
Autologous Barrier Membrane in Guided Bone and Tissue Regeneration: A 2-Years Prospective Case Series Pilot Study
*Corresponding Author(s):
Saptarshi MukherjeeDepartment Of Dental Surgery, Oral Implantologist, FICOI, Kolkata, India
Tel:+91 9163633444,
Email:cureassurance@gmail.com
Abstract
Background: Autologous Blood Concentrates have been subjected to various controversial debates in the past decade relating to its use in current dental surgeries. Its use as a stand-alone Barrier Membrane in a Guided Bone Regeneration has been questioned time and again due to its fast resorption rate. These randomized clinical trials are a collective attempt to shed some light in favour of its use as an independent barrier membrane in guided bone regeneration procedures.
Materials and Methods: 25 randomly selected patients in between the age group of 30-65 years were rehabilitated with 36 end osseous implant based prosthesis with the implant sites having some form of horizontal bone defects and needed augmentation procedures. These implants were placed and simultaneously GBR was done using 60:40 ratios of Autogenous to Xenografts (Bovine Origin) along with stand-alone membranes formed with Autologous Blood concentrates. All the implants were placed with an intention of not re-entering the site for any 2nd implant recovery surgeries. A minimum of 12 months’ follow-up of all the implant sites were monitored along with 1stmonth and 6thmonth CBCTs were examined.
Results: At 6 months’ follow-up, 30 out of 36 implants augmented with GBR and Autologous Blood Concentrate membranes, showed an average of 25% of graft width loss (0.65mm on average) and the rest 6 out of 36 implants showed a 40% on an average of graft loss due to remodelling. The residual bone graft that wasn’t lost was found to be stable without any Xenograft scattering in each evaluated case.
Conclusion: Barrier membranes formed out of Autologous Blood Concentrates used independently over a horizontal bone defect augmentation can give predictable outcomes if the site is not meant to be further re-entered for implant uncovering or any tissue regeneration procedures.
Keywords
Autologous blood concentrates; Barrier membranes; Guided bone regeneration; Horizontal bone defects; Xenografts scattering
INTRODUCTION
It’s almost two decades since the introduction of Autologous Blood Concentrates into dental surgeries for enhancing the healing abilities of a surgical wound. Initially, the main objective was to develop a procedure where platelet concentrates could be introduced into wounds by efficiently utilizing body’s natural healing capacity. Platelet Rich Plasma (PRP) and Platelet Rich Growth Factor (PRGF) were the first generation of Autologous Blood Concentrates (ABC) that was introduced. But the lacked efficacy due to their by-products which made it mandatory that an anticoagulant has to be simultaneously used with the First Generation Blood concentrates. Now with modified centrifugation and removing the anticoagulants, a more potent 2nd Generation of Blood concentrates has been derived and commonly known as Platelet Rich Fibrin (PRF) [1-4].
The main purpose of placing a barrier membrane to cover a GBR procedure is to protect the slow turning over and maturing bone graft from the fast growing soft tissue, infiltrating into the bone graft and hindering with the regeneration of the new bone to some extent. A variety of barrier membranes have been used to accomplish this purpose, including a PRF membrane. But earlier it was believed that PRF resorts in about 20 days, which makes it inefficient as a barrier membrane that has to protect the grafted site for a longer span of time.
With current trends of PRF researches and experiments it is understood that PRF can not only accomplish regeneration of soft tissue but when required and under strict protocols can protect the underlying bone tissues from any incoming pathogens while facilitating angiogenesis to the underlying bone graft structure.
MATERIALS AND METHODS
Patient enrolment
All patients referred or registered were from January 2018 to March 2020 for single or multiple implant placements in the Maxillary and Mandibular jaw in between 2nd bicuspids with the following criteria of inclusions and exclusions.
Following Inclusion Criteria were applied:
- • All patients were between the ages of 30 to 65 years.
- • Some form of Horizontal bone defects was present or was expected after implant placement.
- • The site of GBR did not have any notable infection as evaluated from the CBCT and IOPA.
- • Adequate oral hygiene as expressed by the modified plaque index and modified sulcus bleeding index from Mombelli, et al.
- • Sufficient mesio-distal space for placing an Implant and restoring it.
- • Sufficient bone height for placing the planned implant without the requirement of any vertical bone augmentation.
- • The patient is capable of understanding and giving a written consent for the procedure.
Following Exclusion Criteria were applied:
- • Medical and general contraindications for surgical procedures.
- • Presence of any active or uncontrolled periodontal disease.
- • Severe Bruxism.
- • An Active Smoker or Tobacco chewer.
- • History of Radiotherapy to the head and neck region or any metabolic bone disorder like osteoporosis.
Surgical and grafting procedure
25 randomly selected patients favouring the inclusion criteria mentioned above were rehabilitated for missing tooth/teeth with 36 end osseous dental implants that were from four different companies which had SLA treated surfaces and platform switching connections with internal Hex. The length of the minimum implant was 8mm and the maximum was 16mmwith required diameter as per the site dimension. After the placement of implants either the site was Contour Augmented or had some form of Dehiscence or Fenestration [5,6].
All the patients were given an antibiotic prophylaxis (1g Amoxicillin or 600mg Clindamycin if allergic to penicillin) 1 hour before surgery as well as a 0.2% chlorhexidine mouthwash (twice daily for 10 days) for oral disinfection.
The surgeries were performed maintaining all the sterilization protocols and under local inject able anaesthetics. Implants were placed as pre-planned with CBCT and planning software. The sites of horizontal defects were then grafted with 60% of autogenous bone harvested from potent sites in the particular jaw where the implant/ implants are being place with a bone scrapper or trephine burs (Figures 1 and 2) and the rest 40% was Xenograft (Bio-0ss, Giestlich Pharma) of Bovine Origin. The graft particles were mixed with a few drops of saline in a bone well (Figure 3).
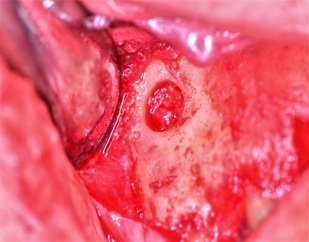 Figure 1: Area of bone harvested using trephine bur.
Figure 1: Area of bone harvested using trephine bur.
 Figure 2: Autologous harvested bone.
Figure 2: Autologous harvested bone.
 Figure 3: Mixture of autologous & xenograft in 60:40 ratio.
Figure 3: Mixture of autologous & xenograft in 60:40 ratio.
Phlebotomy was done from the Median Cubital Vein in the Antecubital fossa. Blood was collected in Red Cap Glass tubes (10ml) and was placed in the centrifuge in a balanced sequence. Without the anti-coagulants in the Red Cap Tubes the collected blood will clot and for that reason, centrifugation must be done immediately after the collection of blood. As per protocols, the derived blood in the tubes were centrifuged for 12 minutes at an RPM of 2700 (750g). From the tubes solid gel like structure of Fibrin Matrix was retrieved carefully separating the attached red blood corpuscles. These Fibrin Gels were placed in a PRF box and were pressed with its lid for 5mins to form it intomembranes made of fibrin mesh also known as PRF membranes (Figure 4).
 Figure 4: PRF matrix in gel form.
Figure 4: PRF matrix in gel form.
The pre-mixed bone graft particles were then carefully packed into the site of defects and then by using bone tags on the apical aspect of the graft and using the healing abutments (per-mucosal abutments) on the coronal aspect (Poncho Technique) (Figure 5) for stabilizing the PRF membranes the entire graft membrane complex was secured. Flaps were then closed tension-free and Vertical mattress suture followed by an interrupted suture was place with a resorb able suture material.
The surgeries were planned to be done in a single appointment and no further intervention was essentially contemplated for either implant recovery via 2nd surgery or any further soft tissue enhancing graft procedures were done.
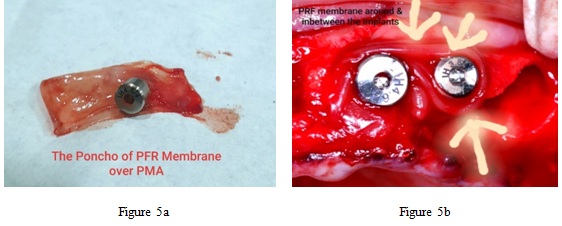 Figure 5: Showing poncho technique to stabilize the PRF membrane.
Figure 5: Showing poncho technique to stabilize the PRF membrane.
RESULTS
At follow-ups of 1 month and 6 months’ post-surgery with evaluations from CBCT and clinical investigation, it was seen that 30 out of 36 implants augmented with GBR and Autologous Blood Concentrate (PRF) membranes, showed an average of 25% of graft width loss (0.65mm on average) and the rest 6 out of 36 implants showed a 40% on an average of graft loss due to remodelling (Figures 6 and 7). The residual bone graft that wasn’t lost was found to be stable without any Xenograft scattering in each evaluated case.
Physical follow-ups at a year’s time showed good and stable new bone formation upon radiographic analysis (Figure 8).
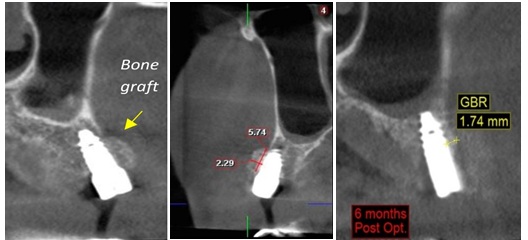 Figure 6: Case 1 showing CBCT of 1st month & 6th month post-op.
Figure 6: Case 1 showing CBCT of 1st month & 6th month post-op.
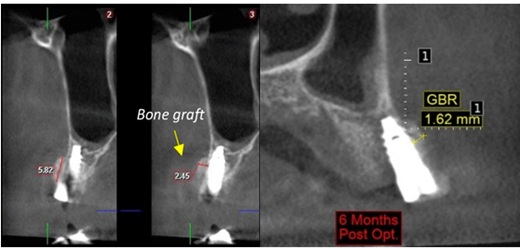 Figure 7: Case 2 showing CBCT of 1st month & 6th month post-op.
Figure 7: Case 2 showing CBCT of 1st month & 6th month post-op.
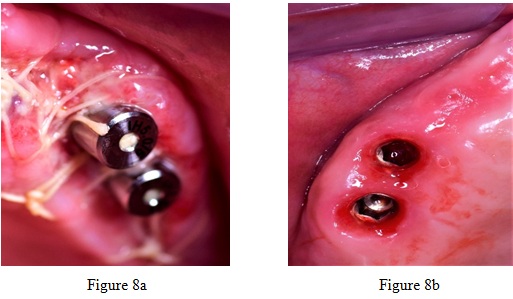 Figure 8: Showing 10 days & 6 months post surgery healing.
Figure 8: Showing 10 days & 6 months post surgery healing.
CONCLUSION
Following years of research worldwide and the above clinical evaluations of all the patients, it can be stated that Autologous Blood concentrates can be predictably utilized as a standalone barrier membrane for horizontal bone defects where re-entry is not expected.
The advantage is that the periosteum, which additionally contains many progenitor cells, is in contact with living cells from the PRF matrix as opposed to being entirely blocked by a non-living collagen membrane. Also an added enhancement of soft tissue quality and a remarkable quick healing of the surgical site is noticed with the use of PRF membranes.
REFERENCES
- Miron RJ, Choukroun J (2017) Platelet Rich Fibrin in Regenerative Dentistry: Biological Background and Clinical Indications: Biological Background and Clinical Indications, One. Wiley Online Library, New Jersey, USA.
- Eren G, Atilla G (2014) Platelet-rich Fibrin in the Treatment of Localized Gingival Recessions: A Split-Mouth Randomized Clinical Trial. Clin Oral Investig 18: 1941-1948.
- Kang YH, Jeon SH, Park JY, Chung JH, Choung YH, et al. (2011) Platelet-Rich Fibrin is a Bioscaffold and Reservoir of Growth Factors for Tissue Regeneration. Tissue engineering Part A 17: 349-359.
- Roy S, Driggs J, Elgharably H, Biswas S, Findley M, et al. (2011) Platelet-rich Fibrin Matrix Improves Wound Angiogenesis via Inducing Endothelial Cell Proliferation. Wound Repair Regen 19: 753-766.
- Simonpieri A, Choukroun J, Del Corso M, Sammartino G, Dohan Ehrenfest DM (2011) Simultaneous Sinus-Lift and Implantation Using Microthreaded Implants and Leukocyte- And Platelet-Rich Fibrin as Sole Grafting Material: A Six-Year Experience. Implant Dentistry 20: 2-12.
- Panda S, Sankari M, Satpathy A, Jayakumar D, Mozzati M, et al. (2016) Adjunctive Effect of Autologus Platelet-Rich Fibrin to Barrier Membrane in the Treatment of Periodontal Intrabony Defects. J Craniofac Surg 27: 691-696.
Citation: Mukherjee S (2020) Autologous Barrier Membrane in Guided Bone and Tissue Regeneration: A 2-Years Prospective Case Series Pilot Study. J Surg Curr Trend Innov 4: 041.
Copyright: © 2020 Saptarshi Mukherjee, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

