Different Cell Types Involved in Mediating Concanavalin A Induced Liver Injury: A Comprehensive Overview
*Corresponding Author(s):
Claude LibertDepartment Of Biomedical Molecular Biology, Ghent University, Ghent, Belgium
Tel:+32 93313700,
Email:Claude.Libert@IRC.VIB-UGent.be
Abstract
Autoimmune Hepatitis (AIH) is a chronic liver disease where presentation of a self-antigen peptide by professional antigen presenting cells initiates liver damage and drives the differentiation of uncommitted CD4 helper T cells to a Th1 and Th17 response. However, the details of the pathogenic mechanisms remain unclear and current treatments stay non-specific, making the need for reliable animal models high. Due to the activation and recruitment of T lymphocytes to the liver, the Concanavalin A (ConA) model is a proper animal model to investigate most forms of acute liver injury in humans such as AIH, acute liver failure and acute viral hepatitis in which T cell infiltration is observed. Intravenous injection of ConA leads to severe liver damage by necrotic cell death of hepatocytes and release of alanine and aspartate transaminases into the blood. In this review, we present an overview of the current understanding of the model, with an emphasis on the cell types involved in ConA-induced liver injury.
Keywords
ABBREVIATIONS
ALF: Acute Liver Failure
AIH: Autoimmune Hepatitis
ConA: Concanavalin A
GalN: D Galactosamine
HSCs: Hepatic Stellate Cells
KCs: Kupffer cells
LSECs: Liver Sinusoid Endothelial Cells
MDSCs: Myeloid Derived Suppressor Cells
MHC: Major Histocompatibility
ROS: Reactive Oxygen Species
SCID: Severe Combined Immunodeficiency Syndrome
TCR: T-cell Receptor
KEY POINTS
• Concanavalin A (ConA) induced T cell infiltration and liver injury is a model for acute hepatitis
• Important cell types include most white blood cells as well as stromal cells
• The ConA model has successfully been used to discover new pathways in hepatitis research
• New therapeutic interventions for hepatitis were discovered using the ConA model
INTRODUCTION
Autoimmune hepatitis
AIH animal models
ConA induced liver injury
Despite the above differences between the ConA model and human AIH, this model is considered as one of the best models for mimicking and studying certain mechanisms of AIH, and it has played an important role in AIH drug development [9]. However, decades of ConA-induced hepatitis research demonstrate that the model suffers from substantial variability resulting in inconsistency of reported results. The most important factors to take into account when using this model are the dose, genetic background, gender and age of the mice and microbial status of the animal facility. For more details we refer to a recently published standard operating procedure [18]. In this review, we present an overview of the current understanding of the cell types (Table 1) and molecules which are involved in ConA-induced liver injury. In addition, we will summarize successful therapeutic interventions and their effects on important factors and cell types in disease development.
| Cell type | Strategy for cell type depletion or adoptive transfer or adaptive transfer | Liver injury (ALT/AST induction in serum) |
Reference | Therapeutic intervention targeting specific cell type [P] prophylactic [T] therapeutic |
| T and B cells | SCID mice | Abolished | 17 | Dexamethasone [17,20] [P] Cyclosporin A [17] [P] FK506 [17] [P] Ginsenoside Rg1 [21] [P] Baicalein [22] [P] Tetrandrine [23] [P] CpG ODN [24] [P] Salvianolic acid A [25] [P] |
| Athymic nude mice | Abolished | 17 | ||
| RAG -/- mice | Abolished | 19 | ||
| Anti Thy 1.2 antibody | Abolished | 17 | ||
| CD4+ T cells | Anti CD4 antibody | Abolished | 17,26,27 | NCPP [28] [P] Dexmedetomidine [29] [P] Salidroside [30] [P] Ascaris Suum Extract [31] [T] Salvionalic Acid A [25] [P] Fraxinellone [32] [P] Pentoxyfilline [33] [P] Suppressive ODN [34] [P] [T] ApoA-II [35] [P] [T] |
| CD8 + T cells | Anti CD8 antibody | Not effected | 17,26,36 | |
| Kupffer cells | gadolinium chloride (GdCl3) | Abolished | 37-39 | TP-58 [41] [T] Edaravone [40] [P] Free radical scavengers [42] [P] Andrographolide [38] [P] N-acetylcysteine (NAC) [21] [P] Hesperidin [43] [P] Curcumin [44] [P] Edaravone [41] [P] |
| liposome-encapsulated dichloromethylene bisphosphonate (Cl2MBP) | Reduced | 40 | ||
| silica particles | Abolished | 17 | ||
| Neutrophils | Anti Gr1 antibody anti MIP-2antibody |
Reduced Reduced |
27,45,46 | |
| Eosinophils | Anti CCR3 antibody | Reduced | 47 | |
| NKT cells | CD1d -/- mice | Reduced | 48,49 | Glucocerebroside [52] [P] Prednisolone [53] [P] Retinoic Acid [54] [P] CGX [55] [P] CpG ODN [24] [P] DT56a [56] [T] |
| B2M -/- mice | Reduced | 50 | ||
| Vα14 -/- mice | Abolished | 19 | ||
| anti NK1.1 antibody | Reduced | 39 | ||
| CD39 -/- mice | Reduced | 51 | ||
| NK cells | Anti-asialo-GM1 (ASGM1) antibody | Not effected Reduced |
36,41,50,57 36, 58,59 |
CpG ODN [24] [P] |
| CD4+ CD25+ Tregs |
Anti CD25 antibody Adoptive transfer Tregs |
Exacerbated Reduced |
60, 61 60 |
DT56a [56] [T] |
| Tr1 cells | Adoptive transfer in vitro generated Tr1 cells | Reduced | 62 | |
| γδ T cells | TCRδ –/- mice | Exacerbated | 63,64 | |
| MDSCs | adoptive transfer MDSCs in naïve mice | Reduced | 20,65 | Dexamethasone [20] [P] Cannabidiol [66] [T] IL-25 [67] [P] + [T] GW4064 [68] [P] |
Table 1: Studies showing the importance of different cell types that are involved in ConA-mediated liver inflammation and therapeutic approaches used to manipulate them.
T CELLS
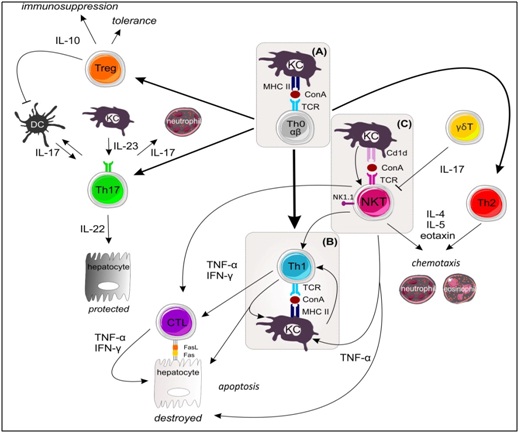
ConA activates T cells to proliferate, produce cytokines and migrate to the liver [46]. Several adhesion molecules play an important role in accumulation of CD4+ T cells but not CD8+ T cells [76]. By helping APCs and cytotoxic CD8+ T cells, CD4+ helper T cells are important in the initiation of an adaptive immune response. CD4+ T cells are divided into four major subsets based on their secreted cytokines and expression profile of transcription factors, namely, Th1, Th2, Th17 and regulatory T cells (Tregs) [77]. The involvement of and interplay between these T cell subsets in ConA-induced liver injury will be discussed in the following sections and is summarized in Figure 2.
Th1 cells
IL-12 is produced mainly by monocytes, macrophages and Dendritic Cells (DCs) [89] and plays an important role in the production of Th1 cytokines as well as Th2 cytokines by activation of Signal transducer and activator of transcription (STAT)4 [90,91]. IL-12 is detrimental in ConA-induced liver injury model as its administration exacerbates the signs of ConA-induced hepatitis. Additionally, its neutralization ameliorates them by reducing the expression of Th1 and Th2 cytokines [82,92]. In contrast, recent results using IL12 and STAT4 deficient mice reveal a protective role of IL12 induced activation of STAT-4 via indirect or direct down regulation of FasL in Natural Killer T (NKT) cells. These conflicting results indicate that IL-12 and STAT4 may play a dual role in ConA-induced hepatitis by promoting cytokine production and inhibiting NKT cell function [93].
Th17 cells
IL-22 is known to protect hepatocytes from the destructive effect of IFN-γ produced by different immune cells [105]. IL-22 has no direct effect on immune cells but affects liver tissue by activating STAT3 and Akt in hepatocytes, leading to the induction of anti-apoptotic and cell survival genes. The hepatoprotective role of IL-22 in ConA-induced injury was confirmed by treating IL-22 deficient mice [105]. It is important to note that Th17 cells are not the only source of IL-22, for example, Notch signaling in CD4+ T cells is crucial for IL-22 expression in a Th17 independent manner [107]. Interestingly, IL-22 seems to be pro-inflammatory in the presence of IL-17 but protective in its absence [108]. IL-23 does not initiate Th17 cell differentiation but acts as a survival cytokine maintaining Th17 responses. Th17 master regulators STAT3 and RORγt are essential for the expression of its receptor [109]. In ConA-induced hepatitis, the induction of IL-23 expression and Notch activation results in the production of IL-22 which is critical for protection. IL-23 induces the expression of IL-17 as well, which is detrimental in the pathogenesis, showing that IL-23 has both a hepatotoxic and a protective role [103].
As mentioned above, STAT3 is an important transcription factor involved in the induction of the Th17 specific cytokine profile. It has opposing roles in myeloid and T cells in ConA-induced hepatitis. In myeloid cells it inhibits STAT1 and NF-κB activation; thereby inhibiting hepatitis by downregulating the production of various cytokines such as IL-6, IL-12 and IFN-γ. In T cells, it leads to the expression of Th17 specific inflammatory cytokines, including IL-17, which might play a detrimental role in ConA-induced liver injury [102].
Regulatory T cells (Tregs)
The inhibitory function of Tregs is mediated by TGF-β, which is needed to suppress the injury caused by effector T cells [61]. Depleting CD25+ cells aggravated ConA-induced injury, and treating mice with CD4+ CD25+ Tregs made them less susceptible; this illustrates the therapeutic potential of Tregs [62]. For example, galectin-9 plays an important role in inducing apoptosis of TIM-3 positive activated CD4+ T cells and is predominantly expressed on Tregs [115-117]. In ConA-induced hepatitis, TIM3 neutralization, which blocks the effects of the endogenous ligand of galectin-9, aggravates liver injury by inhibiting the suppressive activities of Tregs [26,118]. Moreover, administration of galectin-9 ameliorates injury by downregulating the number of CD4+ effector T cells and their infiltration and cytokine production in the liver [26]. This example shows that the number of Tregs is important in overcoming ConA-induced hepatitis and interfering with Treg amount either exacerbates or improves injury.
ConA also induces Tr1 cells which secrete IL-10 and are Foxp3 negative [63]. IL-10 produced by Tregs and KCs plays a prominent role in the induction of tolerance. Interestingly, one single sublethal dose of ConA can induce tolerance within eight days. This tolerogenic state is characterized by an anti-inflammatory cytokine profile, with IL-10 as one of the major cytokines [62]. Restoring self-tolerance could be a valid therapeutic strategy because deficiency of Treg number or function plays a key role in ConA induced hepatitis. For a detailed review on using regulatory T cells to treat liver disease we refer to [119]. However, the role of Tregs in the pathogenesis of AIH and other autoimmune diseases is not clear. Recent reports show that Treg frequency and function are not impaired in AIH but the liver infiltrating Tregs are not able to block disease progression [120].
γδ T cells
NKT CELLS
NKT cells are a special subset of lymphocytes that express both the TCR with the invariant VαJα-chain and NK cell associated markers, including NK1. 1. These cells are considered to form a bridge between the innate and the adaptive immune systems. NKT cells are enriched in the liver and constitute about 30% of hepatic lymphocytes [127]. There are two distinct subpopulations of NKT cells: type I (95% of NKT cells) and type II (5% of NKT cells) [128]. Type I invariant iNKT cells have a highly restricted TCR repertoire, in contrast to the type II non-invariant NKT cells, which express a wide range of TCRs [129,130]. The crucial role of NKT cells in the pathogenesis of ConA-induced hepatitis has been shown by different studies using NKT deficient mice, depleting antibodies or NOD mice partially iNKT deficient [19,37,49,50,51,92,131]. Although NKT cells are one of the predominant effector cells in ConA-induced liver injury, they are rapidly depleted from the liver by Fas/FasL mediated apoptosis [49].
Activation of NKT cells is mediated by presentation of lipid antigens by CD1d but needs additional signals from KCs, LSECs, Hepatic Stellate Cells (HSCs), Myeloid Derived Suppressor Cells (MDSCs) and hepatocytes [132]. One of the most important signals is IL-12, produced mainly by APCs, which acts on iNKT via the IL-12R and triggers increased production of IL-4 [92,133]. In ConA-induced hepatitis, NKT cells are required for IL-4 induction which is essential for TNF-α production and liver damage [51]. Once they are activated, NKT cells release large amounts of Th1 cytokines (IFN-γ, TNF-α and IL-2) and Th2 cytokines (IL-4, IL-5, IL-10 and IL-13) [130]. NKT cells directly induce hepatocyte death through release of TNF-α or perforins/granzyme B and/or upregulation of FasL [134]. Next to this, upon TCR engagement iNKT cells can rapidly produce a mixture of cytokines involved in the differentiation of Th0 cell into Th1 or Th2 cells [135-137]. So by releasing of IFN-γ, NKT cells can indirectly induce hepatocyte death by driving a Th1 biased phenotype and converting CD8+ T lymphocytes into cytotoxic T lymphocytes (CTLs). (Figure2) It is clear that NKT cells play a well-established detrimental role in the course of ConA-induced injury. Hence, interfering with the development, recruitment or activation of NKT cells in the liver could be an effective therapeutic strategy.
HEPATOCYTES
Upon ConA challenge, IL-1β serum levels are increased, and this cytokine is a key mediator in the pathogenesis of the disease [84,85]. Hepatocytes are the major source of both circulating and hepatic IL-1Rα, which plays a critical role in the resolution of hepatic damage [138]. After ConA stimulation, TNF-α induces liver damage by activating both TNFR1 and TNFR2 [80,139]. However, TNF-α is not the only factor inducing apoptosis in the liver. Hepatocytes are very prone to Fas/FasL-mediated apoptosis because of their strong expression of Fas [140]. This type I integral membrane protein of the TNF receptor family transduces cell death signals and administration of a monoclonal agonistic Fas stimulating antibody leads to massive hepatocyte apoptosis and consequent liver damage [141-143]. However, the role of Fas/FasL in ConA-induced hepatitis remains controversial. Several studies have shown that blocking Fas/FasL signaling using antibodies, deficient mice or siRNA prevents ConA-induced liver injury [81,134,144,145]. In contrast, others researchers did not demonstrate a significant role for Fas/FasL but claimed that perforin-mediated cytotoxicity is important [69,85,134,146]. Interestingly, deficiency of caspase 1, which is a common intermediate of TNF-α and FasL signaling, also leads to protection against ConA-induced hepatitis [85,147].
As mentioned above, IL-22 targets epithelial cells such as hepatocytes and plays an essential antihepatotoxic role in ConA-induced injury [105,148-150]. The protective role of IL-22 is mediated through activation of STAT3 and subsequent induction of the anti-apoptotic proteins Bcl2 and Bcl-xL [148,149]. Next to this, strong IL-33 expression in hepatocytes is controlled by NKT cell activation [50]. IL-33, a new member of the IL-1 family, reduces the severity of liver damage caused by ConA stimulation by recruiting regulatory T cells to the liver. The expression of IL-33 is dependent on TNF-Related Apoptosis-Inducing Ligand (TRAIL) but not on TNF-α and FasL [151]. TRAIL induces apoptosis by binding to death receptors, but it also initiates necroptosis [152,153]. TRAIL induces necroptosis by activating PARP-1 in a RIPK1/3 dependent manner, which leads to depletion of ATP and subsequently to programmed necrosis [154]. Since TRAIL is a crucial mediator of ConA-induced liver injury, this type of damage is driven not only by apoptosis but also by necroptosis [155]. Furthermore, elevated RIPK1 and RIPK3 expression indicate that necroptosis occurs during pathogenesis [156].
MYELOID-DERIVED SUPPRESSOR CELLS
Myeloid Derived Suppressor Cells (MDSCs) expand during cancer, infection and inflammation, and have an outstanding ability to suppress T cell responses. This heterogeneous pool of myeloid cells has the morphology of granulocytes and monocytes [157]. Murine MDSCs are characterized by surface co-expression of Gr-1 and CD11b and are further subdivided into two major populations: CD11b+ Ly6G+ Ly6Clow granulocytic MDSCs and CD11b+ Ly6G- Ly6Chigh monocytic MDSCs [158]. MDSCs use various mechanisms to influence both innate and adaptive immune responses. Most importantly, they deprive T cells of L-cysteine and L-arginine, generate oxidative stress, and can activate or expand Tregs (for a review on MDSC functions we refer to 159,160). To facilitate the suppression of T cell responses, MDSCs need iNOS, Arginase 1 and Reactive Oxygen Species (ROS) [161]. Recent findings suggest that the accumulation of MDSCs is linked to different types of inflammatory diseases such as inflammatory bowel disease [162] but also hepatitis B and C infections [66,163-165]. Moreover, in ConA-induced liver injury an expansion of MDSCs in the liver was observed and these cells suppressed the proliferation of an early T cell subset population [20].
Different therapeutic interventions have been shown to be associated with the induction and/or expansion of MDSCs during ConA-induced liver inflammation [20,66,67]. For example, treatment with glucocorticoids, which are strong anti-inflammatory molecules working through the GC receptor GR, led to accumulation of MDSCs and protected against liver injury [20,166]. A specific liver MDSC population with an unique surface marker expression profile was recently identified. Moreover, Zhu et al., demonstrated that only this population could inhibit CD4+ T cell responses and this inhibitory capacity depends on de novo expression of iNOS and subsequent NO production upon CD4+ T cell contact [65]. In human AIH patients, evidence for the presence of MDSCs is limited [167]. However, accumulation of CD11b + cells in liver biopsies has been reported [168]. Data on MDSC function in mouse models of AIH show that exhaustion of MDSCs and impairment of MDSC effector functions are part of the pathogenic mechanisms of the disease. Restoring the MDSC suppressive capacity or increasing the accumulation of MDSCs could thus be a promising therapeutic strategy for AIH. However, specific methods to generate these cells ex vivo so that they will be suitable for administration to patients will be challenging [159]. Especially because MDSCs are not terminally differentiated cells so depending on their surrounding milieu, they may mature into other myeloid cell types.
NATURAL KILLER CELLS
The cytotoxic and cytokine response of Natural Killer (NK) cells depends on their interaction with other cells of the immune system, such as T cells and NKT cells, as well on the cytokine environment [169]. In ConA-induced hepatitis, large numbers of NK cells are recruited from the bone marrow and spleen to the liver. Activation of NK cells requires other types of immune cells, such as NKT cells and T cells, and depends strongly on IFN-γ production [58]. Since NK cells need other cells in order to perform their function, it could be expected that in vivo depletion of NK cells alone does not ameliorate ConA-induced liver injury [36,51,58,170].
KUPFFER CELLS
The liver resident macrophages or KCs constitute the first line of defense in the liver sinusoids that is in direct contact with all kinds of danger signals, such as microbial debris and gut derived pathogenic microbes. KCs phagocytose this exogenous material and degrade it, but they are also an important source of various inflammatory mediators. During disease, KCs interact with different neighboring cell types and the mediators they produce are central in influencing these cells. For a more detailed overview of the different cell-cell interactions and macrophage associated mediators in liver injury, we refer to Yang et al., [171].
Macrophages differentiate into M1 or M2 subsets, which are characterized by distinct markers and functions. The switch between pro-inflammatory M1 and anti-inflammatory M2 macrophages is controlled by mediators in the micro-environment of macrophages. A shift in the M1-M2 balance is correlated with the state of inflammation [171]. At the onset and during the first phase of acute hepatitis, macrophages exhibit a classical M1 phenotype and are hepatotoxic. In this phase, ConA activates KCs to produce large amounts of pro-inflammatory cytokines and promotes liver injury [80,172]. The essential role of KCs in disease development has been demonstrated by various research groups, who showed that KC depletion represses liver injury [38-40, 87]. Not surprisingly, depleting KCs does not affect the ability of ConA to induce the expression of important cytokines such as TNF-α and IFN-γ, suggesting that KCs are not the main producers of these factors. In contrast, the production and release of superoxide ROS is reduced after KC depletion; these species are critical in ConA-induced hepatitis [37,39]. In the second phase of hepatitis, macrophages display an M2-like phenotype, which is characterized by the production of anti-inflammatory and regenerative cytokines such as IL-10, TGF-β and IL-6 [171]. M2 macrophages counteract the overwhelming pro-inflammatory events during the resolution phase of ConA-induced hepatitis [173]. The most important mediators involved in suppressing inflammation and promoting tissue repair have been reviewed by Yang et al., and will not be discussed further [171]. In a third phase, six hours after ConA challenge, the total number of KCs decreases but the remaining KCs have a more potent pro-inflammatory phenotype characterized by high IL-1β secretion [174]. As mentioned before, IL-1β is known as a critical pro-inflammatory cytokine during liver injury [175].
NEUTROPHILS
Neutrophils are the first cell type to arrive at the site of inflammation in response to chemotactic factors. They migrate from the bloodstream to their target, where they attempt to resolve inflammation [78,176,177]. ConA challenge induces a recruitment of neutrophils to the liver within four hours. ConA can bind and activate neutrophils with consequent expression of adhesion molecules and release of reactive oxidants [179]. Accumulation of neutrophils is seen in the necrotic areas in the liver [47,48]. The activation and recruitment of neutrophils is a crucial step in the pathogenesis of ConA-induced liver inflammation [45,46]. Depleting neutrophils with an anti Gr1 antibody does not completely prevent but represses liver injury and leukocyte trafficking. Moreover, Bonder et al., showed that without neutrophils ConA cannot induce the recruitment of T cells and subsequent liver injury [46]. Activation of neutrophils leads to the expression of various mediators, such as IFN-γ, TNF-α and IL-4, which are known crucial factors in the development of ConA-induced liver injury, as discussed before [45].
EOSINOPHILS
Eosinophils are important in mediating tissue damage through the release of toxic granule proteins, pro-inflammatory cytokines, chemokines and lipids. The activation of NKT cells in ConA-induced hepatitis results in a strong IL-4/STAT-6 dependent production of eotaxin and IL-5, leading to eosinophil and neutrophil infiltration into the liver [95]. IL-5 is not only responsible for the maturation, proliferation and differentiation of eosinophils but also, together with eotaxin, for the recruitment of eosinophils to the liver. The importance of eosinophils in ConA-induced liver inflammation was shown using the CCR3 antibody to deplete eosinophils. Treatment with this antibody and neutralization of IL-5 led to protection against ConA-induced injury [48]. Next to IL-5, Smad3 is an essential mediator of TGF-β signaling and is important for the viability of eosinophils in vitro, supporting the idea that upon ConA stimulation, Smad3 promotes eosinophil function and survival [180,181].
DENDRITIC CELLS
In contrast to conventional DCs (CD11c+CD11b+ B220-) present in the spleen and other organs, hepatic DCs are thought to be plasmacytoid (CD11c+CD11b- B220+) [182]. Hepatic DCs are weak activators of immune responses and are believed to be more immunosuppressive and to have a somewhat immunotolerance phenotype [183,184]. During the early stages after ConA challenge (3-6h), the absolute number of DCs in the liver decreases, but during the later stages (12-24h) their number increases again. Using adoptive transfers, Tomiyama et al., showed that only hepatic DCs could suppress the hepatic damage induced by ConA and this might be related to their ability to suppress IL-12 secretion and the subsequent Th1 response [184].
STROMAL CELLS
Stromal cells lining the hepatic sinusoids are the first cells to encounter gut-derived or systemic antigens. There are two major types, both of which can modulate the immune response: Liver Sinusoidal Endothelial Cells (LSECs) and Hepatic Stellate Cells (HSCs).
LSECs are the most abundant non parenchymal cell population in the liver. They are unique endothelial cells that line the hepatic sinusoids and define the space of Disse. Although LSECs express low levels of MHC II and co-stimulatory molecules, they are scavengers that compete with DCs for the uptake of antigens circulating in the liver [185]. By physically interacting with DCs, LSECs can reduce the antigen presenting function of DCs [186]. After physical contact, DCs lose their ability to prime naive T cells due to reduced expression of co-stimulatory molecules. LSECS can also promote CD4+ T cell migration in a chemokine dependent way [187]. It was recently shown that the liver sinusoidal endothelium contains an intracellular transport system for chemokines to induce local recruitment of circulating T cells. During ConA-induced inflammation, LSECs internalize CXCL12 via CXCR4 mediated endocytosis, leading to enhanced CD4+ T cell migration [188]. LSECs can also reduce CD4+ T cell activity and promote the induction of Tregs using membrane bound TGF-β [114,189,190].
HSCs are present in the perisinusoidal space between LSECs and hepatocytes. In normal conditions, HSCs (Ito cells) are important for Vitamin A storage and for regulation of blood flow in the sinusoids. In inflammatory conditions, HSCs form a second line of defense by limiting the effector function of T cells that have extravasated and thereby they prevent tissue damage. HSCs produce large amounts of anti-inflammatory mediators like IL-10 and TGF-β [191]. By doing so, they can suppress DC function [192]. In addition, HSCs can induce NKT cell response by producing IL-15, present peptides to CD4 and CD8+ T cells, and induce T cell priming [193]. Next to LSECs, HSCs are the second APCs that reside in the liver. HSCs also facilitate the differentiation of inflammatory monocytes into MDSCs, which impair T cell function and proliferation [194]. Moreover, HSCs can induce and expand Tregs but the mechanism is not known [195,196]. For a full overview of hepatic immune regulation by stromal cells we refer to Schildberg et al., [197].
THERAPEUTIC INTERVENTIONS
Because of the great amount of cell types and mediators involved in the pathogenesis of the ConA induced liver inflammation; a lot of different therapeutic strategies have been tested in the last decades (depicted in the last column of Table 1). In most studies, strategies which inhibit T cell and macrophage activation leading to reduced cytokine production and liver damage are used. Known immunomodulatory agents like corticosteroids, cyclosporine A, paeoniflorin, glucocerebroside, curcumin and flavonoids -quercetin, hesperidin and baicalein- were able to reduce ConA associated cytokine production [17,22,43,53,119,198,200]. Unfortunately, most of the reported therapies are only effective when administered prophylactically (marked as [P] in table 1) and some even lose their potential when used therapeutically (marked as [T] in table 1). For example, pentoxyfilline, a strong suppressor of TNF-α release, is able to rescue the phenotype when administered prophylactically but the effect was lost when used therapeutically [33,201]. Luckily, there have been some studies identifying valid therapeutic strategies. For example, Y-40138 significantly suppressed the development of ConA-induced hepatitis by suppression of TNF-α, MIP-2, IL-4 and IFN-γ production [201]. Additionally, both quercetin and TP58, a novel thienopyridine derivative protect against liver injury by inhibiting NF-κB mediated inflammation [41,198]. Other strategies targeting regulatory T cells and/or NKT cell function have also been described. For example, oral administration of DT56a, a natural product isolated from soybeans, redistributed the CD4+ CD25+ T cells resulting in less liver damage [57]. New and interesting therapeutic opportunities arose when immunosuppressive MDSCs were shown to be induced upon ConA stimulation [20]. Later studies using dexamethasone show that its positive effect is specifically due to an increased MDSC induction [20]. Next to this, cannabidiol, a non-psychoactive cannabinoid and IL-25 treatment can reduce ConA induced inflammation and protect mice from liver injury by triggering the induction of MDSCs, opening a new window for therapeutic strategies [66,67].
CONCLUSION
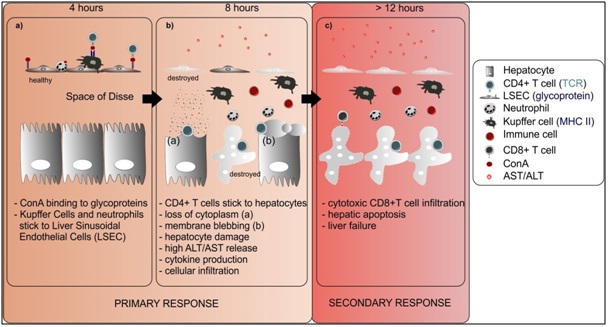
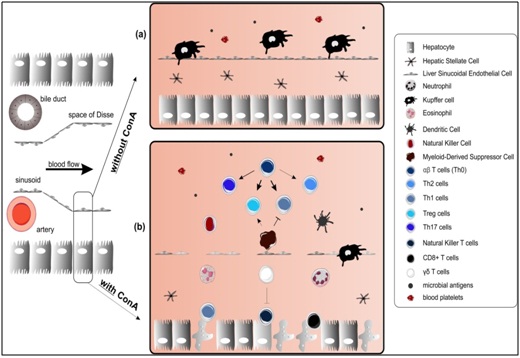
The ConA model mimics important features of acute liver injury. The immune activation/infiltration and the involvement of T cells in this model make it particularly relevant to study acute liver injury. In this review we presented an overview of the different cell types and their mediators driving ConA-induced hepatitis. Decades of research have shown that the pathogenesis is more complex than anticipated by Tiegs et al., in 1992 (shown in figure 1) [17]. The great variety of immune cells, hepatocytes and stromal cells that play crucial roles in the development of the disease and the fact that different cell types influence the effector functions of their neighboring cells by producing different cytokines, chemokines and other effector molecules (shown in Figure 2) are responsible for this high complexity. It is now clear that ConA-induced hepatitis is the result of a well-orchestrated interaction network (Figure 3) and novel mediators are still being identified meaning that the full complexity of pathogenesis is not yet understood. Fortunately, successful therapeutic strategies targeting a specific mediator or cell type have been already identified. However, recent findings, like the induction of MDSCs, have great therapeutic potential but have not been investigated to a great extent and should be focused on in the future.
(a) In normal conditions, oxygenated blood passes through the sinusoids containing blood platelets and microbial antigens. The area between the liver sinusoid endothelial cells (LSECs) and hepatocytes is called the space of Disse and contains stellate cells which control the blood flow. Immune cells reside in the sinusoids but are in close contact with the LSECs.
(b) After intravenous ConA injection, different immune cell types are recruited to the liver and infiltrate the parenchym causing inflammation and cell death resulting in liver failure.
REFERENCES
- Czaja AJ (2010) Animal models of autoimmune hepatitis. Expert Rev Gastroenterol Hepatol 4: 429-443.
- Webb GJ, Hirschfield GM (2016) Using GWAS to identify genetic predisposition in hepatic autoimmunity. J Autoimmun 66: 25-39.
- de Boer YS, van Gerven NM, Zwiers A, Verwer BJ, van Hoek B et al. (2015) Genome-wide association study identifies variants associated with autoimmune hepatitis type 1. Gastroenterology 147: 443-452.
- Czaja AJ (2002) Autoimmune hepatitis after liver transplantation and other lessons of self-intolerance. Liver Transpl 8: 505-513.
- Liberal R, Grant CR, Mieli-Vergani G, Vergani D (2013) Autoimmune hepatitis: a comprehensive review. J Autoimmun 41: 126-139.
- Yüksel M, Laukens D, Heindryckx F, Van Vlierberghe H, Geerts A, et al. (2014) Hepatitis mouse models: from acute-to-chronic autoimmune hepatitis. Int J Exp Pathol 95: 309-320.
- Czaja AJ (2009) Current and future treatments of autoimmune hepatitis. Expert Rev Gastroenterol Hepatol 3: 269-291.
- Wang HX, Liu M, Weng SY, Li JJ, Xie C, et al. (2012) Immune mechanisms of Concanavalin A model of autoimmune hepatitis. World J Gastroenterol 18: 119-125.
- Bonito AJ, Aloman C, Fiel MI, Danzl NM, Cha S, et al. (2013) Medullary thymic epithelial cell depletion leads to autoimmune hepatitis. J Clin Invest 123: 3510-3524.
- Hintermann E, Ehser J, Christen UC (2012) The CYP2D6 animal model: how to induce autoimmune hepatitis in mice. J Vis Exp 3: 3644.
- Bogdanos DP, Dalekos GN (2008) Enzymes as target antigens of liver-specific autoimmunity: the case of cytochromes P450s. Curr Med Chem 15: 2285-2292.
- Christen U, Hintermann E (2015) An Update on Animal Models of Autoimmune Hepatitis: Are we There Yet? Curr Pharm Des 21: 2391-2400.
- Jaeckel E, Hardtke-Wolenski M, Fischer K (2011) The benefit of animal models for autoimmune hepatitis. Best Pract Res Clin Gastroenterol 25: 643-651.
- Soares PA, Nascimento CO, Porto TS, Correia MT, Porto AL, et al. (2011) Purification of a lectin from Canavalia ensiformis using PEG-citrate aqueous two-phase system. J Chromatogr B Analyt Technol Biomed Life Sci 879: 457-460.
- Lis H, Sharon N (1986) Biological properties of lectins. In: Liener JE, Sharon N, Goldstein IJ (eds.). The lectins: properties, functions and applications in biology and medicine. Acadamic Press, Inc, London. Pg no: 256-291.
- Tiegs G, Hentschel J, Wendel A (1992) A T cell-dependent experimental liver injury in mice inducible by concanavalin A. J Clin Invest 90: 196-203.
- Heymann F, Hamesch K, Weiskirchen R, Tacke F (2015) The concanavalin A model of acute hepatitis in mice. Lab Anim 49: 12-20.
- Leist M, Wendel A (1996) A novel mechanism of murine hepatocyte death inducible by concanavalin A. J Hepatol 25: 948-959.
- Kaneko Y, Harada M, Kawano T, Yamashita M, Shibata Y et al. (2000) Augmentation of Valpha14 NKT cell-mediated cytotoxicity by interleukin 4 in an autocrine mechanism resulting in the development of concanavalin A-induced hepatitis. J Exp Med 191: 105-114.
- Diao W, Jin F, Wang B, Zhang CY, Chen J, et al. (2014) The protective role of myeloid-derived suppressor cells in concanavalin A-induced hepatic injury. Protein Cell 5: 714-724.
- Cao L, Zou Y, Zhu J, Fan X, Li J (2013) Ginsenoside Rg1 attenuates concanavalin A-induced hepatitis in mice through inhibition of cytokine secretion and lymphocyte infiltration. Mol Cell Biochem 380: 203-210.
- Zhang Y, Shan L, Hua Y, Wang D, Zeng H, et al. (2013) Baicalein selectively induces apoptosis in activated lymphocytes and ameliorates concanavalin a-induced hepatitis in mice. PLoS One 8: 69592.
- Feng D, Mei Y, Wang Y, Zhang B, Wang C, et al. (2008) Tetrandrine protects mice from concanavalin A-induced hepatitis through inhibiting NF-kappaB activation. Immunol Lett 121: 127-133.
- Zhang H, Gong Q, Li JH, Kong XL, Tian L, et al. (2010) CpG ODN pretreatment attenuates concanavalin A-induced hepatitis in mice. Int Immunopharmacol 10: 79-85.
- Xu X, Hu Y, Zhai X, Lin M, Chen Z, et al. (2013) Salvianolic acid A preconditioning confers protection against concanavalin A-induced liver injury through SIRT1-mediated repression of p66shc in mice. Toxicol Appl Pharmacol 273: 68-76.
- Lv K, Zhang Y, Zhang M, Zhong M, Suo Q (2012) Galectin-9 ameliorates Con A-induced hepatitis by inducing CD4(+)CD25(low/int) effector T-Cell apoptosis and increasing regulatory T cell number. PLoS One 7: 48379.
- Zhang S, Liang R, Luo W, Liu C, Wu X, et al. (2013) High susceptibility to liver injury in IL-27 p28 conditional knockout mice involves intrinsic interferon-γ dysregulation of CD4+ T cells. Hepatology 57: 1620-1631.
- Liu C, Gao S, Qu Z, Wang Q, Zhu F, et al. (2012) NCPP treatment alleviates ConA-induced hepatitis via reducing CD4+T activation and NO production. Immunopharmacol Immunotoxicol 34: 962-967.
- Wang H, Hu B, Zou Y, Bo L, Wang J, et al. (2014) Dexmedetomidine premedication attenuates concanavalin A-induced hepatitis in mice. J Toxicol Sci 39: 755-764.
- Hu B, Zou Y, Liu S, Wang J, Zhu J, et al. (2014) Salidroside attenuates concanavalin A-induced hepatitis via modulating cytokines secretion and lymphocyte migration in mice. Mediators Inflamm 2014: 314081.
- Nascimento WC, Silva RP, Fernandes ES, Silva MC, Holanda GC, et al. (2014) Immunomodulation of liver injury by Ascaris suum extract in an experimental model of autoimmune hepatitis. Parasitol Res 113: 3309-3317.
- Sun Y, Qin Y, Gong FY, Wu XF, Hua ZC, et al. (2009) Selective triggering of apoptosis of concanavalin A-activated T cells by fraxinellone for the treatment of T-cell-dependent hepatitis in mice. Biochem Pharmacol 77: 1717-1724.
- Shirin H, Bruck R, Aeed H, Frenkel D, Kenet G, et al. (1998) Pentoxifylline prevents concanavalin A-induced hepatitis by reducing tumor necrosis factor alpha levels and inhibiting adhesion of T lymphocytes to extracellular matrix. J Hepatol 29: 60-67.
- Li N, Liu Y, Li S, Fu C, Zhou R et al. (2015) Protective role of synthetic oligodeoxynucleotides expressing immunosuppressive TTAGGG motifs in concanavalin A-induced hepatitis. Immunol Lett 151: 54-60.
- Yamashita J, Iwamura C, Sasaki T, Mitsumori K, Ohshima K, et al. (2011) Apolipoprotein A-II suppressed concanavalin A-induced hepatitis via the inhibition of CD4 T cell function. J Immunol 186: 3410-3420.
- Li B, Sun R, Wei H, Gao B, Tian Z (2006) Interleukin-15 prevents concanavalin A-induced liver injury in mice via NKT cell-dependent mechanism. Hepatology 43: 1211-1219.
- Nakashima H, Kinoshita M, Nakashima M, Habu Y, Shono S, et al. (2008) Superoxide produced by Kupffer cells is an essential effector in concanavalin A-induced hepatitis in mice. Hepatology 48: 1979-1988.
- Okamoto T, Maeda O, Tsuzuike N, Hara K (1998) Effect of gadolinium chloride treatment on concanavalin A-induced cytokine mRNA expression in mouse liver. Jpn J Pharmacol 78: 101-103.
- Shi G, Zhang Z, Zhang R, Zhang X, Lu Y, et al. (2012) Protective effect of andrographolide against concanavalin A-induced liver injury. Naunyn Schmiedebergs Arch Pharmacol 385: 69-79.
- Hatano M, Sasaki S, Ohata S, Shiratsuchi Y, Yamazaki T, et al. (2008) Effects of Kupffer cell-depletion on Concanavalin A-induced hepatitis. Cell Immunol 251: 25-30.
- Li Y, Wang ZL, He F, Wu Y, Huang W, et al. (2012) TP-58, a novel thienopyridine derivative, protects mice from concanavalinA-induced hepatitis by suppressing inflammation. Cell Physiol Biochem 29: 31-40.
- Shirin H, Aeed H, Alin A, Matas Z, Kirchner M, et al. (2010) Inhibition of immune-mediated concanavalin a-induced liver damage by free-radical scavengers. Dig Dis Sci 55: 268-275.
- Li G, Chen MJ, Wang C, Nie H, Huang WJ, et al. (2014) Protective effects of hesperidin on concanavalin A-induced hepatic injury in mice. Int Immunopharmacol 21: 406-411.
- Tu CT, Han B, Yao QY, Zhang YA, Liu HC et al. (2012) Curcumin attenuates Concanavalin A-induced liver injury in mice by inhibition of Toll-like receptor (TLR) 2, TLR4 and TLR9 expression. Int Immunopharmacol 12: 151-157.
- Hatada S, Ohta T, Shiratsuchi Y, Hatano M, Kobayashi Y (2005) A novel accessory role of neutrophils in concanavalin A-induced hepatitis. Cell Immunol 233: 23-29.
- Bonder CS, Ajuebor MN, Zbytnuik LD, Kubes P, Swain MG (2004) Essential role for neutrophil recruitment to the liver in concanavalin A-induced hepatitis. J Immunol 172: 45-53.
- Nakamura K, Okada M, Yoneda M, Takamoto S, Nakade Y, et al. (2001) Macrophage inflammatory protein-2 induced by TNF-alpha plays a pivotal role in concanavalin A-induced liver injury in mice. J Hepatol 35: 217-224.
- Louis H, Le Moine A, Flamand V, Nagy N, Quertinmont E, et al. (2002) Critical role of interleukin 5 and eosinophils in concanavalin A-induced hepatitis in mice. Gastroenterology 122: 2001-2010.
- Takeda K, Hayakawa Y, Kaer L, Matsuda H, Yagita H et al. (2000) Critical contribution of liver natural killer T cells to a murine model of hepatitis. Proc Natl Acad Sci USA 97: 5498-5503.
- Arshad MI, Rauch M, L'Helgoualc'h A, Julia Vr, Leite-de-Moraes MC et al. (2011) NKT cells are required to induce high IL-33 expression in hepatocytes during ConA-induced acute hepatitis. Eur J Immunol 41: 2341-2348.
- Toyabe S, Seki S, Iiai T, Takeda K, Shirai K, et al. (1997) Requirement of IL-4 and liver NK1+ T cells for concanavalin A-induced hepatic injury in mice. J Immunol 159: 1537-1542.
- Beldi G, Wu Y, Banz Y, Nowak M, Miller L, et al. (2008) Natural killer T cell dysfunction in CD39-null mice protects against concanavalin A-induced hepatitis. Hepatology 48: 841-852.
- Margalit M, Abu Gazala S, Ghazala SA, Alper R, Elinav E et al. (2005) Glucocerebroside treatment ameliorates ConA hepatitis by inhibition of NKT lymphocytes. Am J Physiol Gastrointest Liver Physiol 289: 17-25.
- Kwon HJ, Won YS, Park O, Feng D, Gao B (2014) Opposing effects of prednisolone treatment on T/NKT cell- and hepatotoxin-mediated hepatitis in mice. Hepatology 59: 1094-1106.
- Lee KA, Song YC, Kim GY, Choi G, Lee YS, et al. (2012) Retinoic acid alleviates Con A-induced hepatitis and differentially regulates effector production in NKT cells. Eur J Immunol 42: 1685-1694.
- Shin JW, Wang JH, Kim HG, Park HJ, Bok HS, et al. (2010) CGX, a traditional Korean medicine ameliorates concanavalin A-induced acute liver injury. Food Chem Toxicol 48: 3308-3315.
- Shabat Y, Lichtenstein Y, Zolotarov L, Ben Ya'acov A, Ilan Y (2013) Hepatoprotective effect of DT56a is associated with changes in natural killer T cells and regulatory T cells. J Dig Dis 14: 84-92.
- Miyagi T, Takehara T, Tatsumi T, Suzuki T, Jinushi M et al. (2004) Concanavalin a injection activates intrahepatic innate immune cells to provoke an antitumor effect in murine liver. Hepatology 40: 1190-1196.
- Zhao N, Hao J, Ni Y, Luo W, Liang R, et al. (2011) Vγ4 γ? T cell-derived IL-17A negatively regulates NKT cell function in Con A-induced fulminant hepatitis. J Immunol 187: 5007-5014.
- Beraza N, Malato Y, Sander LE, Al-Masaoudi M, Freimuth J, et al. (2009) Hepatocyte-specific NEMO deletion promotes NK/NKT cell- and TRAIL-dependent liver damage. J Exp Med 206: 1727-1737.
- Wei HX, Chuang YH, Li B, Wei H, Sun R, et al. (2008) CD4+ CD25+ Foxp3+ regulatory T cells protect against T cell-mediated fulminant hepatitis in a TGF-beta-dependent manner in mice. J Immunol 181: 7221-7229.
- Erhardt A, Biburger M, Papadopoulos T, Tiegs G (2007) IL-10, regulatory T cells, and Kupffer cells mediate tolerance in concanavalin A-induced liver injury in mice. Hepatology 45: 475-485.
- Ye F, Yan S, Xu L, Jiang Z, Liu N, et al. (2009) Tr1 regulatory T cells induced by ConA pretreatment prevent mice from ConA-induced hepatitis. Immunol Lett 122: 198-207.
- Zhao N, Ni Y, Zhao L, Wu Z, Yin Z (2014) [Protective role of γδ T cells in concanavalin A-induced liver injury]. Zhonghua Gan Zang Bing Za Zhi 22: 58-62.
- Li H, Dai F, Peng Q, Gan H, Zheng J, et al. (2015) Myeloid-derived suppressor cells suppress CD4+ and CD8+ T cell responses in autoimmune hepatitis. Mol Med Rep 12: 3667-3673.
- Hegde VL, Nagarkatti PS, Nagarkatti M (2011) Role of myeloid-derived suppressor cells in amelioration of experimental autoimmune hepatitis following activation of TRPV1 receptors by cannabidiol. PLoS One 6: 18281.
- Sarra M, Cupi ML, Bernardini R, Ronchetti G, Monteleone I, et al. (2013) IL-25 prevents and cures fulminant hepatitis in mice through a myeloid-derived suppressor cell-dependent mechanism. Hepatology 58: 1436-1450.
- Zhang H, Liu Y, Bian Z, Huang S, Han X, et al. (2014) The critical role of myeloid-derived suppressor cells and FXR activation in immune-mediated liver injury. J Autoimmun 53: 55-66.
- Gossard AA, Lindor KD (2012) Autoimmune hepatitis: a review. J Gastroenterol 47: 498-503.
- Gantner F, Leist M, Lohse AW, Germann PG, Tiegs G (1995) Concanavalin A-induced T-cell-mediated hepatic injury in mice: the role of tumor necrosis factor. Hepatology 21: 190-198.
- Nagata T, McKinley L, Peschon JJ, Alcorn JF, Aujla SJ et al. (2008) Requirement of IL-17RA in Con A induced hepatitis and negative regulation of IL-17 production in mouse T cells. J Immunol 181: 7473-7479.
- Mizuhara H, O'Neill E, Seki N, Ogawa T, Kusunoki C, et al. (1994) T cell activation-associated hepatic injury: mediation by tumor necrosis factors and protection by interleukin 6. J Exp Med 179: 1529-1537.
- Tiegs G (2007) Cellular and cytokine-mediated mechanisms of inflammation and its modulation in immune-mediated liver injury. Z Gastroenterol 45: 63-70.
- Bowen DG, Warren A, Davis T, Hoffmann MW, McCaughan GW, et al. (2002) Cytokine-dependent bystander hepatitis due to intrahepatic murine CD8 T-cell activation by bone marrow-derived cells. Gastroenterology 123: 1252-1264.
- Azuma T, Watanabe N, Yagisawa H, Hirata H, Iwamura M, et al. (2000) Induction of apoptosis of activated murine splenic T cells by cycloprodigiosin hydrochloride, a novel immunosuppressant. Immunopharmacology 46: 29-37.
- Azuma T, Watanabe N, Yagisawa H, Hirata H, Iwamura M, et al. (2000) Induction of apoptosis of activated murine splenic T cells by cycloprodigiosin hydrochloride, a novel immunosuppressant. Immunopharmacology 46: 29-37.
- Hammerich L, Heymann F, Tacke F (2011) Role of IL-17 and Th17 cells in liver diseases. Clin Dev Immunol 2011: 345803.
- O'Garra A, Arai N (2000) The molecular basis of T helper 1 and T helper 2 cell differentiation. Trends Cell Biol 10: 542-550.
- Murphy KM, Reiner SL (2002) The lineage decisions of helper T cells. Nat Rev Immunol 2: 933-944.
- Küsters S, Gantner F, Künstle G, Tiegs G (1996) Interferon gamma plays a critical role in T cell-dependent liver injury in mice initiated by concanavalin A. Gastroenterology 111: 462-471.
- Tagawa Y, Sekikawa K, Iwakura Y (1997) Suppression of concanavalin A-induced hepatitis in IFN-gamma(-/-) mice, but not in TNF-alpha(-/-) mice: role for IFN-gamma in activating apoptosis of hepatocytes. J Immunol 159: 1418-1428.
- Nicoletti F, Di Marco R, Zaccone P, Salvaggio A, Magro G et al. (2000) Murine concanavalin A-induced hepatitis is prevented by interleukin 12 (IL-12) antibody and exacerbated by exogenous IL-12 through an interferon-gamma-dependent mechanism. Hepatology 32: 728-733.
- Glimcher LH, Murphy KM (2000) Lineage commitment in the immune system: the T helper lymphocyte grows up. Genes Dev 14: 1693-1711.
- Mizuhara H, O'Neill E, Seki N, Ogawa T, Kusunoki C, et al. (1994) T cell activation-associated hepatic injury: mediation by tumor necrosis factors and protection by interleukin 6. J Exp Med 179: 1529-1537.
- Ksontini R, Colagiovanni DB, Josephs MD, Edwards CK 3rd, Tannahill CL, et al. (1998) Disparate roles for TNF-alpha and Fas ligand in concanavalin A-induced hepatitis. J Immunol 160: 4082-4089.
- Maeda S, Chang L, Li ZW, Luo JL, Leffert H, et al. (2003) IKKbeta is required for prevention of apoptosis mediated by cell-bound but not by circulating TNFalpha. Immunity 19: 725-737.
- Fotin-Mleczek M, Henkler F, Samel D, Reichwein M, Hausser A, et al. (2002) Apoptotic crosstalk of TNF receptors: TNF-R2-induces depletion of TRAF2 and IAP proteins and accelerates TNF-R1-dependent activation of caspase-8. J Cell Sci 115: 2757-2770.
- Schümann J, Prockl J, Kiemer AK, Vollmar AM, Bang R, et al. (2003) Silibinin protects mice from T cell-dependent liver injury. J Hepatol 39: 333-340.
- Gee K, Guzzo C, Che Mat NF, Ma W, Kumar A (2009) The IL-12 family of cytokines in infection, inflammation and autoimmune disorders. Inflamm Allergy Drug Targets 8: 40-52.
- Cho SS, Bacon CM, Sudarshan C, Rees RC, Finbloom D, et al. (1996) Activation of STAT4 by IL-12 and IFN-alpha: evidence for the involvement of ligand-induced tyrosine and serine phosphorylation. J Immunol 157: 4781-4789.
- Morinobu A, Gadina M, Strober W, Visconti R, Fornace A, et al. (2002) STAT4 serine phosphorylation is critical for IL-12-induced IFN-gamma production but not for cell proliferation. Proc Natl Acad Sci USA 99: 12281-12286.
- Zhu R, Diem S, Araujo LM, Aumeunier A, Denizeau J, et al. (2007) The Pro-Th1 cytokine IL-12 enhances IL-4 production by invariant NKT cells: relevance for T cell-mediated hepatitis. J Immunol 178: 5435-5442.
- Wang Y, Feng D, Wang H, Xu MJ, Park O, et al. (2014) STAT4 knockout mice are more susceptible to concanavalin A-induced T-cell hepatitis. Am J Pathol 184: 1785-1794.
- O'Garra A (2000) Commitment factors for T helper cells. Curr Biol 10: 492-494.
- Jaruga B, Hong F, Sun R, Radaeva S, Gao B (2003) Crucial role of IL-4/STAT6 in T cell-mediated hepatitis: up-regulating eotaxins and IL-5 and recruiting leukocytes. J Immunol 171: 3233-3244.
- Veldhoen M, Hocking RJ, Atkins CJ, Locksley RM, Stockinger B (2006) TGFbeta in the context of an inflammatory cytokine milieu supports de novo differentiation of IL-17-producing T cells. Immunity 24: 179-189.
- Wilson NJ, Boniface K, Chan JR, McKenzie BS, Blumenschein WM, et al. (2007) Development, cytokine profile and function of human interleukin 17-producing helper T cells. Nat Immunol 8: 950-957.
- Ivanov II, McKenzie BS, Zhou L, Tadokoro CE, Lepelley A et al. (2006) The orphan nuclear receptor RORgammat directs the differentiation program of proinflammatory IL-17+ T helper cells. Cell 126: 1121-1133.
- Lafdil F, Miller AM, Ki SH, Gao B (2010) Th17 cells and their associated cytokines in liver diseases. Cell Mol Immunol 7: 250-254.
- Yan S, Wang L, Liu N, Wang Y, Chu Y (2012) Critical role of interleukin-17/interleukin-17 receptor axis in mediating Con A-induced hepatitis. Immunol Cell Biol 90: 421-428.
- Yasumi Y, Takikawa Y, Endo R, Suzuki K (2007) Interleukin-17 as a new marker of severity of acute hepatic injury. Hepatol Res 37: 248-254.
- Lafdil F, Wang H, Park O, Zhang W, Moritoki Y, et al. (2009) Myeloid STAT3 inhibits T cell-mediated hepatitis by regulating T helper 1 cytokine and interleukin-17 production. Gastroenterology 137: 2125-2135.
Citation: Ballegeer M, Libert C (2016) Different Cell Types Involved in Mediating Concanavalin A Induced Liver Injury: A Comprehensive Overview. J Gastroenterol Hepatology Res 1: 001.
Copyright: © 2016 Marlies Ballegeer, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.