 ± 0.6 |
0.5 ± 0.7 |
0.04 |
|
Δ CO |
0.49 ± 1.12 |
1.19 ± 2.6 |
0.02 |
|
Δ EF |
-0.33 ± 9.8 |
0.2 ± 6.8 |
0.8 |
|
Δ EDV |
−28.35 ± 81 |
0.3 ± 51 |
0.24 |
|
Δ ESV |
−26.26 ± 88 |
1 ± 46 |
0.003 |
|
Δ LA vol |
− 0.15 ± 0.4 |
− 0.08 ± 0.6 |
0.59 |
|
Δ RA vol |
−2.6 ± 5.3 |
− 3.7 ± 6.5 |
0.53 |
Table 2: Clinical and Hemodynamic Changes from Baseline to hemodynamic monitoring discontinuation or discharge.
Survival analysis
The combined outcome of rehospitalizations and death from cardiovascular causes occurred in 33 patients (60%) in the refractory group and 46 patients (52%) in the non-refractory group (Hazard Ratio [HR] in the responder group 0.84, 95% Confidence Interval [CI] 0.51-1.26; p > 0.05). A total of 14 deaths (25%) due to cardiovascular events occurred in the in the refractory group and 18 (20%) in the non-refractory group (HR 1.38, 95% CI 0.15-12.3; p > 0.05) (Figures 1-3). Rehospitalization for HF occurred in 26 patients (47%) in the refractory group and 38 (43%) in the non-refractory group (HR 0.60, 95% CI 0.24-1.5; p > 0.05).
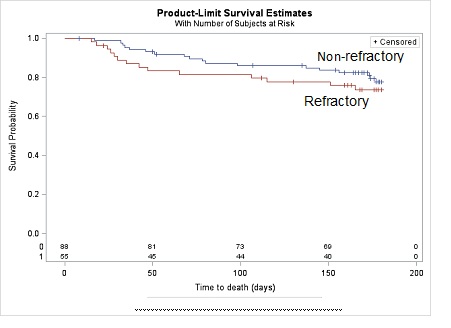 Figure 1: Survival Estimates by Response versus non-Response to Therapy.
Figure 1: Survival Estimates by Response versus non-Response to Therapy.
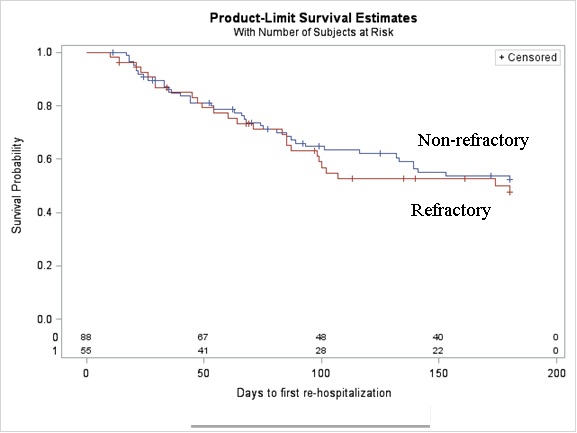 Figure 2: Readmission Estimates by Response versus non-Response to Therapy.
Figure 2: Readmission Estimates by Response versus non-Response to Therapy.
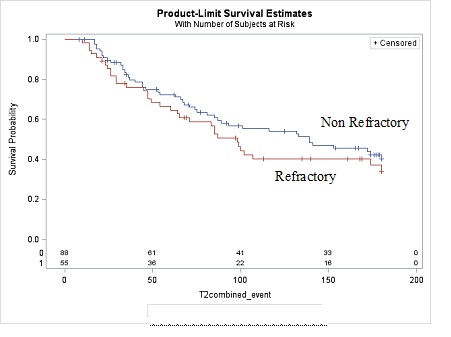 Figure 3: Combined Outcome for Heart Failure Events by Response versus non-Response to therapy.
Figure 3: Combined Outcome for Heart Failure Events by Response versus non-Response to therapy.
DISCUSSION
In this study of PAC-directed-therapy patients from the ESCAPE trial, we identified two distinct groups of patients based on the response in LV filling pressures to therapy. Those classified as refractories exhibited <4 mmHg or <28% reduction in RA pressure and <8 mmHg or <32% reduction in PCWP. In our study 38% were refractory to therapy. A recent study on the ESCAPE trial data showed that a high RA pressure/PCWP ratio, primarily driven by increased RA pressure, was associated with adverse events at 6 months but not with mortality [8]. They hypothesized that an exaggerated PCWP elevation response might lead to right ventricle dysfunction. It is possible that the refractory patients may have more extensive ventricular damage, making recovery less feasible. This finding correlates with the traditional view that a failing ventricle cannot improve its function despite a decrease in LV filling pressure, likely related to its position on the Frank Starling curve.
Refractory patients were not significantly different regarding baseline characteristics, i.e. they did not appear sicker except for lower BP and lower hemoglobin levels. The refractory group demographics showed that they were more likely to be Caucasian and have lower systolic BP, supine HR, baseline weight, and hemoglobin levels. Previous studies using ESCAPE, Organized Program to Initiate Lifesaving Treatment in Hospitalized Patients with Heart Failure (OPTIMIZE-HF), and the Heart function Assessment Registry Trial in Saudi Arabia (HEARTS) data have shown that lower systolic BP is associated with higher in-hospital and post-discharge mortality rates in ADHF [9-11]. In our findings the refractory patients had worsening of renal function and a lesser decrease in LA and LV volumes and a lesser increase in CO despite treatment. Testani, et al. showed that ESCAPE trial patients with larger admission-to-discharge systolic BP changes were significantly more likely to have worsening renal function (odds ratio 1.9, p = 0.017) [12]. Lowered hemoglobin levels are also considered to be poor prognostic markers. Ueda et al. (2014) showed that low hemoglobin levels were an independent risk factor for worsening renal failure within 1 year of discharge [13]. In an analysis of the Acute Decompensated Heart Failure Syndromes Registry (ATTEND), Kajimoto, et al. (2014) demonstrated that renal dysfunction coupled with anemia resulted in a higher risk for rehospitalization or mortality in patients with and without reduced ejection fractions (HR 1.65 and 1.54, respectively) [14]. The poor prognosis associated with the lower systolic BP and anemia could possibly be associated with the very advanced HF state but surprisingly our analysis did not demonstrate that refractory patients had differences in other variables associated with disease severity such as BNP, BUN, Creatinine, NYHA class, MVO2 EF, sodium and JVD.
Among the changes in hemodynamic findings, refractory patients exhibited an increase in creatinine, decreased weight loss, and reduced CO improvement. Analyses of the ESCAPE trial demonstrated that increased creatinine levels were a major mortality risk factor and that increased RA pressure/PCWP ratio was associated with higher baseline creatinine11 and reduced creatinine clearance [9]. Ueda, et al. (2014) used an >0.3 mg/dL or 25% increase in serum creatinine as their definition of worsening renal function [13]. It is possible that the increased creatinine observed in the refractory group may be associated with increasing renal and right ventricle dysfunction that may render therapies ineffective.
In regards to the weight loss, a study analyzing the ESCAPE trial showed no significant differences between weight change and mortality or rehospitalization, which was consistent with another trial using diuretics to reduce weight but not with a trial using ultrafiltration [15]. In another ESCAPE study, weight loss, along with increased CO, was associated with improved LVEF at the 3-month follow up [16]. Given these results, it seems likely that the decreased weight loss in refractories does not contribute to mortality or rehospitalization, but may be a result of the decreased recoverability of the ventricle. This is supported by the reduced CO improvement, which is also associated with poor LVEF recovery.
It should be noted that refractory patients did show greater reductions in LA volume, LVEDV, and LVESV compared with non-refractory patients. This should be a positive result. Studies of heart failure following myocardial infarction demonstrated that lower LA volume decreased the risk of heart failure [17]. Another study of systolic heart failure patients showed that reduced LVEDV decreased the risk of cardiovascular events [18]. Had the volume reductions been isolated results, it seems likely that improved outcomes would be observed; however, these improvements were likely insufficient to overcome the apparent LV failure presenting in these patients and the CO remained poor.
Overall, despite the poor prognostic indicators for the refractory patients to guideline directed medical therapy for heart failure, there was no significantly increased hazard for the refractory group with either mortality or rehospitalization, even though we were able to identify a subgroup of patients characteristics where further investigation is necessary (Caucasians with low weight, anemia and decreased BP at presentation). It is possible that these patients progressed beyond the point where current clinical or laboratory tests could distinguish between treatment non-refractory and refractory patients. Therefore, more studies are needed to identify adequate responsiveness markers for Advanced Acute Decompensated Heart Failure (ADHF).
STUDY LIMITATIONS
There are several limitations in our study. One limitation was the short-term follow-up period, which was only 180 days. Also, the number of patients in the trial with complete sets of laboratory and hemodynamic data at presentation and discharge was relatively small, which limited the study’s power. While limited power did not alter some of our findings, likely because of the large effect size, associations with factors that had smaller effect sizes may not have been detected. Although the trial is one of the largest contemporary datasets with detailed hemodynamic information regarding decompensated HF, because of its study design, hemodynamics data was only available for slightly less than one-half of the patients. The failure to find a difference in HF outcomes, including HF rehospitalizations and cardiovascular death, was possibly due to the small sample size, as there was a tendency toward a worse prognosis in the refractory patients. Additionally, since the treating physicians were not blinded to either medical-guided data or hemodynamics data, it is likely that treatment strategies were modified in response to those variables. Moreover, the hemoglobin, weight, and supine and resting BP values can be influenced by numerous factors, which were not controlled for in this study.
CONCLUSION
Our findings indicate that lack of significant change in PCWP, RAP, LA and LVEDV is seen in 30% of patients hospitalized with ADHF despite aggressive strategies. Additionally, were there no HF outcome differences between patients who did and did not respond to hemodynamics-guided therapy. While these results are internally consistent and in line with plausible pathophysiologic mechanisms, the small sample size and other methodological limitations may have impacted our results. Future research is necessary to replicate these findings in other cohorts and to identify optimal surrogate markers of successful AHDF treatment with the goal of reducing rehospitalizations and cardiovascular deaths.
REFERENCES
- Lloyd-Jones D, Adams R, Carnethon M, De Simone G, Ferguson TB, et al. (2009) Heart disease and stroke statistics--2009 update: A report from the american heart association statistics committee and stroke statistics subcommittee. Circulation 119: 21-181.
- Gheorghiade M, Follath F, Ponikowski P, Barsuk JH, Blair JEA, et al. (2010) Assessing and grading congestion in acute heart failure: A scientific statement from the acute heart failure committee of the heart failure association of the european society of cardiology and endorsed by the european society of intensive care medicine. European journal of heart failure 12: 423-433.
- Androne AS, Katz SD, Lund L, LaManca J, Hudaihed A, et al. (2003) Hemodilution is common in patients with advanced heart failure. Circulation 107: 226-229.
- Jain P, Massie BM, Gattis WA, Klein L, Gheorghiade M (2003) Current medical treatment for the exacerbation of chronic heart failure resulting in hospitalization. Am Heart J 145: 3-17.
- Stevenson LW, Tillisch JH, Hamilton M, Luu M, Chelimsky-Fallick C, et al. (1990) Importance of hemodynamic response to therapy in predicting survival with ejection fraction less than or equal to 20% secondary to ischemic or non ischemic dilated cardiomyopathy. Am J Cardiol 66: 1348-1354.
- Damman K, van Deursen VM, Navis G, Voors AA, van Veldhuisen DJ, et al. (2009) Increased central venous pressure is associated with impaired renal function and mortality in a broad spectrum of patients with cardiovascular disease. J Am Coll Cardiol 53: 582-588.
- Yancy CW, Jessup M, Bozkurt B, Redfield MM, Butler J, Casey Jr DE, et al. (2013) 2013 ACCF/AHA guideline for the management of heart failure: A report of the american college of cardiology foundation/american heart association task force on practice guidelines. J Am Coll Cardiol 62: 147-239.
- Drazner MH, Velez-Martinez M, Ayers CR, Reimold SC, Thibodeau JT, et al. (2013) Relationship of right- to left-sided ventricular filling pressures in advanced heart failure: Insights from the escape trial. Circulation. Heart Failure 6: 264-270.
- Gheorghiade M, Abraham WT, Albert NM, Greenberg BH, O'Connor CM, et al. (2006) Systolic blood pressure at admission, clinical characteristics, and outcomes in patients hospitalized with acute heart failure. JAMA 296: 2217-2226.
- O'Connor CM, Hasselblad V, Mehta RH, Tasissa G, Califf RM, et al. (2010) Stevenson LW. Triage after hospitalization with advanced heart failure: The escape (evaluation study of congestive heart failure and pulmonary artery catheterization effectiveness) risk model and discharge score. J Am Coll Cardiol 55: 872-878.
- Alhabib KF, Elasfar AA, Alfaleh H, Kashour T, Hersi A, et al. (2014) Clinical features, management, and short- and long-term outcomes of patients with acute decompensated heart failure: Phase i results of the hearts database. European journal of heart failure. Eur J Heart Fail 16: 461-469.
- Testani JM, Coca SG, McCauley BD, Shannon RP, Kimmel SE (2011) Impact of changes in blood pressure during the treatment of acute decompensated heart failure on renal and clinical outcomes. European journal of heart failure 13: 877-884.
- Ueda T, Kawakami R, Sugawara Y, Okada S, Nishida T, et al. (2014) Worsening of renal function during 1 year after hospital discharge is a strong and independent predictor of all-cause mortality in acute decompensated heart failure. Journal of the American Heart Association 3: 001174.
- Kajimoto K, Sato N, Keida T, Sakata Y, Takano T (2014) Acute Decompensated Heart Failure Syndromes I. Associations of anemia and renal dysfunction with outcomes among patients hospitalized for acute decompensated heart failure with preserved or reduced ejection fraction. Clin J Am Soc Nephrol 9: 1912-1921.
- Mehta RH, Rogers JG, Hasselblad V, Tasissa G, Binanay C, et al. (2009) Pulmonary Artery Catheterization Effectiveness Trial I. Association of weight change with subsequent outcomes in patients hospitalized with acute decompensated heart failure. Am J Cardiol 103: 76-81.
- Guglin M, Verma S, Chen R (2013) Association between weight loss and improvement of ventricular systolic function in advanced heart failure. Congest Heart Fail 19: 186-191.
- Verma A, Pfeffer MA, Skali H, Rouleau J, Maggioni A, et al. (2011) Incremental value of echocardiographic assessment beyond clinical evaluation for prediction of death and development of heart failure after high-risk myocardial infarction. American Heart Journal 161: 1156-1162.
- 18. Cameli M, Righini FM, Lisi M, Bennati E, Navarri R, et al. (2013) Comparison of right versus left ventricular strain analysis as a predictor of outcome in patients with systolic heart failure referred for heart transplantation. Am J Cardiol 112: 1778-1784.
Citation: Vieira JL, Diaz MA, Patton M, Valdes P, Foth C, et al. (2020) Association between Clinical Parameters and Hemodynamics in Patients with Refractory Cardiomyopathy. J Cardiol Stud Res 5: 016.
Copyright: © 2020 Jefferson Vieira, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

