
Designing a Model for Implementing Clinical Guidelines in Iran
*Corresponding Author(s):
Sorush NiknamianMilitary Medicine Department, Liberty University, United States
Email:so.niknamian@gmail.com
Abstract
Background
Clinical guidelines refer to a developed scientific statement to help physicians and patients for decision-making about the best care for special clinical conditions which can be an important document to shape evidence-based medicine.
Objective
This study has been conducted with the aim of designing a model for implementing clinical guidelines in Iran to enhance the quality of services.
Methods
The current research which has been of descriptive analytical type has been performed through combined quantitative-qualitative method in 2018. The statistical population consisted of 400 health managers and experts, and it was conducted through multistage sampling method in five regions of the country (North, South, Center, East, and West). Overall, 20 academic experts were chosen from each university. For data collection, a researcher-made questionnaire was used, and to measure the face and content validity, Content Validity Ratio (CVR) was used, while for determining the reliability, test-retest method as well as Cronbach alpha coefficient with a correlation of 0.934 was utilized. Also, to determine the validity of the items, exploratory factor analysis with Varimax rotation as well as Lisrel 8.8 and SPSS 24 was employed. Finally, to determine the fitness of the model, fitness indices were used.
Results
Out of the 63 factors identified in the questionnaire, based on CVR, 47 factors were confirmed by the experts. In the exploratory factor analysis, six economic (with seven components), organizational (with nine components), organizational culture (with eight components), the clinical guidelines feature (with eight components), insurance (with seven components), trusteeship of the healthcare system (with eight components) were identified as the main dimensions, accounting for 52.35% of the total variance of the variables. The economic dimension had the maximum effect on implementing clinical guidelines (0.90), while the clinical guidelines feature (0.63) and organizational culture (0.63) showed the minimum extent of effect on implementing clinical guidelines. After performing Confirmatory Factor Analysis (CFA), all factors were shown to lie within a desirable range and the model indicated goodness of fit and was confirmed.
Conclusion
Alongside the trend of corresponding and developing CPG, their usage should be planned. Evidently, imposing the mentioned interventions with the ultimate goal of sustainable behaviour change in providing healthcare services requires contribution of all practitioners, presentation of suitable facilities for implementing clinical guidelines based on evidence, time and personnel management, training methodology and planning, developing the necessary infrastructure, supervision, and developing professional and legal motivation.
Keywords
Clinical guidelines; Evidence-based medicine; Facilitator; Implementing clinical guidelines; Obstacles
Introduction
Background
The main reason of the different performance in healthcare is the gap between knowledge generation and its consequences. To fill this gap, in recent years, evidence-based method has attracted a great deal of attention [1,2]. Evidence-based method is an international priority in an attempt to improve the quality of healthcare and has been defined as integrating clinical experience with high quality evidence and patient priority [3,4]. Clinical guidelines refer to a developed scientific statement to help the patient and physician for decision-making about the best care for special clinical conditions, which can be an important document to shape evidence-based medicine [5,6]. Development and implementation of instructions aim to organize and present the best available evidence to support clinical decision-making in order to improve the health care quality, outcome of patients, effectiveness, and reduction of costs [7-11]. Accordingly, through different ways, clinical instructions help the executors to improve professional performance, quality of care, and the outcome of patients [12].
In countries such as USA and the Netherlands, around 30-40% of patients do not receive evidence-based healthcare. About 20% or more of the patients receive the treatments that are not required or are even sometimes harmful. Indeed, there is a gap between what is currently available and the best evidence-based medical treatment [13]. Not following instructions may result in inessential diagnosis and inadequate or unfocused treatment [14]. Evidence clearly suggests that there are many obstacles against the use of evidence and its implementation. The major obstacles include lack of time, lack of knowledge, lack of medical resources, negative attitude to evidence, and financial constraints [15]. In the investigation by Kabana et al, it was found that lack of awareness, limited familiarity, and disagreeing with clinical guidelines are the main obstacles in approving clinical guidelines. Furthermore, this investigation concluded that the main obstacle can be attributed to the simple fact that sometimes physicians are not aware of the existence of special clinical guidelines [12]. In addition, the findings of a qualitative study indicated that the obstacles for creating, developing, and implementing clinical guidelines include the context of practice, evidence-based healthcare systems, professionality of individuals, supportive policymakers and political context, innovation, and patients [16].
A study in Tehran indicated that only 31.8% of physicians were aware of clinical instructions for presenting health care services. The obstacles against implementation, according to these people, included lack of awareness of development of clinical instructions and evidence-based performance (37%), long way for employing clinical instructions (70%), unavailability (60%), lack of skill of physicians in using clinical guidelines (31%), and eventually absence of the necessary conditions and facilities (65%) [17]. Furthermore, the authors developed a framework in which these obstacles were categorized into three main groups: the obstacles related to physician’s knowledge (e.g. lack of awareness and unfamiliarity), obstacles affecting the attitude of physicians (e.g. absence of agreement and lack of motivation), and external factors (patient associated factors, the feature of guidelines, and environmental factors) [6,18,19]. Furthermore, the results obtained from a study in Iran showed that 39% of the participants had very little familiarity with EBM databases, and only 12.7% of these databases were very familiar [20].
The implementation strategies should be based on the current knowledge about effective interventions and evaluating the potential obstacles to approve guidelines [21,22]. Evaluating the obstacles and facilitators to implement clinical instructions is the first step in adapting and approving localized evidence [23]. Clinical instructions can improve the quality of healthcare services, but currently there are a number of challenges in adopting and implementing the instructions [8]. Considering the legal necessity to produce CPG in Iran, localized clinical instructions are rarely used to offer clinical services [16,17]. Therefore, recognizing and investigating these obstacles to enhance the quality of services seem to be essential. On the other hand, developed and translated clinical instructions are sparse in Iran, and their distribution is also limited or has become stopped [16].Therefore, given the investigations performed in Iran, there are problems in implementation of the guidelines including absence of adequate studies about the obstacles of implementation and unavailability of a suitable executive model. Therefore, to motivate the authorities to select development policies and use clinical guidelines, this study has been conducted with the aim of presenting a model for developing a system of producing and implementing clinical guidelines.
Objectives
This study has been conducted with the aim of designing a model for implementing clinical guidelines in Iran.
Methods
The present research which is a descriptive analytical study has been conducted by combined quantitative-qualitative method in 2018 across Iranian medical sciences Universities to achieve a suitable model for implementing clinical guidelines in Iran.
Setting
This study has been conducted in 2018 across medical sciences universities that are the executive of clinical guidelines in Iran.
Sample
The research population in the first stage was used from the common and available models regarding implementation of clinical guidelines as information resources. The second stage involved the professors of the fields of healthcare services management, the health economy, and the experts of developing clinical guidelines in Ministry of health and health policy-making. In the third and fourth stages, field study was done by academic professors, students, and executives of clinical guidelines as well as experienced experts in the field of healthcare system across the country. The sample size in the third and fourth stages consisted of 400 health managers and experts, chosen through multistage sampling method across five regions of the country (North, South, East, West, and East). Each region by allocating for universities and five quotas was chosen and then introduced into the cluster. Overall, 20 academic experts were chosen from each university.
Data collection
The required data were collected through investigating the literature of all related studies and performed on obstacles, facilitating factors, effective factors, awareness, and attitude in Iranian and global clinical guidelines in relevant databases. Further, collection of field data was performed using a researcher-made questionnaire to measure the influential factors in designing a model for implementing clinical guidelines. The questionnaire was designed in two sections. The first section included gender, age, and level of education, management background, workplace, and academic field. The second section consisted of 47 items about the influential factors in implementing clinical guidelines in the form of Likert scale ranging from absolutely agree [5], to absolutely disagree [1]. The responsivity rate was 100%.
Validity and reliability
To determine the qualitative face validity, a 20-person specialized panel was formed with the presence of healthcare specialists and experts of developing clinical guidelines to resolve the ambiguities of factors, lack of fit of factors, the objective and presentation of comments, whose comments were applied in the questionnaire. To examine the content validity quantitatively, Content Validity Ratio (CVR) coefficient was used, where the minimum acceptable value of this index based on the number of assessors was 0.42. In this study, out of the 72 variables in the initial questionnaire, given the CVR value, 47 variables were introduced into the final questionnaire. In order to examine reliability along the questionnaire, test-retest method was used. Accordingly, the questionnaire was distributed among 20 specialists and experts in two stages and the data were collected. After completion, the data were introduced into SPSS 24. For reliability over time, the correlation of the responses of the experts along the two stages of data collection was calculated, which was obtained as 0.934.
Data analysis
The collected data were analysed using Exploratory Factor Analysis (EFA) to determine the main effective components for implementing clinical guidelines, while Confirmatory Factor Analysis (CFA) was utilized to confirm the model. In addition, to analyse the current item, principal components analysis method with vertical rotation using vari max technique was employed. By performing exploratory factor analysis for the questionnaire and through vertical rotation, six main factors were identified. To investigate the goodness of fit of the measurement instrument (questionnaire), CFA was utilized with covariance approach via Lisrel 8.8 software. In addition, to investigate the type of data distribution, skewness and kurtosis coefficients were used. Finally, to investigate the research questions and hypotheses, independent one sample t-test as well as Friedman ranking test was used by SPSS 24.
Ethical considerations
To perform the research, official permission was presented as introduction letter to the research Department of the universities of interest. Completion of the questionnaire was performed with complete consent of the subjects, while ensuring confidentiality of the identity of the individuals.
Results
The results indicated that the mean age of the participants was 40.53 years old with a standard deviation of 6.86. The maximum frequency in the variable of gender was related to men with 230 (57.5%) subjects, while the maximum frequency regarding the level of education was associated with medical subspecialty with 118 individuals (29.5%). Considering working background, the greatest frequency was related to the group of 15-20 years with a frequency of 105 (26.3%). Also, regarding management background, the maximum frequency was related to the group of 5-10 years with the frequency of 128 (32%). Finally, the results showed that most people in the academic field of healthcare management with the frequency of 86 (21.5%) were respondent (Table 1).
|
Variables |
No. (%) |
|
|
SEX |
Male |
230(42.5) |
|
Female |
170(57.5) |
|
|
Age |
<30 |
46(11.5) |
|
30-35 |
43(10.8) |
|
|
35-40 |
77(19.3) |
|
|
40-45 |
90(22.5) |
|
|
>45 |
144(36) |
|
|
Education Level |
Master's Degree |
69(17.3) |
|
PhD |
76(19) |
|
|
Public Doctorate |
56(14) |
|
|
Medical expertise |
64(16) |
|
|
Super specialist doctor |
118(29.9) |
|
|
Fellowship |
17(4.3) |
|
|
Job Experience |
<5 |
38(9.5) |
|
5-10 |
65(16.3) |
|
|
10-15 |
94(23.5) |
|
|
15-20 |
105(26.3) |
|
|
20> |
98(24.5) |
|
|
Management Experience |
<5 |
76(19) |
|
5-10 |
128(32) |
|
|
11-20 |
121(30.3) |
|
|
>20 |
75(18.8) |
|
|
Field of Study |
Healthcare service management |
86(21.5) |
|
Medical |
76(19) |
|
|
Nursing |
40(10) |
|
|
Urologist |
20(5) |
|
|
Internist |
42(10.5) |
|
|
Specialist of infectious diseases |
2(./5) |
|
|
Pediatrician |
24(6) |
|
|
Medical files |
1(./3) |
|
|
Cardiologist |
14(3.5) |
|
|
Traditional medicine |
1(./3) |
|
|
Policymaking |
2(./5) |
|
|
Emergency specialist |
13(3.3) |
|
|
Neurologist |
7(1.8) |
|
|
General surgeon |
5(1.3) |
|
|
Health economics |
8(2) |
|
|
Midwifery |
9(2.3) |
|
|
Medical nutrition |
3(./8) |
|
|
Gynecologist |
8(2) |
|
|
ENT specialist |
5(1.3) |
|
|
Orthopedist |
4(1) |
|
|
Endocrinologist |
19(4.8) |
|
|
ocular surgeon |
7(1.8) |
|
|
Medical education |
4(1) |
|
Table 1: Demographic characteristics of the Study.
To investigate data fitness, for exploratory factor analysis, KMO test as well as Bartlett test was used. Since KMO was 0.911 and the significance number of Bartlett test was sig<0.05, the data were fit for conducting exploratory factor analysis (Table 2).
|
Significance level (p-value) |
Degree of freedom (df) |
Bartlett's Test of Sphericity |
KMO test |
|
0.00 |
1081 |
8073.36 |
0.911 |
Table 2: KMO and Bartlett's Test.
To identify the most important and effective influential components on implementing clinical guidelines and detect the loading value of each variable on the principal components, exploratory factor analysis via principal components analysis with rotation was used. By performing exploratory factor analysis with vertical rotation, six factors were identified. Overall these six factors accounted for 52.35% of the variance (Table 3).
|
Component |
Initial Eigenvalues |
Extraction Sums of Squared Loadings |
Rotation Sums of Squared Loadings |
||||||
|
Total |
% of Variance |
Cumulative % |
Total |
% of Variance |
Cumulative % |
Total |
% of Variance |
Cumulative % |
|
|
Economic |
12.521 |
26.639 |
26.639 |
12.521 |
26.639 |
26.639 |
4.652 |
18.796 |
18.796 |
|
Organizational |
3.901 |
8.300 |
34.940 |
3.901 |
8.300 |
34.940 |
4.458 |
8.508 |
27.304 |
|
Insurance |
1.975 |
4.203 |
39.142 |
1.975 |
4.203 |
39.142 |
3.752 |
7.486 |
34.793 |
|
Stewards |
1.920 |
4.086 |
43.228 |
1.920 |
4.086 |
43.228 |
3.684 |
6.695 |
41.488 |
|
Clinical |
1.564 |
3.328 |
46.556 |
1.564 |
3.328 |
46.556 |
3.469 |
5.963 |
47.451 |
|
Organizational Culture |
1.336 |
2.843 |
49.400 |
1.336 |
2.843 |
49.400 |
3.204 |
4.903 |
52.354 |
Table 3: Total explained variance of the Studied Components.
Extraction Method
Principal Component Analysis
To obtain a meaningful structure from factorial loads, based on conventional methods and using orthogonal rotation, the extracted factors were transferred to new axes which have non-right angle. Therefore, in exploratory factor analysis, 47 variables were categorized into six main principal components including the factors of insurance, organizational culture, the feature of clinical guidelines, economic factors, organizational factors, and trusteeship of the healthcare system. The insurance tariffs (0.668) (insurance factors), support of managers and decision-makers institutions (0.705) (out of healthcare system trusteeship factors), availability of budget and resolving budget constraints (0.644) (out of economic factors), developing adaptation of behaviour and approving guidelines with the factorial load of 0.538 (among organizational factors), adequate notification to the patient (0.819) (out of control factors), and existence of a clear protocol for production and adaptation (0.747) (out of the feature of clinical guidelines) are the most important influential factors on implementing clinical guidelines (Table 4).
|
Question |
The features of clinical guidelines |
Organizational culture |
Insurance |
Healthcare system trusteeship |
Organizational factors |
Economic factors |
|
|
B1 |
Correcting the payment system |
|
|
.585 |
|
|
|
|
B2 |
Aggregating the resources of insurer organizations |
|
|
.464 |
|
|
|
|
B3 |
Optimal and strategic purchase of services |
|
|
.490 |
|
|
|
|
B4 |
Insurance tariffs |
|
|
.668 |
|
|
|
|
B5 |
Supplying financial resources |
|
|
.647 |
|
|
|
|
B6 |
Preparing, designing and developing incentive packages by insurers |
|
|
.513 |
|
|
|
|
B7 |
Integrated function of insurer organizations |
|
|
.487 |
|
|
|
|
T1 |
The necessity of integrated healthcare system |
|
|
|
.414 |
|
|
|
T2
|
Improving the structure and resolving the methodological problems of guidelines |
|
|
|
.404 |
|
|
|
T3 |
Developing regulations and bylaws and supervising the implementation |
|
|
|
.476 |
|
|
|
T4 |
Existence of a practical evidence-based system |
|
|
|
.637 |
|
|
|
T5 |
Paying attention to professional features and the strategy of implementing instructions |
|
|
|
.444 |
|
|
|
T6 |
Support of managers and decision-makers institutions |
|
|
|
.705 |
|
|
|
T7 |
Time and personnel management |
|
|
|
.417 |
|
|
|
T8 |
Resolving simulation in clinical setting |
|
|
|
.430 |
|
|
|
E1 |
The necessary investments for realizing implementation of guidelines |
|
|
|
|
|
.413 |
|
E2 |
Financial profit in the using clinical guidelines |
|
|
|
|
|
.448 |
|
E3 |
Developing the necessary infrastructures for implementing guidelines |
|
|
|
|
|
.508 |
|
E4 |
Caring for the cost of access to evidence |
|
|
|
|
|
.465 |
|
E5 |
Caring for clinical uncertainty |
|
|
|
|
|
.459 |
|
E6 |
Availability of budget and resolving budget constraints |
|
|
|
|
|
.644 |
|
E7
|
Multidimensionality of health phenomenon |
|
|
|
|
|
.469 |
|
S1 |
Proper structure of the organization and development of the organization |
|
|
|
|
.487 |
|
|
S2 |
Interaction and cooperation in development sectors to implement the clinical guidelines |
|
|
|
|
.430 |
|
|
S3 |
Holding training courses in the academic education and constant training |
|
|
|
|
.426 |
|
|
S4 |
Caring for clinical guidelines in policymaking under macro conditions |
|
|
|
|
.481 |
|
|
S5 |
developing behavior adaptation and approving guidelines in case of availability of evidence and the alternative to the best method |
|
|
|
|
.538 |
|
|
S6 |
Ability, skill, and experience employing the research findings in the healthcare |
|
|
|
|
.425 |
|
|
S7 |
Familiarity, developing motivation, and attempts for teamwork
|
|
|
|
|
.412 |
|
|
S8 |
changing the attitude of employees to reliability, importance, and usefulness of instructions |
|
|
|
|
.463 |
|
|
S9 |
Attracting the cooperation and contribution of beneficiaries |
|
|
|
|
.417 |
|
|
L1 |
The potential of localizing the guidelines in the country |
.453 |
|
|
|
|
|
|
L2 |
Implementability of guidelines across all patients |
.676 |
|
|
|
|
|
|
L3 |
Practicalizing guidelines |
.485 |
|
|
|
|
|
|
L4 |
Convenience in implementing clinical guidelines |
.672 |
|
|
|
|
|
|
L5 |
Access to higher-quality evidence |
.698 |
|
|
|
|
|
|
L6 |
sufficient production and publication in the number and distribution of clinical guidelines |
.656 |
|
|
|
|
|
|
L7 |
availability of a clear protocol for production and adaptation |
.747 |
|
|
|
|
|
|
L8 |
resolving the ambiguity of clinical guidelines |
.594 |
|
|
|
|
|
|
F1 |
corresponding to patient needs |
|
.535 |
|
|
|
|
|
F2 |
interaction between physician and patient |
|
.624 |
|
|
|
|
|
F3 |
adequate notification to the patient |
|
.819 |
|
|
|
|
|
F4 |
developing collaboration and commitment for following up the course of treatment by the patient |
|
.587 |
|
|
|
|
|
F5 |
taking the patient preferences into account |
|
.554 |
|
|
|
|
|
F6 |
correspondence with previous experiences of using guidelines |
|
.451 |
|
|
|
|
|
F7 |
understanding the patient need to guidelines and patient adaptation |
|
.438 |
|
|
|
|
|
F8
|
physician understanding, generalizing the research findings to the patient population |
|
.439 |
|
|
|
|
|
Mean ± SD |
4.630±.41 |
4.59±.43 |
4.59±.42 |
4.66±.34 |
4.62±.34 |
4.54±.46 |
|
Table 4: Rotated factor matrixes.
Furthermore, to analyse the confirmed factor, the data and the studied responses were used according to the proposed structure. The results of fitness indices for the model are presented in (Table 5).
Suggesting approval of the studied index. Therefore, in general and considering the calculated indices, goodness of fit of the model can be concluded (Figure 1).
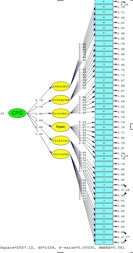 Figure 1: Suggesting approval of the studied index.
Figure 1: Suggesting approval of the studied index.
Discussion
The present research has been conducted with the aim of identifying the most important effective factors on creating a system for developing and implementing clinical guidelines and the loading value of each factor on the principal components in the context of Iran. Clinical Performance Guidelines (CPGs) are considered as one of the knowledge products with the aim of improving clinical services, helping in decision-making for physicians and patients, a means for insurance with the aim of controlling the costs, and helping governments in policymaking or prioritizing healthcare [24].
In this study, based on exploratory factor analysis 47 effective variables on implementing clinical guidelines were investigated in the form of six principal components including organizational culture, organizational factors, healthcare system trusteeship, feature of clinical guidelines, insurance, and economic factors. In the present study using a questionnaire, the extent of the impact of 47 variables was measured on implementing clinical guidelines for designing the model for implementing the guidelines.
Six groups of the influential factors were identified after analysis. In this regard, some of these factors have also been mentioned in other frameworks and models. For example, in “coordinated implementation model” the most important factors were organizational setting, education, economic factors, social, and administrative factors and eventually the personal environment of individuals and the role of information, motivation, existence of regulations, patient conditions [25]. Furthermore, Thrones mentioned factors including knowledge, attitude, and skills of patient and physician, organizational and economic factors as the obstacles against implementing guidelines. In this study, also the effect of support of managers, thought of leaders, professionality of individuals, organizational efficiency, the characteristic of colleague groups, and other service providers have been mentioned [26]. In another model which also propounds the clinical performance setting, a combination of economic, organizational, cultural, and policymaking effects has been mentioned as influential factors. In addition, it emphasizes the effect of the ability of implementation, awareness, and attitude of physicians [16]. Financial interventions in the present study referred to disciplinary and motivational interventions. Generally, financial interventions focus on service providers or patients [27]. As long as financial incentives and the payment system are not corrected, adhering to clinical guidelines cannot be expected [11].
In other studies, the obstacles against implementing clinical guidelines have been mentioned as knowledge, attitude, and behaviour change of patients and physicians, features of clinical guidelines including clarity and simplicity [28], environmental factors such as lack of adequate time and resources and the payment system problems [16]. Furthermore, in other relevant studies, the most common obstacles against implementation of guidelines in the healthcare system have been mentioned as research obstacles, limited support of colleagues, and obstacles of change [29]. Poor communication, deficiency of technical abilities of policymakers, limited capacity for conducting clinical research, constraint of resources, legal processes, infrastructures, equipment, and the recognized market and resolving the budget constraints, lack of valid support for altering the leadership and diminished leadership power were also mentioned as the main obstacles against implementing guidelines [5,12, 30-33]. In addition, factors such as presence of contradictory instructions and environmental factors such as lack of time, refund system [5], organizational constraints, and the increased understanding of the need to implementing clinical guidelines have been mentioned as obstacles against implementing clinical guidelines [34].
Absence of an evidence-based healthcare system has been one of the main reasons behind the attempts of economic development of Evidence-Based Practice (EBP) over the past decade in Iran [35]. Indeed, absence of an evidence-based healthcare system and improper political support on macro scale in Iran as a developing country have challenged the creation of a system for developing and implementing Clinical Performance Guidelines (CPG ) [36]. Studies conducted in Iran on the awareness and attitude of physicians about CPG have suggested inadequate familiarity of physicians as the most important factor in not implementing clinical guidelines [20,37,38].
Inadequate production and publication of CPGs might be one of the reasons of the unfamiliarity with CPG by physicians. In a study, most physicians stated that they had access to a limited number of CPGs and used them in their clinical practice. The findings of a study in the US indicated that over half of physicians have changed their clinical methods based on CPG, while 3% had no belief in it and did not use it in their practice [39]. Successful implementation of the guidelines involves development, publication, and implementation [6]. The organizations involved in production of guidelines should:
1) Develop the strategies for publicizing guidelines for timely delivery of information.
2) Reduce the complexity of clinical guidelines for physicians.
3) Produce clinical guidelines which are simple and understandable [40].
In qualitative studies conducted on the influential factors on implementing clinical guidelines, the factors were:
- Approving the subject and content.
- Approving the references.
- Organizational factors.
- Patient characteristics.
- The strategies of publication of journals [28].
A supportive organizational culture can act as a catalyst for EBP implementation with respective factors including available information, abundant sources for developing change, and skilful personnel to change the practice, having prerequisite skill and powerful leadership [41].
Therefore, the results of the present study and the similar results from other studies suggest that alongside the trend of Corresponding and Developing CPG, planning should also be done for their implementation. Evidently, imposing the mentioned interventions with the ultimate goal of sustainable behaviour change in healthcare service provision, contribution of all practitioners and presentation of suitable facilities for implementing clinical guidelines based on evidence, time and personnel management, training methodology and planning, developing the necessary infrastructures, existence of adequate supervision and leadership and developing professional and legal motivation are required.
Research limitations
Lack of awareness about clinical guidelines in some of the selected universities across the five chosen regions, no implementation of guidelines, and incompatibility with guidelines and uncooperativeness of some medical sciences universities.
Acknowledgement
This work has extracted from thesis of PhD. The authors would thank all who cooperated in our study, including the heads and employees of Ministry of Health and Medical Education.
Authors’ Contribution
Seyed Mojtab Hosseini and Mohammadkarim Bahadori developed the study concepts and design. Farzaneh Kimiaeimehr collected the data. Mohammadkarim Bahadori, Ali Maher and Farzaneh Kimiaeimehr analyzed and interpreted the data. Farzaneh Kimiaeimehr and Mohammadkarim Bahadori wrote the manuscript. Khalil Alimohammadzadeh revised and edited the manuscript. All authors read and approved the final manuscript.
Financial Disclosure
The authors declare no financial interests related to the material in the manuscript.
Funding/Support
The authors declare no funding/support.
Conflict of Interest
The authors declare no conflict of interest.
References
- Grol R, Grimshaw J (2003) From best evidence to best practice: Effective implementation of change in patients care. Lancet 362: 1225-30.
- Institute of Medicine (US) Committee on Clinical Practice Guidelines (1992) In: Field MJ, Lohr KN (eds,), Guidelines for Clinical Practice: From Development to Use. National Academies Press, Washington, USA.
- Richard G, Michel W, Michel E (2005) Improving patient care: The implementation of change in clinical practice. Elsevier Butterworth Heinemann Edinburgh 290.
- Nitu K, Jane D, George M, Cynthia B, N. SR. Guide Line implementability appraisal Yale Center for Medical Informatics,: New Haven, CT; 2011.
- Tord F, Johan H, Mats B, Aberg WA, Yvonne F (2010) Implementing clinical guidelines in psychiatry: A qualitative study of perceived facilitators and barriers. BMC Psychiatry 10: 1-10.
- Florian F, Kerstin L, Kristina K, Wolfgang G, Alexander K (2016) Barriers and Strategies in Guideline Implementation-A Scoping Review. Healthcare 4: 1-16.
- Alderaman MH, Furberg DC, Kostis JB, Laragh JH, Psaty MB, et al. (2002) Hypertension Guidelines: Criteria That might make them more clinically useful. Am J Hypertens 15: 917-923.
- Jaewon Y, Changsu H, Ho-Kyoung Y, Chi-Un P, Min-Jeong K, Sun-Young P, et al. Experiences and barriers to implementation of clinical practice guideline for depression in Korea. BMC Psychiatry. 2013;13(150):1-14.
- Fretheim A, Schünemann HJ, Oxman AD. Improving the use of research evidence in guideline development :5. Group processes. Health Res Policy Syst. 2006;4(17):27.
- van DSA, Marjo M, Bart SJ, Geert R, Henri K, der SMN-v, et al. Effectiveness of peer assessment for implementing a dutch physical therapy low back pain guideline:A cluster randomized controlled trial Phys Ther. 2014;94(10):1396-409.
- Reza M, Zahra B-S. How to Implement Clinical Practice Guidelines in Iran. Iranian Red Crescent Medical Journal. 2013;15(11). doi:10.5812/ircmj.9702.
- Ankele F, .C SM, Veer AJd, Patriek M. Factors influencing the implementation of clinical guidelines for health care professionals: a systematic meta-review. BMC Med Inform Decis Mak. 2008;8(1):38. doi:10.1186/1472-6947-8-38.
- J LM, David G. Are complementary medicine practitioners implementing evidence based practice? Complement Ther Med. 2011;19(3):128-36. doi:10.1016/j.ctim.2011.04.002. [PubMed:21641517].
- .MN VS. Long-term adherence to a local guideline on postoperative body temperature measurement: Mixed methods analysis. J Eval Clin Pract. 2012;18(4):841-7.
- Christian W, Andrzej L, Andrzej B, Bela M, Silvia P, Josep B. Barriers to implementation of evidence-based electrical therapies and the need for outcome research: role of European registries. Europace: European Pacing, Arrhythmias, and Cardiac Electrophysiology Journal of the Working Groups on Cardiac Pacing, Arrhythmias, and Cardiac Cellular Electrophysiology of the European Society of Cardiology. 2011;13:II18– II20.
- Zahra B-S, Sima N, Bahareh Y, Saharnaz N, Reza M. Barriers of Clinical Practice Guidelines Development and Implementation in Developing Countries: A Case Study in Iran. Int J Prev Med. 2013;4(3):340-8.
- Leila M, Saharnaz N, Reza M, Arash R, Jaleh G. Only One Third of Tehran's Physicians are Familiar with 'Evidence-Based Clinical Guidelines. Int J Prev Med. 2013;4(3):349-57.
- Sarah M, Manizheh A, Atefeh S, Nima K. Evidence-based medical practice in developing countries: The case study of Iran. J Eval Clin Pract. 2011;17:651-6. doi:10.1111/j.1365-2753.2011.01642.x.
- E LS, GAJ WM, Arie F, C.J SH, M.H CV, Huub W, et al. Study protocol: Cost effectiveness of two strategies to implement the NVOG guidelines on hypertension in pregnancy: An innovative strategy including a computerised decision support system compared to a common strategy of professional audit and feedback,a randomized controlled trial. . Implementation Science 2010;5:68.
- Farbod EA, Aziz R, Mousavi IH, Saber A-A, Khalil K, Feridun M. Evidence- based medicine performance among health care providers in Iranian hospitals: A nationwide survey. Med J Islam Repub Iran. 2017;31:77. doi:https://doi.org/10.14196/mjiri.31.77.
- R GA, C BM, A PV, Louise L-C, M GJ. How can we improve guideline use. A conceptual framework of implementability? Implem Sci. 2011;6: 26. Implementation Science 2011;6:26.
- Marjolein L, JudithMZegers-van S, .P WG, S BJ. Why don't physicians adhere to guideline recommendations in practice? An analysis of barriers among Dutch general practitioners. Implementation Science 2009;4(54). doi:doi:10.1186/1748-5908-4-54.
- S BJ, .M GRPT, M ZJO, H ST, der BAv, H.G M. Characteristics of effective clinical guidelines for general practice. Br J Gen Pract. 2003;53:15-9.
- Pille T, Marika R, Jarno H, Helvi T, Mari M, Suzanne H, et al. Barriers and facilitators to the implementation of clinical practice guidelines: A cross-sectional survey among physicians in estonia. BMC Health Serv Res. 2012;12:455.
- D CM, S RC, R PN, .W WA, H WM, C AP-A. Why don’t physicians follow clinical practice guidelines. A framework for improvement? . JAMA. 1999;282(15):1458–65.
- Thorsen T, Mäkelä M. Changing professional practice: theory and practice of clinical guidelines implementation. DSI Danish Institute for Health Services Research and Development; 1999.
- Alan B, Robert.G B. Analyzing qualitative data. New York:: Taylor & Francis e-Library; 2002.
- Arash R, Martin.p E, Ian R. Falling on stony ground. A qualitative study of implementation of clinical guidelines’ prescribing recommendations in primary care? . Health Policy. 2008;85:148-61. doi:10.1016/j.healthpol.2007.07.011.
- Homayoun S-B, Sadegh TJ, Saber A-A. Barriers to evidence-based medicine: a systematic review. Journal of evaluation in clinical practice. 2014;20(6):793-802.
- A HA, Adrijana C, J WP, Azza E-S, Harith G, Hossein M, et al. National policy-makers speak out: are researchers giving them what they need? Health Policy and Planning. 2011;26:73-82. doi:doi:10.1093/heapol/czq020.
- D OA, Atle F, J SH, SURE. Improving the use of research evidence in guideline development: introduction. Health Res Policy Syst 2006;4:12.
- A DD, Anne T-V. Translating guidelines into practice.A systematic review of theoretic concepts, practical experi ence and research evidence in the adoption of clinical practice guidelines 1997;157(4):408-16.
- Michel W, der Wtv, richard G. Implementing guidelines and innovations in general practice: which interventions are effective? Bitish Journal of General Practice 1998;48:991-7.
- Mazrou SHA. Expected benefits of clinical practice guidelines: Factors affecting their adherence and methods of implementation and dissemination. Journal of Health Specialties. 2013;1(3).
- Zahra B-S, Reza M. Evidence-based health care, past deeds at a glance, challenges and the future prospects in Iran. Iranian J Publ Health. 2012;41(12):1-7.
- Reza M, Bahareh Y, Saharnaz N, Jaleh G, Sharareh A. Strengthening evidence-based decision-making: is it possible without improving health system stewardship? Health Policy Plan. 2012;27:499-504.
- Sara A-A, Akbar S, Farhad H. Knowledge and attitudes of trainee physicians regarding evidence-based medicine: a questionnaire survey in Tehran, Iran. J Eval Clin Pract. 2008;14(5):775-9.
- Morteza G, Saber A-A, Fatemeh PA, Mozhgan F, Mohammad M, Taraneh T. A systematic review on barriers, facilities, knowledge and attitude toward evidence-based medicine in Iran. J Anal Res Clin Med. 2015;3(1):1-11. doi:10.15171/jarcm.2015.001.
- M WR, K SL, M WR. Family physicians’ opinions and attitudes to three clinical practice guidelines. J Am Board Fam Pract. 2004;17:150-7.
- Heiko S, Mei S, E PD, Tanja B, Julie F-H, Krishna A. Aravamudhan K. Barriers to implementing evidence-based clinical guidelines: a survey of early adopters The Journal of Evidence-based Dental Practice. 2010;10(4):195-206. doi:10.1016/j.jebdp.2010.05.013.
- Angela K, L SL, Lehan MM, G TM. Facilitating the implementation of evidence-based practice through contextual support and nursing leadership Journal of Healthcare Leadership. 2015:729-39.
Citation: Sorush Niknamian (2022) Designing a Model for Implementing Clinical Guidelines in Iran. J Phys Med Rehabil Disabil 8: 72.
Copyright: © 2022 Sorush Niknamian, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

