
Consideration to a New Standard for the Diagnosis of Lumbar Central Canal Stenosis by Quantitative MRI Assessment
*Corresponding Author(s):
Motoyuki IwasakiDepartment Of Neurosurgery, Otaru City Hospital, Hokkaido, Japan
Tel:+81 134251211,
Email:jzesso801@yahoo.co.jp
Abstract
The first aim of this study is to assess the Dural Sac Cross-sectional Area (DSCA) of the lumbar spinal canal in healthy young individuals with the appropriate posture when using MRI. A secondary aim was to determine if obtained DSCA values are correlated with each symptom. The group of 15 healthy individuals (group I) were scanned in the flexed and extended knee posture. The DSCA and Spinal Canal Cross-sectional Area (SCCA) were measured at the center of the pedicle level of each vertebral body. A group of 42 symptomatic patients (group II) were scanned, and the correlation with each symptom was statistically evaluated. In group (I), the mean DSCA was 178 ± 5.1 mm2 in the flexed posture and 158.9 ± 5.6 mm2 in the extended knee posture. At each level, there was a significantly smaller DSCA in the extended posture. In group (II), the mean DSCA was almost two-third of that in group (I). The DSCA at L1/2 and L2/3 showed a mild correlation with the Japanese Orthopedic Association score - IV (JOA - IV) (urinary disturbance score) (r2 = 0.16 and 0.21). Minimum DSCA was significantly correlated with JOA - IC (intermittent claudication) (p = 0.0003, r2 = 0.278). The number of inter vertebral levels with severe and moderate stenosis was assessed and a significant negative correlation was found with JOA - IC and IV (p = 0.0046 and 0.0275. r2 = 0.184 and 0.116, respectively).
It should be noted that these imaging values should be always considered with matched lumbar symptoms.
Keywords
ABBREVIATIONS
COSMIC: Fat - suppressed three - dimensional Coherent Oscillatory State acquisition for the Manipulation of Image Contrast
DSCA: Dural Sac Cross - sectional Area
JOA: Japanese Orthopedic Association score
SCCA: Spinal Canal Cross - sectional Area
INTRODUCTION
Previously, many morphologic spinal canal studies have been done using a plain radiograph or CT with or without a contrast medium [1-4] and cadaver [5]. Recent advancements in radiological techniques have enabled us to perform a faster and less invasive diagnosis, which resulted in the development of a new assessment method for the spinal canal with magnetic resonance imaging [6-12]. Fat - suppressed three - dimensional Coherent Oscillatory State acquisition for the Manipulation of Image Contrast (COSMIC) is a sequence based on fast imaging employing steady - state acquisition, which are more appropriate angle sliced spinal canal images taken in shorter time. COSMIC images are obtained from reconstructed original 3D scanned images, which can easily produce various thin sliced planes.
Several previous studies also measured anteroposterior diameter of the lumbar spinal canal or Dural Sac Cross-sectional Area (DSCA) of patients involved disc level, and then attempted to apply the numerical data to lumbar symptoms statistically, which resulted in no correlation (Table 1) [2,13-16]. However, the previous studies’ designs may have negatively affected the results. Moreover, some studies use upright MRI and axial loading of the spine [2,8,11,13,14]. Those studies suggest the importance of a more appropriate condition while taking images despite whether or not it is an expensive and complicated method. We decided on a unified, reasonable, and easy posture while taking images to avoid variable values in the same individual without using upright MRI or axial loading of the spine for daily clinical decisions.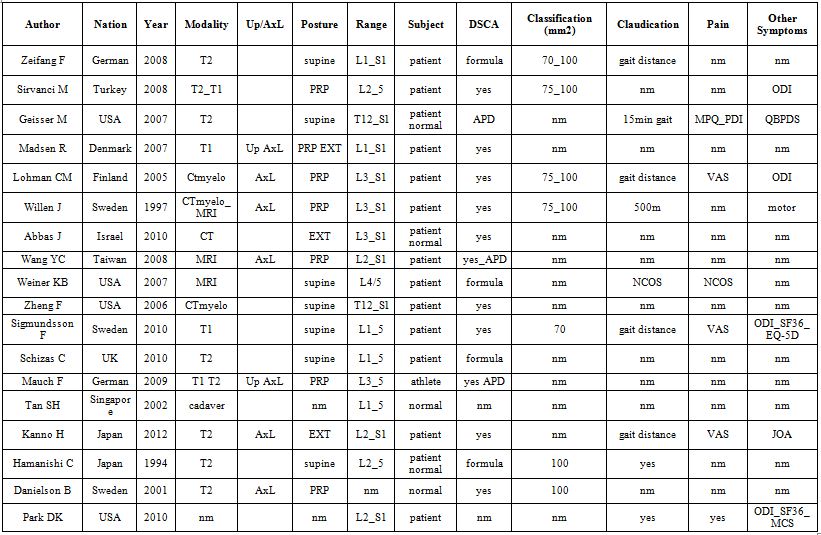
Table 1: Recent representative studies of lumbar canal stenosis.
Previous standards for lumbar central canal stenosis were <75 mm2 or 100 mm2 in the DSCA [2,3,15,16], which clinicians might not recognize as “lumbar canal stenosis” at a glance. Those standards could lead to many unimproved cases after decompression surgery. Furthermore, clinicians have to realize there may be coexisting stenotic lesions along with spinal canal stenosis when making decisions, as the Spine Patient Outcomes Research Trial (SPORT) study suggested [17]. This study might help achieve better patient outcomes when we decide to surgically treat lumbar central canal stenosis with decompression.
OBJECTIVE
Clarifying the correlation between lumbar symptoms and 3 - dimensional radiographical data leads to clinical decision making for all the spine surgeons.
MATERIAL AND METHODS
Fifteen healthy Japanese individuals and 16 patients with lumbago and/or leg pain were examined in two separate studies. A MRI was taken in the supine position by using the three - dimensional COSMIC method without axial loading.
Group (I) were 15 healthy individuals (mean age: 36.3 ± 3.3 years, 7 men and 8 women) who were scanned in the flexed and extended knee posture from Lumbar vertebra 1 (L1) to Sacral vertebra 1 (S1). Axial views were taken parallel to each intervertebral line (L1/2 - L5/S1) to acquire accurate slices by 3D remodeling. The DSCA and the Spinal Canal Cross-sectional Area (SCCA) at the center of the pedicle level of each vertebral body were measured in each posture to obtain the mean values of healthy Japanese people. We also assessed whether such an easy change of supine postures could make a significant difference.
Group (II) including 42 patients (mean age: 71.7 ± 1.8 years old, 26 men and 16 women) were enrolled. Each patient had a history of at least 12 weeks of symptoms of low back, leg pain or gait disturbance (e.g., intermittent claudication) and the degree of severity was ranged from mild to severe. They were scanned in the extended knee posture only (spinal extended position) to obtain the parameters, and then the correlation with each symptom was statistically evaluated. In order to assess the morphology of the spinal stenosis in 3 - dimension, the graphical parameters included as explanatory variables were the minimum DSCA in each patient’s lumbar spine, the number of intervertebral levels with a DSCA < 40 mm2 (severe stenosis) and < 80 mm2 (moderate stenosis). Outcome measures included as dependent variables were VAS, total Japanese Orthopedic Association - lumbar score (total JOA), JOA - lumbago score that consists JOA - IA, JOA - gait ability that matches the JOA - IC and JOA - urinary disturbance which corresponds to the JOA - IV.
STATISTICS
JMP 8 statistical software was used for statistical analysis in the study. When investigating the difference between groups, the Tukey - Kramer method was used. When investigating the correlation between graphical parameters and outcome measures, Pearson’s correlation was used. The level of significance was set at 0.05.
RESULTS
Group (I)
| DSCA | SCCA | |||
| Knee-flexed | Knee-extended | Knee-extended | ||
| Mean value | Group (I) | 178 ± 5.1 | 158.9 ± 5.6 | 203.6 ± 7.1 |
| Group (II) | 104.4 ± 4.0 | 196.5 ± 3.5 |
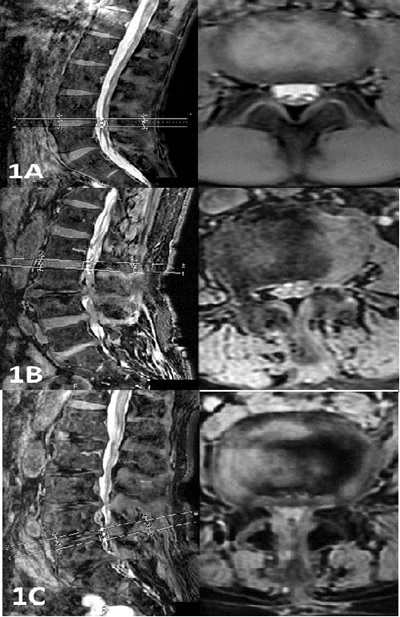
Figure 1: Examples of COSMIC images (A) Healthy individual/DSCA = 154 mm2, (B) moderate stenosis/DSCA = 72 mm2, and (C) severe stenosis/DSCA = 36 mm2.
| Group (I) | Group (II) | ||||
| Knee-flexed | Knee-extended | Knee-extended | Knee-extended | Knee-extended | |
| Mean DSCA | Mean DSCA | Mean SCCA | Mean DSCA | Mean SCCA | |
| L1/2 | 192.4 | 182.1 | 231.4 (L1) | 152.0 | 218.8 (L1) |
| L2/3 | 180.1 | 164.5 | 223.3 (L2) | 120.1 | 211.8 (L2) |
| L3/4 | 180.6 | 158.3 | 201.7 (L3) | 80.4 | 191.6 (L3) |
| L4/5 | 174.5 | 156.8 | 191 (L4) | 71.7 | 177.5 (L4) |
| L5/S1 | 162.2 | 132.9 | 170.9 (L5) | 123.8 | 173.0 (L5) |
Table 3: Mean DSCA and SCCA at each disc level in both groups.
Group (II)
| Number of severely affected levels | 25 |
| Number of moderately affected levels | 75 |
| Number of patients who have severely affected single level | 15 |
| Number of patients who have severely affected multilevel | 4 |
| Number of patients who have moderately affected single level | 13 |
| Number of patients who have moderately affected multilevel | 25 |
| Number of patients who underwent previous lumbar surgery | 4 |
The mean minimum DSCA was 47.6 ± 4.7 mm2. There were no significant correlations between the minimum DSCA and VAS, total JOA, JOA - IA, and JOA - IV (p = 0.3345, 0.1243, 0.1634, and 0.1024, respectively). However, a significant correlation was found between minimum DSCA and JOA - IC (p = 0.0003, r2 = 0.278) (Table 5); therefore, the patient who had a lower minimum DSCA was more likely to have intermittent claudication.
| Minimum DSCA | # <40 mm2 DSCA | # <80 mm2 DSCA | |
| VAS | 0.3345 | 0.6515 | 0.5663 |
| total JOA | 0.1243 | 0.1681 | 0.0715 |
| JOA-IA | 0.1634 | 0.4186 | 0.6153 |
| JOA-IC | 0.0003* (0.278) | 0.0046* (0.184) | 0.0090* (0.159) |
| JOA-IV | 0.4366 | 0.0275* (0.116) | 0.0079* (0.164) |
DISCUSSION
Previous studies of lumbar canal stenosis attempted to assess the relationship between symptoms and acquired values using various imaging modalities. There were some reports that found negative results [2,13-16]; however, we thought that the presence of more accurate study conditions when assessing the acquired values would lead to less pseudo-negative results, which would benefit clinicians who have to accurately diagnose conditions in outpatient settings daily.
In the group (I) session, healthy young Japanese individuals had almost the same DSCA (mean: 158.9 ± 5.6 mm2) regardless of the intervertebral level, except for between L1/2 and L5/S1 in the extended knee posture. We thought the constant DSCA value could be acceptable as a definition when assessing spinal canal stenosis. To regulate the settings conveniently when taking an MRI, the knee posture was assessed. The extended knee posture significantly made the DSCA narrower compared to the flexed knee posture. The extended knee posture was thought to be ideal due to its convenience without using either axial loading or upright MRI. Moreover, the COSMIC method always provides an accurate cross-sectional plane by using 3D remodeling. Previous reports did not necessarily specify the posture when taking images and the precision of the angle of the acquired plane. Recently, the cross-sectional area can be instantly measured by drawing the contour of the dural sac without any formula, such as Hamanishi et al., [9,12,16,18].
In the group (II) session, each patient’s minimum DSCA did not correlate with low back pain scores or urinary disturbance, such as VAS and JOA-IA/IV, but did correlate with JOA-IC, which indicates that even one level severe stenosis could cause gait disturbance as expected. Sigmundsson et al., reported that minimal DSCA did not correlate with estimated walking distance as a preoperative status [10]; however, this quantitative parameter does not accord with neurological intermittent claudication, which indicates that further research is needed.
It is known that some patients with radiographic spinal stenosis remain asymptomatic [19,20]; however, some patients have symptoms such as neurological claudication or pain. Ishimoto et al., reported that severe central stenosis was significantly associated with clinical symptoms, but only 17.5% of participants with severe central stenosis were symptomatic [21]. It is unclear why such differences are seen. We should also clarify the definition of “spinal stenosis.” The Wakayama Spine Study qualitatively rated canal stenosis according to the classification included in a general guideline [22]. Until recently, a DSCA of < 100 mm2 was most often used as a definition of spinal stenosis [6,18] while recent studies use a value of 70 - 80 mm2 as a definition of spinal stenosis [10,23]. Some authors divided the extent of stenosis in two points (DSCA of 75 -100 mm2) [2,3,15]. However, most of the studies have failed to demonstrate positive results when examining the relationship with symptoms. We considered the necessity of a clinically relevant classification, and as mentioned above, DSCA of < 40 mm2 was used as the objective diagnostic criterion for “severe spinal stenosis”, and DSCA of < 80 mm2 as “moderate spinal stenosis.” As a result, the number of severely or moderately affected levels did not correlate with the VAS of leg pain but did correlate with JOA - IC as intermittent claudication and JOA - IV as urinary disturbance, however, the severity of stenosis did not make difference. In a recent multicenter randomized trial, the Spine Patient Outcome Research Trial (SPORT), patients with multilevel spinal stenosis (with or without degenerative spondylolisthesis) were more likely to report neurogenic claudication [17]. They also reported that single level spinal stenosis patients were more likely to have a radicular distribution of their pain and depression compared to patients with more than 3 levels of stenosis. When we consider these results, the initial stage of spondylosis, such as one level of stenosis and a radiographically mildly compressed level, even though that is multilevel lesion, would more likely cause leg pain. In the later stage, spondylosis has developed to cause intermittent claudication by wide-expanding toward sagittal and axial, affecting the multilevel or single level. In short, the results of our study and the SPORT may have analyzed the same phenomenon from a different aspect.
Throughout the present study, total JOA and JOA - IA (low back pain) never correlated with DSCA. A few authors stated that total lumbar JOA scores were significantly correlated only with postoperative lower extremity VAS scores but not with the preoperative VAS and VAS for low back pain [24], which corresponds with our results. Low back pain occurs from various pathologies; therefore, we believe the total JOA score, including lumbar pain, is less likely to be associated with the radiographical values.
There were few reports that mentioned the relationship between the degree of dural sac compression and bladder symptoms. Tsai et al., concluded that anteroposterior diameter of the dural sac on an MRI was correlated with the American Urological Association Symptom Score [25]. Previous research has reported that the incidence of urinary disturbance was significantly higher in patients with disc herniation at L1/2 and 2/3 compared to lower levels (L3/4 - L5/S1) [26], which is similar to the result of the current study.
Finally, it should be noted that from a spine surgeon perspective, the decision for surgery will never be based on some DSCA value, but rather on both imaging data and clinical presentation because our study could not show the moderate/strong correlation between any imaging value and symptoms.
CONCLUSION
The COSMIC method is a useful, fast, and noninvasive technique to quantify the lumbar canal and dural sac. When researching spinal stenosis, the posture used when performing MRI has to be specified to obtain accurate numeric values. The patient analysis revealed that upper lumbar stenosis was correlated with urinary disturbance, neurogenic claudication was correlated with the minimum DSCA. The number of affected levels was correlated with intermittent claudication and urinary disturbance. The JOA and VAS of leg pain never correlated with radiographical stenotic values.
REFERENCES
- Abbas J, Hamoud K, May H, Hay O, Medlej B, et al. (2010) Degenerative lumbar spinal stenosis and lumbar spine configuration. Eur Spine J 19: 1865-1873.
- Lohman CM, Tallroth K, Kettunen JA, Lindgren KA (2006) Comparison of radiologic signs and clinical symptoms of spinal stenosis. Spine (Phila Pa 1976) 31: 1834-1840.
- Willén J, Danielson B, Gaulitz A, Niklason T, Schönström N (1997) Dynamic effects on the lumbar spinal canal: axially loaded CT - myelography and MRI in patients with sciatica and/or neurogenic claudication. Spine (Phila Pa 1976) 22: 2968-2976.
- Zheng F, Farmer JC, Sandhu HS, O’Leary PF (2006) A novel method for the quantitative evaluation of lumbar spinal stenosis. HSS J 2: 136-140.
- Tan SH, Teo EC, Chua HC (2002) Quantitative three-dimensional anatomy of lumbar vertebrae in Singaporean Asians. Eur Spine J 11: 152-158.
- Danielson B, Willén J (2001) Axially loaded magnetic resonance image of the lumbar spine in asymptomatic individuals. Spine (Phila Pa 1976) 26: 2601-2606.
- Kanno H, Ozawa H, Koizumi Y, Morozumi N, Aizawa T, et al. (2012) Dynamic change of dural sac cross-sectional area in axial loaded magnetic resonance imaging correlates with the severity of clinical symptoms in patients with lumbar spinal canal stenosis. Spine (Phila Pa 1976) 37: 207-213.
- Mauch F, Jung C, Huth J, Bauer G (2010) Changes in the lumbar spine of athletes from supine to the true-standing position in magnetic resonance imaging. Spine (Phila Pa 1976) 35: 1002-1007.
- Schizas C, Theumann N, Burn A, Tansey R, Wardlaw D, et al. (2010) Qualitative grading of severity of lumbar spinal stenosis based on the morphology of the dural sac on magnetic resonance images. Spine (Phila Pa 1976) 35: 1919-1924.
- Sigmundsson FG, Kang XP, Jönsson B, Strömqvist B (2011) Correlation between disability and MRI findings in lumbar spinal stenosis: a prospective study of 109 patients operated on by decompression. Acta Orthop 82: 204-210.
- Wang YC, Jeng CM, Wu CY, Chang HY, Huang YC, et al. (2008) Dynamic effects of axial loading on the lumbar spine during magnetic resonance imaging in patients with suspected spinal stenosis. J Formos Med Assoc 107: 334-339.
- Weiner BK, Patel NM, Walker MA (2007) Outcomes of decompression for lumbar spinal canal stenosis based upon preoperative radiographic severity. J Orthop Surg Res 2: 3.
- Geisser ME, Haig AJ, Tong HC, Yamakawa KS, Quint DJ, et al. (2007) Spinal canal size and clinical symptoms among persons diagnosed with lumbar spinal stenosis. Clin J Pain 23: 780-785.
- Madsen R, Jensen TS, Pope M, Sørensen JS, Bendix T (2008) The effect of body position and axial load on spinal canal morphology: an MRI study of central spinal stenosis. Spine (Phila Pa 1976) 33: 61-67.
- Sirvanci M, Bhatia M, Ganiyusufoglu KA, Duran C, Tezer M, et al. (2008) Degenerative lumbar spinal stenosis: correlation with Oswestry Disability Index and MR imaging. Eur Spine J 17: 679-685.
- Zeifang F, Schiltenwolf M, Abel R, Moradi B (2008) Gait analysis does not correlate with clinical and MR imaging parameters in patients with symptomatic lumbar spinal stenosis. BMC Musculoskelet Disord 9: 89.
- Park DK, An HS, Lurie JD, Zhao W, Tosteson A, et al. (2010) Does multilevel lumbar stenosis lead to poorer outcomes?: a subanalysis of the Spine Patient Outcomes Research Trial (SPORT) lumbar stenosis study. Spine (Phila Pa 1976) 35: 439-446.
- Hamanishi C, Matukura N, Fujita M, Tomihara M, Tanaka S (1994) Cross-sectional area of the stenotic lumbar dural tube measured from the transverse views of magnetic resonance imaging. J Spinal Disord 7: 388-393.
- Maus TP (2012) Imaging of spinal stenosis: neurogenic intermittent claudication and cervical spondylotic myelopathy. Radiol Clin North Am 50: 651-679.
- Tsutsumimoto T, Shimogata M, Yui M, Ohta H, Misawa H (2012) The natural history of asymptomatic lumbar canal stenosis in patients undergoing surgery for cervical myelopathy. J Bone Joint Surg Br 94: 378-384.
- Ishimoto Y, Yoshimura N, Muraki S, Yamada H, Nagata K, et al. (2013) Associations between radiographic lumbar spinal stenosis and clinical symptoms in the general population: the Wakayama Spine Study. Osteoarthritis Cartilage 21: 783-788.
- Suri P, Rainville J, Kalichman L, Katz JN (2010) Does this older adult with lower extremity pain have the clinical syndrome of lumbar spinal stenosis? JAMA 304: 2628-2636.
- Malmivaara A, Slätis P, Heliövaara M, Sainio P, Kinnunen H, et al. (2007) Surgical or nonoperative treatment for lumbar spinal stenosis? A randomized controlled trial. Spine (Phila Pa 1976) 32: 1-8.
- Haro H, Maekawa S, Hamada Y (2008) Prospective analysis of clinical evaluation and self-assessment by patients after decompression surgery for degenerative lumbar canal stenosis. Spine J 8: 380-384.
- Tsai CH, Chou EC, Chou LW, Chen YJ, Chang CH, et al. (2010) The evaluation of bladder symptoms in patients with lumbar compression disorders who have undergone decompressive surgery. Spine (Phila Pa 1976) 35: 849-854.
- Iwasaki M, Akino M, Hida K, Yano S, Aoyama T, et al. (2011) Clinical and radiographic characteristics of upper lumbar disc herniation: ten-year microsurgical experience. Neurol Med Chir (Tokyo) 51: 423-426.
Citation: Iwasaki M, Koyanagi I, Houkin K (2016) Consideration to a New Standard for the Diagnosis of Lumbar Central Canal Stenosis by Quantitative MRI Assessment. J Clin Stud Med Case Rep 3: 028.
Copyright: © 2016 Motoyuki Iwasaki, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

