
Case Report: Platypnea-Orthodeoxia Syndrome -An Echocardigraphic and Clinical Diagnosis
*Corresponding Author(s):
Florian PapeDepartment Of Cardiology, HELIOS Klinikum Emil Von Behring, Berlin, Germany
Tel:+49 3081021313,
Email:florianpape@helios-kliniken.de
Abstract
Our report concerns a 75 year old lady with severe dyspnea primarily during physical activity in the upright position. Interestingly, the patient reports that some activities, such as swimming for example, are tolerated well.
The cardiopulmonary exercise testing detected highly restricted exercise tolerance plus hypoxemia at rest and increase during exercise. The transesophageal echocardiography showed a spontaneous right-to-left shunt through a patent foramen ovale. In addition, we found an elongated and dilated ascending aorta with almost horizontal outlet from the left ventricle.
We suspected a platypnea-orthodeoxia syndrome. The patent foramen ovale was closed interventionally with an Amplatzer-PFO-occluder. Subsequently, the patient improved significantly.
ABBREVIATIONS
AaDO2: Alveolar-arterial Oxygen Difference
Ao: Aorta
AT: Anaerobic Threshold
BF: Breathing Frequency
BR: Breathing Reserve
BSA: Body Surface Area
CPET: Cardiopulmonary Exercise Testing
HR: Heart Rate
LV: Left Ventricle
OUES: Oxygen Uptake Efficiency Slope
PA: Pulmonary Artery
paCO2: Partial Pressure of Carbon Dioxide
paO2: Partial Pressure of Oxygen
PCPW: Pulmonary-Capillary Wedge Pressure
PFO: Patent Foramen Ovale
PVR: Pulmonary Vascular Resistance
RA: Right Atrium
RER: Respiratory Exchange Ratio
RR: Blood Pressure (Riva-Rocci)
RV: Right Ventricle
SVR: Systemic Vascular Resistance
TEE: Transesophageal Echocardiography
VD: Dead Space Ventilation
VE: Minute Volume
VO2: Oxygen Uptake
VCO2: Carbondioxide Output
VT: Tidal Volume
CASE PRESENTATION
Our report concerns a 75 year old lady with severe dyspnea that progressed after a thoracic vertebral body fracture in 2009. The dyspnea was present at any physical activity in upright position such as walking. On the other hand, other types of physical stress, such as swimming, were tolerated without restrictions.
Her medical history included obstructive pulmonary disease (GOLD stage II) with emphysema. The patient had received supplemental oxygen consisting of three liters via nasal cannula in the months prior to presenting at our clinic. She suffered from osteoporosis which led to thoracic vertebral fractures in 2009 and 2011. In addition, systemic Arterial hypertension, mild renal insufficiency and repetitive events of deep venous thrombosis were reported.
The physical examination showed a cardiopulmonary compensated patient with clinical signs of airway obstruction. The laboratory tests revealed a slightly elevated hs-troponin, but normal d-dimers and brain natriuretic peptides. The oxygen partial pressure in sitting position at room air was 57 mmHg and did not improve significantly with six liters of oxygen via nasal cannula. In the supine position the oxygen partial pressure was 64 mmHg without oxygen supply. X-ray examination of the chest showed bilateral flattened diaphragms, overinflated apical lung segments, an elongated and ectatic ascending aorta, and degenerative changes in the thoracic vertebra.
The lung function test revealed a Forced Expiratory Volume (FEV1) of 61% predicted, the cardiopulmonary exercise testing a highly restricted exercise tolerance with limited maximal oxygen consumption of 8.8 l/min/kg (55% of the target), plus an hypoxemia at rest with an increase during exercise (CPET 1, Table 1). Pulmonary embolism was excluded by thoracic computer tomography.
| Parameter [unit] | CPET 1 | CPET 2 | Change | Change in % |
| Exercise capacity | ||||
| maximal workload [watts] | 9 (17% pred) | 50 (96% pred) | 41 | 82 |
| peak VO2 [ml/min/kg] | 8.8 (55% pred) | 14.6 (93% pred) | 5.8 | + 39.7 |
| VO2@AT [ml/min/kg] | 8.7 | 12.8 | 4.1 | 32 |
| peak RER | 1.01 | 1.09 | 0.08 | 7.3 |
| Cardiovascular System | ||||
| maximal HR [1/min] | 108 (74% pred) | 129 (89% pred) | 21 | +16.2 |
| Peak RR (syst./diast.) [mmHg] | 120 / 91 | 160 / 111 | 2 | 1.388889 |
| peak O2-pulse [ml/beat] | 5.7 (72% pred) | 7.9 (101% pred) | 2.2 | + 27.8 |
| OUES [l/min/log(l/min)] | 1404 (97 % pred) | 1794 (123 % pred) | 390 | +21.7 |
| Ventilatory System | ||||
| maximal BF [1/min] | 33 (79% pred) | 35 (83% pred) | 2 | + 5.7 |
| peak VE [l/min] | 34 (65% pred) | 44 (84% pred) | 10 | + 22.7 |
| BR [%] | 24 (87% pred) | 15 (54% pred) | -9 | - 37.5 |
| Gas-Exchange System and Dead Space Fraction | ||||
| maximal AaDO2 [mmHg] | 72.38 | 36.88 | -35.5 | -49 |
| VE vs. VCO2 slope | 40 | 31 | -9 | - 22.5 |
| paO2 at rest [mmHg] | 57.3 | 76.9 | 19.6 | + 25.4 |
| peak paO2 [mmHg] | 51.6 | 82.2 | 30.6 | + 37.2 |
| paCO2 at rest [mmHg] | 24.7 | 29.3 | 4.7 | +15.6 |
| peak paCO2 [mmHg] | 25.5 | 31.3 | 5.8 | + 18.5 |
| peak VD/VT | 24 | 20 | -4 | -16.6 |
Table 1: Cardiopulmonary Exercise Testing before (CPET 1) and after PFO closure (CPET 2).
A suspected intrapulmonary right-to-left shunt was not found in the lung scan. The transthoracic echocardiography exhibited normal left ventricular systolic function. The Transesophageal Echocardiogram (TEE) showed a spontaneous right-to-left shunt through a Patent Foramen Ovale (PFO, Figure 1a and Figure 1b). The invasive hemodynamic test excluded pulmonary hypertension (Table 2) in the supine position at rest. In addition, an elongated and dilated ascending aorta (angiographically 51 mm) with almost horizontal outlet from the left ventricle was detected. No signs or symptoms of coronary heart disease found.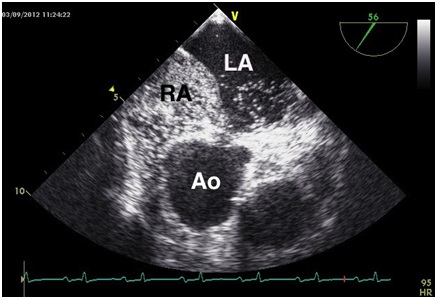
Figure 1a: Transesophageal echocardiography performed in the left lateral position.
Spontaneous right-to-left shunt through a patent foramen ovale visualized by bubbles (agitated saline).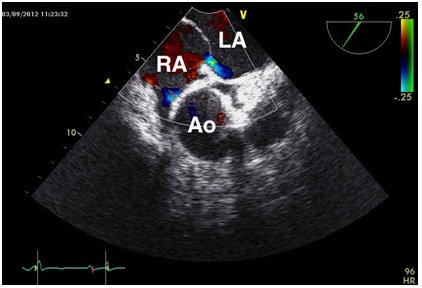
Figure 1b: Transesophageal echocardiography performed in the left lateral position.
Spontaneous right-to-left shunt through a patent foramen ovale visualized by color doppler.
| Parameter [unit] | Pressure values | Standard values |
| PCWP a / v / m [mmHg] | 11/10/7 | 4-12 / 5-15 / <12 |
| PA systolic / diastolic / mean [mmHg] | 21 / 7 / 12 | 15-30 / 4-12 / 9-19 |
| RV [mmHg] | 25 / 4 | 30 / 0-8 |
| RA a / v / m: [mmHg] | 6/5/4 | 3-9 / 2-10 / <8 |
| LV [mmHg] | 128 / 5 | <140 / 5-12 |
| Ao [mmHg] | 116 / 70 / 91 | <140 / 90 / 105 |
| Heart-index [l/min/m² BSA] | 2.27 | 2.8 - 4.2 |
| PVR [dyn x sec x cm-5] | 241 | 45 - 120 |
| SVR [dyn x sec x cm-5] | 1753 | 770 - 1.500 |
Table 2: Heamodynamic parameter before PFO-closure.
In the summary of findings we suspected an intracardiac shunt by foramen ovale, clinically evident as platypnea-orthodeoxia syndrome. The patent foramen ovale was closed interventionally with an Amplatzer-PFO-occluder (Figure 2). Subsequently, the patient improved significantly. The cardiopulmonary exercise testing in the post-interventional phase also showed significant improvement (CPET 2, Table 2).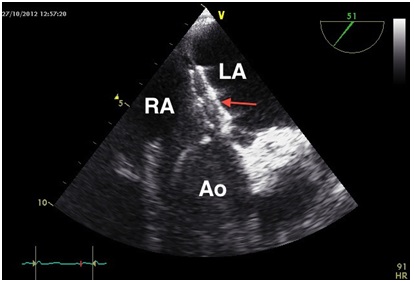
Figure 2: Interventionally closed Patent foramen ovale (arrow: Amplatzer-PFO-occluder 25 mm).
DISCUSSION
The platypnea-orthodeoxia syndrome was first described in 1949 by Burchell et al., [1]. It is not a stand-alone, single disease, but exactly a syndrome that has been reported in many diseases i.e., pneumonectomies [2], hepatopulmonary syndrome [3], adult respiratory distress syndrome [4] or intracardiac shunt [5].
Robin et al., [6] suggested the term "platypnea" to describe the dyspnea produced by assumption of an erect position which was relieved when lying, while the term "orthodeoxia" was coined to describe the accentuated arterial oxygen desaturation in the sitting or standing position improved by the supine one.
The hallmark of the syndrome is, as shown in our case, the postural change of PaO2. Go?mez et al., [3] using a receiver operating characteristic analysis determined in a 5% or 4 mmHg fall in upright PaO2 the cut-off threshold for the PaO2 decrease that can be defined an upright-induced deoxygenation (orthodeoxia).
By definition, the syndrome is characterized by a right-to-left shunt without fixed pulmonary hypertension. The etiology of the problem needs consideration in each case of a right-to-left shunt with normal right atrial pressures. As Cheng asked: "What causes water to flow uphill?" [7].
There are two basic components leading to a position-dependent right-to-left shunt: Firstly, there must be an anatomical shunt such as an inter atrial connection. To detect a shunt, a TEE can be helpful. The shunt can be either a patent foramen ovale, an atrial septal defect, or a fenestered inter atrial aneurysm [8,9]. A patent foramen ovale as underlying anatomical shunt is reported in 80% of the cases described in the medical literature [9]. A patent foramen ovale is usually asymptomatic, detected in a total of about 25% in adults [5,10,11].
Secondly, there has to be a functional component that promotes the right-to-left shunt. In a review published by Rodrigues et al., [11] two general mechanisms for the functional part are discussed: Hemodynamic changes leading to a temporary increase of pressure in the right atrium as well as flow phenomena which redirect the blood flow the direction of the anatomical shunt.
In our case no hemodynamic changes were obvious. The position-dependent deformation of inter atrial septum by the dilated aorta, the almost horizontal outlet that causes the direct passage of venous blood from the inferior vena cava directly towards inter atrial connection are possible flow phenomena. In addition, the compression of the right atrium led to a "spinnaker effect" of inter atrial septum, so that the preexisting anatomical defect became more relevant [9,12] (Figure 3). These effects have been discussed as possibilities.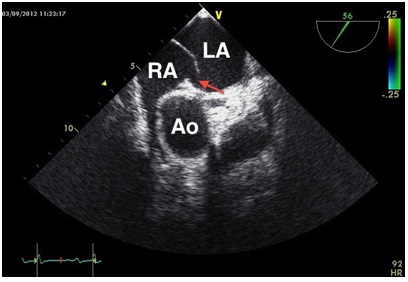
Figure 3: "Spinnaker effect" of the interatrial septum (arrow: patent foramen ovale).
As described above, the dilated ascending aorta seems to play a central role in the occurrence of the syndrome [9,13]. In a compilation of cases by Eichler et et al., [12] 63% of the patients with platypnea-orthodeoxia syndrome showed an aortic root of > 40 mm. Sanikommu et al., [8] compared anatomical factors in patients with diagnosed platypnea-orthodeoxia syndrome: Six of the seven patients had an elongated ascending aorta with horizontal outlet of the left ventricle.
Most of the patients with platypnea-orthodeoxia syndrome described in the scientific literature are older than 75 years [9]. The question is why this syndrome, with an underlying congenital defect, became evident this late in life. In our case the patient described a progression of dyspnea after multiple vertebral body fractures. The fractures resulted in an increase of her kyphoskoliosis. The risk of bone fractures increases with age. A kyphoskoliosis is described in about 9% of the cases [9]. Pathophysiologically the kyphoskoliosis causes a readjustment of the intra-thoracical organs and abets the shunt from right to left [14]. The dilatation of the ascending aorta is most likely the result of a long-standing arterial hypertension. The disease is therefore, despite of the underlying congenital anatomical factors, a disease of old age.
The transesophageal echocardiography performed in the left lateral position revealed, without Vasalva maneuver, a right-to-left shunt visualized by right ventricular "bubbles" (Figure 1). Remarkably the inter atrial septum aneurysm bulges towards the left atrium despite of invasively measured normal right atrial pressure. Kusajima et al., compared various positions during the TEE in patients with platypnea-orthodeoxia syndrome. In sitting and right lateral position they demonstrate a clear right-to-left shunt which declines when the patient is moved into the back or left lateral position [15]. A position change during the TEE examination, a tilt-TEE, could facilitate the diagnosis in some cases, as described by several groups [16]. In our case we assume that the shunt volume increased in upright position.
In summary, it is important to be aware of the platypnea-orthodeoxia syndrome because "one only recognizes what one looks for, and one only looks for what one knows"[17]. Against the background of an aging population the syndrome will become more relevant in future. For diagnosis of this syndrome the patient´s history is very important [18]. When the platypnea-orthodeoxia syndrome is suspected, the measurement of oxygen saturation in the horizontal and vertical position and a TEE to detect an intra-cardiac shunt, possibly with position change, are recommended. Therapeutically an interventional closure of inter atrial connection should be performed.
REFERENCES
- Burchell HB, Helmholz HF Jr, Wood EH (1949) Reflex orthostatic dyspnea associated with pulmonary hypertension. Am J Physiol 159: 563-564.
- LaBresh KA, Pietro DA, Coates EO, Khuri SF, Folland ED, et al. (1981) Platypnea syndrome after left pneumonectomy. Chest 79: 605-607.
- Gómez FP, Martínez-Pallí G, Barberà JA, Roca J, Navasa M, et al. (2004) Gas exchange mechanism of orthodeoxia in hepatopulmonary syndrome. Hepatology 40: 660-666.
- Khan F, Parekh A (1979) Reversible platypnea and orthodeoxia following recovery from adult respiratory distress syndrome. Chest 75: 526-528.
- Pierce CW (2010) Platypnea-orthodeoxia syndrome in an elderly woman with a patent foramen ovale. Can J Cardiol 26: 213-214.
- Robin ED, Laman D, Horn BR, Theodore J (1976) Platypnea related to orthodeoxia caused by true vascular lung shunts. N Engl J Med 294: 941-943.
- Cheng TO (2002) Mechanisms of platypnea-orthodeoxia: what causes water to flow uphill? Circulation 105: 47.
- Sanikommu V, Lasorda D, Poornima I (2009) Anatomical factors triggering platypnea-orthodeoxia in adults. Clin Cardiol 32: 55-57.
- Shiraishi Y, Hakuno D, Isoda K, Miyazaki K, Adachi T (2012) Platypnea-orthodeoxia syndrome due to PFO and aortic dilation. JACC Cardiovasc Imaging 5: 570-571.
- Kizer JR, Devereux RB (2005) Clinical practice. Patent foramen ovale in young adults with unexplained stroke. N Engl J Med 353: 2361-2372.
- Rodrigues P, Palma P, Sousa-Pereira L (2012) Platypnea-orthodeoxia syndrome in review: defining a new disease? Cardiology 123: 15-23.
- Eicher JC, Bonniaud P, Baudouin N, Petit A, Bertaux G, et al. (2005) Hypoxaemia associated with an enlarged aortic root: a new syndrome? Heart 91: 1030-1035.
- Faller M, Kessler R, Chaouat A, Ehrhart M, Petit H, et al. (2000) Platypnea-orthodeoxia syndrome related to an aortic aneurysm combined with an aneurysm of the atrial septum. See comment in PubMed Commons below Chest 118: 553-557.
- Teupe CH, Groenefeld GC (2011) Platypnea-orthodeoxia due to osteoporosis and severe kyphosis: a rare cause for dyspnea and hypoxemia. Heart Int 6: 13.
- Kusajima K, Natsumeda M, Sasaki H, Miyamoto K, Hirokami M, et al. (2011) Right-to-left shunting in the right lateral decubitus and upright positions: a variant of platypnea-orthodeoxia syndrome. J Am Coll Cardiol 57: 9.
- Nakahira A, Matsumura Y, Tatsumi H, Sasaki Y, Hirai H, et al. (2010) Platypnea-orthodeoxia diagnosed by sitting transesophageal echocardiography. Ann Thorac Surg 89: 1284-1286.
- Cheng TO (2004) Platypnea-orthodeoxia and blockpnea as two unrecognized or underdiagnosed causes of medically unexplained dyspnea. Chin Med J (Engl) 117: 1116.
- Ohfuji T, Obase Y, Ikeda M, Obase K, Hayashida A, et al. (2012) A case of platypnea orthodeoxia syndrome: a persistent history taking was the key to the diagnosis. Intern Med 51: 1701-1704.
Citation: Pape F, Schattke S, Richter C, Clemens H, Ammenwerth W, et al. (2015) Case report: Platypnea-Orthodeoxia Syndrome - an Echocardigraphic and Clinical Diagnosis. J Clin Stud Med Case Rep 2: 006.
Copyright: © 2015 Florian Pape, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

