
“Chaos in an Attempt to Appease”- A Case of Mycophenolate Induced Supraventricular Tachycardia
*Corresponding Author(s):
Nkechi MbaebieDepartment Of Internal Medicine, Berkshire Medical Center, Pittsfield, Massachusetts, United States
Tel:+1 5045050690,
Email:nmbaebie@bhs1.org
Abstract
Mycophenolate Mofetil (MMF or CellCept) has been known as a potent agent for immunosuppression especially used in the treatment of ongoing rejection in organ transplant. It is now used for recalcitrant conditions of dire autoimmune disorders especially in the setting of lack of response to first line agents.
MMF causes an array of gastrointestinal side effects such as nausea and vomiting, afebrile diarrhea, diffuse colitis. Others include insomnia, anxiety, easy bleeding and mental/mood disturbances. Although fast and irregular heart beat was identified as a serious but very rare side effect of MMF, its incidence remains very low and rarely reported.
We present a case of new onset supraventricular tachycardia two months after starting MMF for severe scleroderma in the setting of non-response to methotrexate therapy.
Keywords
Drug adverse effects; Mycophenolate mofetil; Supraventricular tachycardia
BACKGROUND
With the soaring increase in solid organ transplant, the need for highly potent immunosuppressive medications for the prevention of allograft rejections and autoimmune conditions has grossly increased.
Mycophenolate Mofetil (MMF or CellCept) has been known as a potent agent for immunosuppression in solid organ transplant and now used for recalcitrant conditions of dire autoimmune disorders especially in the setting of lack of response to first line agents [1].
As with many immunosuppressive therapies, it has numerous side effects but compared to other immunosuppressive agents such as tacrolimus, cyclosporine and azathioprine, it still has a lower toxicity profile making it an attractive alternative when patients with severe autoimmune disorders fail to respond to first line agents [2].
MMF has been known to cause an array of gastrointestinal side effects such as nausea and vomiting, afebrile diarrhea, diffuse colitis, insomnia, anxiety, easy bleeding and mental/mood disturbances [3]. Although fast and irregular heart beat was identified as a serious but very rare side effect of MMF, its incidence remains very low and rarely reported [4].
We present a case of a patient with multiple cardiac comorbidities who presented with new onset supraventricular tachycardia two months after therapy with MMF for severe scleroderma in the setting of non-response to methotrexate therapy.
CASE PRESENTATION
50 year old female with significant history of scleroderma, mixed connective tissue disorder, restrictive lung disease (on three liters of oxygen at home), non-ischemic cardiomyopathy with reduced ejection fraction of 25-30% status post in situ Implantable Cardioverter Defibrillator (ICD), paroxysmal atrial fibrillation on rivaroxaban, atrial flutter status post cardioversion and ablation. She was admitted with symptoms of palpitations, dizziness, lightheadedness, chest pain and shakiness after her ICD fired.
Chest pain was sharp, non -radiating associated with nausea. She also had shortness of breath on exertion with orthopnea. She was recently started on sacubitril/valsartan for heart failure as well as MMF for scleroderma and connective tissue disease just two months prior to presentation.
Examination revealed tachycardia with regular heart rhythm and significant dermatological manifestations of scleroderma (Figure 1).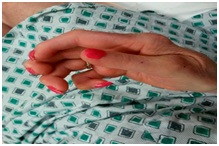
Figure 1: Scleroderma and mixed connective tissue affecting the hands.
INVESTIGATION
Labs revealed a normal thyroid stimulating hormone, potassium of 3.3; troponin peaked at 0.072, electrocardiogram revealed Supraventricular Tachycardia (SVT) with narrow QRS complex. Cardiology team was consulted and the ICD was interrogated which confirmed narrow complex SVT (Figures 2&3).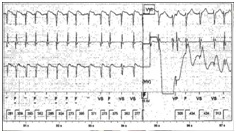
Figure 2: Implantable cardioverter defibrillator strip showing supra ventricular tachycardia with HR >180 which triggered implantable cardioverter defibrillator to fire.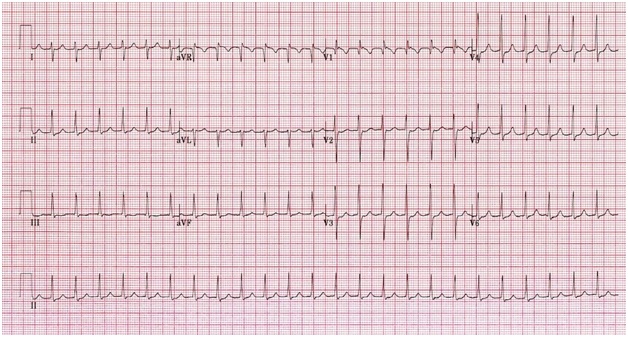
Figure 3: 12 Lead EKG showing SVT.
DIFFERENTIAL DIAGNOSIS
New onset supraventricular tachycardia due to possible drug adverse effect was suspected, other differentials were paroxysmal atrial tachycardia, accessory pathway between the hearts upper and lower chamber, two electrical pathways in the atrioventricular node were all suspected as possible etiologies.
TREATMENT
Patient’s home medication carvedilol was discontinued and metoprolol started as it has less anti-hypertensive effects than carvedilol which is as more effective AV nodal agent. Rheumatology consult obtained suggested discontinuing MMF since it could be a cause of tachycardia’s although rarely reported.
OUTCOME AND FOLLOW UP
Three days after MMF was discontinued, patient became symptom free and SVT changes on EKG disappeared. On four weeks follow up, patient was found to remain in sinus rhythm.
A decision was reached with patient participation to re-challenging her with MMF as this medication curtailed her autoimmune conditions effectively. Just three weeks into this challenge, her symptoms of palpitation returned and interrogation of her ICD revealed re-emergence of SVT. MMF was then discontinued indefinitely and patient was placed on a biologic therapy for her autoimmune conditions.
DISCUSSION
MMF has been a stable in the prevention of allograft rejection. However, it is now used in the treatment of autoimmune conditions such as psoriasis, rheumatoid arthritis, scleroderma and autoimmune uveoretinitis [1].
MMF is converted to mycophenolic acid which non-competitively inhibits the inosine monophosphate dehydrogenase enzymes required for purine synthesis in B and T lymphocytes. This sequentially causes a reduction in humoral and cytotoxic T cell response to immunogenic stimuli hence the effectiveness of this medication in these disease entities [5].
Despite it’s vital use, MMF has been known to cause certain side effects such as nausea, vomiting, afebrile diarrhea, insomnia, anxiety, easy bleeding, more serious side effects are diffuse colitis, mental/mood disturbance and fast and irregular heart beat which our index patient presented with [6]. The exact pathogenesis via which MMF leads to fast or irregular heart beat is unknown, however it is thought to occur due to insult to the cardiac conduction system and formation of toxic immunogenic reactions to the cardio-myocytes [5]. Clinically, MMF related cardiac insult may present as any form of arrhythmia, this untoward effect is very rare and only very few cases has been reported [4].
Our patient presented with SVT two months after starting MMF therapy, although she reported palpitation even sooner than 4 weeks after MMF use. SVT is new in this patient, as patient was adjudged to be stable from the cardiology standpoint prior to the start of this medication. Although our patient had multiple cardiac comorbidities and prior history of arrhythmias, which could have easily predisposed this presentation but the conviction however is that sinus rhythm was achieved with discontinuation of MMF and remains same even when followed up in the outpatient setting after four weeks. To further confirm that this medication was the main inciting agent, a re-challenge with this medication caused a re-emergence of SVT just after three weeks of use.
MMF induced tachyarrhythmia should be considered in a patient presenting with new onset arrhythmia after the start of this medication regardless of comorbidities.
LEARNING POINTS/TAKE HOME MESSAGE
• MMF induced arrhythmias are an uncommon but important clinical condition in patients presenting with palpitation and/or lightheadedness
• Early recognition of MMF induced arrhythmias along with discontinuation of this medication may result in improved outcome and should be considered in all patients presenting with new onset tachyarrhythmia after starting this medication
• A careful review of patient’s medication history and detailed review of their adverse events are very crucial in a patient presenting with new symptoms in the setting of multiple comorbidities
PATIENT PERSPECTIVE
Glad the doctors at Berkshire Medical Center were able to find out that this drug was not good for me and was responsible for my heart beating so fast and causing me so much discomfort. It will be good to thoroughly discuss all possible adverse effects of all medication with a patient before starting the medication so we can know what to expect and what to look out for.
REFERENCES
- Epinette WW, Parker CM, Jones EL, Greist MC (1987) Mycophenolic acid for psoriasis. A review of pharmacology, long-term efficacy, and safety. J Am Acad Dermatol 17: 962-971.
- Behrend M (2001) Adverse gastrointestinal effects of mycophenolate mofetil: Aetiology, incidence and management. Drug Saf 24: 645-663.
- Allison AC, Eugui EM (1996) Purine metabolism and immunosuppressive effects of Mycophenolate Mofetil (MMF). Clin Transplant 10: 77-84.
- Johal K, Ratuapli SK, Lam-Himlin DM, Gurudu SR (2014) Mycophenolate mofetil-induced segmental colitis mimicking ischemic colitis. Case Rep Gastroenterol 8: 95-100.
- Suthanthiran M, Morris RE, Strom TB (1996) Immunosuppressants: Cellular and molecular mechanisms of action. Am J Kidney Dis 28: 159-172.
- Liapis G, Boletis J, Skalioti C, Bamias G, Tsimaratou K, et al. (2013) Histological spectrum of mycophenolate mofetil-related colitis: Association with apoptosis. Histopathology 63: 649-658.
Citation: Mbaebie N, Shaw P (2018) “Chaos in an Attempt to Appease”- A Case of Mycophenolate Induced Supraventricular Tachycardia. J Clin Stud Med Case Rep 5: 055.
Copyright: © 2018 Nkechi Mbaebie, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

