
Journal of Alternative Complementary & Integrative Medicine Category: Medicine
Type: Commentary
Chronic pain in the Skin and Neurogenic Inflammation
*Corresponding Author(s):
James David AdamsDepartment Of Pharmacology And Pharmaceutical Sciences, University Of Southern California, Los Angeles, CA, United States
Tel:+1 3234421362,
Email:jadams@usc.edu
Received Date: Jun 21, 2017
Accepted Date: Jul 19, 2019
Published Date: Jul 26, 2019
Abstract
The treatment of any pain with oral preparations is dangerous and should be replaced by topical preparations or acupuncture. Pain is sensed in the skin, is most effectively and safely treated with topical preparations. Chronic pain is exacerbated in the skin by the pain chemokine cycle. Neurogenic inflammation comes from pain sensed in the skin and increases inflammation throughout the body. Inhibition of skin sensory neuron pain receptors relieves pain and inflammation.
Keywords
Chronic pain; Neurogenic inflammation; Pain; Pain chemokine cycle
INTRODUCTION
Most pain is sensed in the skin, even pain from broken bones, cancer, kidney stones and other internal causes [1,2]. This is because internal pain transmitting neurons communicate with pain sensing neurons in the skin that communicate with the brainstem [1,2]. Chronic pain involves a number of inter-related mechanisms in the skin and other areas [3,4]. Many receptors on skin sensory neurons are responsible for sensing pain and prolonging pain, especially Transient Receptor Potential (TRP) cation channels. Many different TRP channels exist on skin sensory neurons. Populations of specific TRP channels are found on different neurons. The pain chemokine cycle in the skin prolongs and worsens chronic pain [5,6].
Damage to or activation of a sensory neuron in the skin causes the release of chemokines that attract neutrophils and macrophages (Figure 1) into the skin [5,6]. Neutrophils secrete leukotrienes that activate TRP channels on skin sensory neurons causing pain [7]. Leukotrienes can have long half-lives, perhaps allowing them to induce pain in many neighboring neurons over a long period of time. Macrophages contain Cyclooxygenase 2 (COX2) and secrete prostaglandins that cause pain by activating prostaglandin receptors. Prostaglandins can enhance and prolong pain by trans activating TRP channels [5]. The skin contains skin resident T cells that respond to chemokines by releasing IL-17 [8]. Sensory neurons release more chemokines in response to IL-17 [5,6]. Most chronic pain is a self-sustaining pain chemokine cycle that makes the skin into a pain producing organ.
Damage to or activation of a sensory neuron in the skin causes the release of chemokines that attract neutrophils and macrophages (Figure 1) into the skin [5,6]. Neutrophils secrete leukotrienes that activate TRP channels on skin sensory neurons causing pain [7]. Leukotrienes can have long half-lives, perhaps allowing them to induce pain in many neighboring neurons over a long period of time. Macrophages contain Cyclooxygenase 2 (COX2) and secrete prostaglandins that cause pain by activating prostaglandin receptors. Prostaglandins can enhance and prolong pain by trans activating TRP channels [5]. The skin contains skin resident T cells that respond to chemokines by releasing IL-17 [8]. Sensory neurons release more chemokines in response to IL-17 [5,6]. Most chronic pain is a self-sustaining pain chemokine cycle that makes the skin into a pain producing organ.
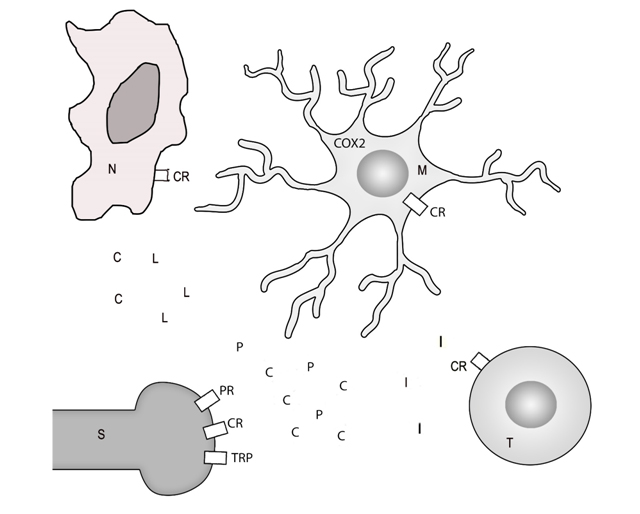 Figure 1: Neurogenic inflammation and chronic pain mechanics - A damaged sensory nerve terminal (S) in the skin releases Chemokines (C) that attract macrophages (M), bind to C Receptors (CR) and induce COX2 in macrophages. Macrophages secreate C that transitive neuronal TRP channels.
Figure 1: Neurogenic inflammation and chronic pain mechanics - A damaged sensory nerve terminal (S) in the skin releases Chemokines (C) that attract macrophages (M), bind to C Receptors (CR) and induce COX2 in macrophages. Macrophages secreate C that transitive neuronal TRP channels.SEXUAL DIFFERENCES IN CHRONIC PAIN
There are clear differences in male and female skin, including thickness, response to hormones and macronutrients [9,10]. These differences may be, in part, responsible for the differences reported in acute pain sensation and responses to acute pain between men and women [11]. However, differences in chronic pain between men and women in relation to skin differences have not been adequately explored.
NEUROGENIC INFLAMMATION
When TRP channels are activated, it causes the release of inflammatory proteins from sensory neurons in the skin, such as Calcitonin Gene-Related Peptide (CGRP), high Mobility Group Box 1 protein (HMGB1), neurokinin A and substance P [12]. This is the basis of neurogenic inflammation. The skin is important in inflammatory diseases such as arthritis and asthma [13].
CGRP released from skin sensory neurons is found at high levels in the synovial fluids of arthritis patients [14]. It is clear that CGRP contributes to the inflammatory process in arthritis. Monoterpenoids that inhibit skin TRP channels may decrease CGRP levels in synovial fluids and may be anti-inflammatory [14]. CGRP also induces pain, contributes to the pain that arthritis patients experience and contributes to chronic pain, which is induced by the pain chemokine cycle [15].
CGRP binds to a G protein coupled receptor called calcitonin receptor-like receptor (CLR,14). CLR activity can be greatly magnified by interactions of the receptor with receptor activity modifying protein (RAMP,14) which contributes to the pain and inflammation induced by CGRP.
HMGB1is secreted by activated macrophages, such as the monocytes/macrophages attracted into the skin by chemokines during the pain chemokine cycle [16]. HMGB1 interacts with Receptor for Advanced Glycation End products (RAGE), and Toll Like Receptors (TLR2 and 4), to induce the secretion of inflammatory cytokines [17]. Tumor necrosis factor α is a cytokine/adipokine that is involved in the induction of inflammation in arthritis. HMGB1 increases the secretion of tumor necrosis factor α from macrophages [18]. Tumor necrosis factor α can travel to remote sites, such as synovial tissues, to increase inflammation.
Neurokinin A is released by skin sensory neurons and enhances pain and inflammation [19]. There are several neurokinin receptors, NK1R, NK2R and NK3R, which are all G protein coupled receptors [20]. Substance P is another kinin that is released from skin sensory neurons [21]. Substance P also interacts with NK1R, NK2R and NK3R to induce pain and inflammation [21]. It is clear that both neurokinin A and substance Pare released from the skin, travel to remote sites and are involved in arthritis and other inflammatory conditions [21].
CGRP released from skin sensory neurons is found at high levels in the synovial fluids of arthritis patients [14]. It is clear that CGRP contributes to the inflammatory process in arthritis. Monoterpenoids that inhibit skin TRP channels may decrease CGRP levels in synovial fluids and may be anti-inflammatory [14]. CGRP also induces pain, contributes to the pain that arthritis patients experience and contributes to chronic pain, which is induced by the pain chemokine cycle [15].
CGRP binds to a G protein coupled receptor called calcitonin receptor-like receptor (CLR,14). CLR activity can be greatly magnified by interactions of the receptor with receptor activity modifying protein (RAMP,14) which contributes to the pain and inflammation induced by CGRP.
HMGB1is secreted by activated macrophages, such as the monocytes/macrophages attracted into the skin by chemokines during the pain chemokine cycle [16]. HMGB1 interacts with Receptor for Advanced Glycation End products (RAGE), and Toll Like Receptors (TLR2 and 4), to induce the secretion of inflammatory cytokines [17]. Tumor necrosis factor α is a cytokine/adipokine that is involved in the induction of inflammation in arthritis. HMGB1 increases the secretion of tumor necrosis factor α from macrophages [18]. Tumor necrosis factor α can travel to remote sites, such as synovial tissues, to increase inflammation.
Neurokinin A is released by skin sensory neurons and enhances pain and inflammation [19]. There are several neurokinin receptors, NK1R, NK2R and NK3R, which are all G protein coupled receptors [20]. Substance P is another kinin that is released from skin sensory neurons [21]. Substance P also interacts with NK1R, NK2R and NK3R to induce pain and inflammation [21]. It is clear that both neurokinin A and substance Pare released from the skin, travel to remote sites and are involved in arthritis and other inflammatory conditions [21].
CONCLUSION
Chronic pain and chronic inflammation are linked by the process of neurogenic inflammation. An initial inflammation results in skin sensory neuron activation and the release of inflammatory proteins from the neurons that can exacerbate inflammation. Chronic inflammation probably leads to chronic pain and more inflammation. On the other hand, an initial pain can cause the release of inflammatory proteins from skin sensory neurons leading to the release of inflammatory proteins that may induce inflammation in the body. Chronic pain may cause the chronic release of inflammatory proteins that enhance pain in other sensory neurons and may lead to chronic inflammation.
Osteoarthritis can be caused by adipokine secretion by visceral adipocytes [22]. The adipokines visfatin, leptin, resist in and tumor necrosis factor α are involved in causing cartilage and bone degradation in osteoarthritis. This degradation leads to pain that is sensed in the skin and establishes the pain chemokine cycle leading to neurogenic inflammation that exacerbates osteoarthritis [3,4].
Treating pain in the skin by inhibiting TRP channels is anti-inflammatory, as has been noted in several case reports [23,24]. Inhibiting skin TRP channels stops the pain chemokine cycle and stops neurogenic inflammation [25]. Effective pain therapy requires a topical preparation that is powerful enough to inhibit a large population of different TRP channels on many different sensory neurons.
Osteoarthritis can be caused by adipokine secretion by visceral adipocytes [22]. The adipokines visfatin, leptin, resist in and tumor necrosis factor α are involved in causing cartilage and bone degradation in osteoarthritis. This degradation leads to pain that is sensed in the skin and establishes the pain chemokine cycle leading to neurogenic inflammation that exacerbates osteoarthritis [3,4].
Treating pain in the skin by inhibiting TRP channels is anti-inflammatory, as has been noted in several case reports [23,24]. Inhibiting skin TRP channels stops the pain chemokine cycle and stops neurogenic inflammation [25]. Effective pain therapy requires a topical preparation that is powerful enough to inhibit a large population of different TRP channels on many different sensory neurons.
REFERENCES
- Ji R, Xu Z, Gao Y (2014) Emerging targets in neuroinflammation-driven chronic pain. Nature Rev Drug Disc 13: 533-549.
- Moran M, McAlexander M, Biro T, Szallasi A (2011) Transient receptor potential channels as therapeutic targets. Nature Rev Drug Disc 10: 601-20.
- Adams JD (2016) The effects of yin, yang and qi in the skin on pain. Medicines 3: 5.
- Adams JD, Wang X (2015) Control of pain with topical plant medicines. Asian Pacific J Trop Biomed 5: 93-95.
- Adams JD (2017) Chronic Pain - Can It Be Cured? J Pharmaceut Drug Devl 4: 105-109.
- Adams JD, Haworth IS, Coricello A, Perri F, Nguyen C, et al. (2017) The treatment of pain with topical sesquiterpenes: Frontiers in Natural Product Chemistry. Bentham Science Publishers, Sharjah, United Arab Emirates.
- Fernandes ES, Vong CT, Quek S, Cheong J, Awal S, et al. (2013) Superoxide generation and leukocyte accumulation: key elements in the mediation of leukotriene B?-induced itch by transient receptor potential ankyrin 1 and transient receptor potential vanilloid 1. FASEB J 27: 1664-1673.
- Heath WR, Carbonne FR (2018) The skin-resident and migratory immune system in steady state and memory: innate lymphocytes, dendritic cells and T cells. Nature Immunol 14: 978-985.
- Hew J, Solon-Biet SM, McMahon AC, Ruohonen K, Raubenheimer D, et al. (2016) The Effects of Dietary Macronutrient Balance on Skin Structure in Aging Male and Female Mice. PLOS One 11: 0166175.
- Dao H, Kazin RA (2007) Gender differences in skin: a review of the literature. Gender Med 4: 308-328.
- Bartley EJ, Fillingim RB (2013) Sex differences in pain: a brief review of clinical and experimental findings. Br J Anaesth 111: 52-58.
- Geppetti P, Nassini R, Materazzi S, Benemei S (2008) The concept of neurogenic inflammation. BJU Int 101: 2-6.
- Chen Y, Lyga J (2014) Brain-skin connection: stress, inflammation and skin aging. Inflam Allergy Drug Targets 13: 177-190.
- Russell FA, King R, Smillie SJ, Kodji X, Brain SD (2014) Calcitonin Gene-Related Peptide: Physiology and Pathophysiology. Physiol Rev 94: 1099-1042.
- Schou WS, Ashina S, Amin FM, Goadsby PJ, Ashina M (2017) Calcitonin gene-related peptide and pain: a systematic review. J Headache Pain 18: 34-51.
- Klune JR, Dhupar R, Cardinal J, Billiar TR, Tsung A (2008) HMGB1: endogenous danger signaling. Mol Med 14: 476-484.
- Sims GP, Rowe DC, Rietdijk ST, Herbst R, Coyle AJ (2010) HMGB1 and RAGE in inflammation and cancer. Ann Rev Immunol 28: 367-388.
- Cao L, Fetterer R, Zhang X, Tuo W (2010) Neospora caninum high mobility group box-1 (HMGB1) stimulates tumor necrosis factor-alpha (TNF-alpha) production by the murine macrophage cell line RAW264.7 (43.26). J Immunol 184: 43.
- Burbach GJ, Kim KH, Zivony AS, Kim A, Aranda J, et al. (2001) The Neurosensory Tachykinins Substance P and Neurokinin A Directly Induce Keratinocyte Nerve Growth Factor. J Invest Dermatol 117: 1075-1082.
- Frisch P, Bilkei-Gorzó A, Rácz I, Zimmer A (2010) Modulation of the CRH System by Substance P/NKA in an Animal Model of Depression. Behav Brain Res 213: 103-108.
- Campos M, Calixto JB (2000) Neurokinin mediation of edema and inflammation. Neuropeptides 34: 3154-3122.
- Adams JD, Parker K (2011) Extracellular and intracellular signaling. Royal Society of Chemistry, London, UK.
- Fontaine P, Wong V, Williams TJ, Garcia C, Adams JD (2013) Chemical Composition and Antinociceptive Activity of California Sagebrush (Artemisia californica). J Pharmacog Phytother 5: 1-11.
- Adams JD, Guhr S, Villasenor E (2019) Salvia mellifera-how does it alleviate chronic pain? Medicines 6: 18.
- Perri F, Coricella A, Adams J (2019) Monoterpenoids: the next frontier in the treatment of chronic pain? J Multidisc Sci J.
Citation: Adams JD (2019) Chronic pain in the Skin and Neurogenic Inflammation. J Altern Complement Integr Med 5: 073.
Copyright: © 2019 James David Adams, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
© 2026, Copyrights Herald Scholarly Open Access. All Rights Reserved!

