
Effectiveness and Tolerability of the Early Chemo & Late Acceleration Protocol for Stage III and IV Head and Neck Cancer
*Corresponding Author(s):
Lyndell E KellyDepartment Of Oncology, Dunedin Public Hospital, Dunedin, New Zealand
Tel:+64 3 474 0999,
Fax:+64 34709689
Email:Lyndell.kelly@southerndhb.govt.nz
Abstract
Background
There are multiple published ways of combining chemotherapy and radiotherapy. There is no doubt that the optimal regimen for safety and efficacy is yet to be found. Studies showing late effects are rare. From first radiobiological principles, the early chemo-late acceleration regimen was devised in the mid 1990’s to obtain optimal cure rates and minimal long-term normal tissue damage.
Methods and Findings
An audit was conducted of 10 years’ experience with this regimen involving 94 patients with a median follow-up of 48 months. The regimen uses Cisplatin 20mg/m2 followed by 2Gy on days 1 - 5 in weeks 1 and 3, no treatment in weeks 2 and 4, then twice daily radiotherapy in weeks 5, 6 and 7.
Local control was 86.8% for the whole group and 92.2% for oropharyngeal cancers. Disease-specific survival was 69.6% and 81.6% respectively. Although the Sydney Swallow Questionnaire raised the spectre of dysphagia in 59% of the group, there was only one case of late aspiration pneumonia, indicating reasonable late toxicity.
Conclusions
This regimen is safe and effective. As techniques evolve, xerostomia and dysphagia should be reduced.
Keywords
ABBREVIATIONS
SCC: Squamous Cell Carcinoma
SLD: Sub-Lethal Damage Repair
PET: Positron Emission Tomography
XRT: X-ray therapy, Radiotherapy
Bd: Twice-Daily
GTV: Gross Tumour Volume
CT: Chemotherapy
HPV: Human Papilloma Virus
PEG: Percutaneous Endoscopic Gastrostomy
AF: Accelerated Fractionation
CRT: Chemo-Radiotherapy, Usually Synchronous
VMAT: Volumetric modified arc therapy
INTRODUCTION
Head and Neck mucosal cancer remains a challenge with multiple regimens in use. The attempt to combine the proven advantages of synchronous chemotherapy with radiotherapy and acceleration has led to multiple regimens in use. Comparisons of regimens has only recently begun. Long-term results raise concerns about quality of survival and other regimens warrant investigation [1].
The Early Chemo Late Acceleration (ECLA) protocol was devised at the Queensland Radium Institute in the mid-1990’s to optimize these radiobiological and clinical principles:
• Re-oxygenation: An early reduction in volume increases oxygenation and therefore radiosensitivity of SCC [2]. By the start of week 5 the cancer is a fraction of its previous size
• Re-population: It is important to keep the overall duration of treatment to around 43 days [3]. It is logical to intensify treatment around the time when repopulation is getting underway, that is, 5-6 weeks
• Accelerated fractionation has been shown in level 1evidence to improve control by about 8% [4]. Although acute effects may be worse, duration of side-effects is shorter [5]
• Sub-lethal Damage (SLD) repair of DNA: By treating macroscopic cancer twice daily we capitalize on slower and less proficient DNA SLD repair by cancer cells while giving normal cells the required 6 hour gap
• Synchronous chemotherapy has been shown in several meta-analyses to improve control by 8% [6]
• Morbidity of fractionated cisplatin is less than either weekly (40mg/m2) or three-weekly (100mg/m2) [7]
In addition, it is sensible to separate side-effects of chemotherapy and radiotherapy.
This is a retrospective study of patients randomly allocated to a particular radiation oncologist (LK) from 2006 to 2015. The aim of the study was to quantify the clinical impression of this regimen and compare it to other published results. No ethics approval was required for this study.
MATERIALS AND METHOD
Patients coming through the New Zealand Southern District Health Board Head and Neck clinic were seen and assessed for synchronous chemoradiotherapy if their disease was of poor prognosis or more advanced than stage 3. If of reasonable general health (Karnovsky>70) they were offered curative treatment. If renal function was poor, they were given either carboplatin or no chemotherapy with radiotherapy. Those patients are not part of this audit.
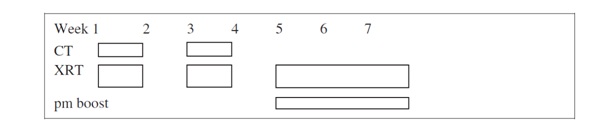
Week 1 - Monday - Friday Cisplatin 20mg/m2 daily Monday to Friday and XRT 10 Gy in 5 fractions to large volumes
Week 2 - no treatment
Week 3 - same as week 1
Week 4 - no treatment
Weeks 5, 6, 7 - Twice daily radiation. Gap of at least 6 hrs, then extra dose to bulky cancer areas. Daily 2Gy to large volume and bd 1.3Gy boost to GTV.
Intensity-Modulated Radiation Therapy (IMRT) was introduced in 2008 with the hope of reducing long-term xerostomia. Two plans were calculated with the afternoon boost as a separate plan, either conformal 3-d or IMRT and the summation checked for hot spots. With the understanding that HPV-induced SCCs are more chemo and radio sensitive, since 2013, the boost dose for p16-positive tumours has been reduced to give a total of 65.6Gy. Routine PET scans have become available only since 2015.
During treatment and recovery, patients are seen in a combined clinic of dietitian, nurse, radiation oncologist, speech language therapist and radiation therapist. Weight is recorded twice weekly. Prophylactic PEGs are not used unless the patient is unable to swallow at diagnosis and have not been necessary. If weight loss is greater than 20% body weight, a naso-gastric tube is used usually in week 7, for duration of up to 4 weeks until recovery. Hospitalization may be required at the completion of treatment for nursing care and feeding stabilization.
All residents of New Zealand have a National Health Identifying (NHI) number, simplifying follow up. Head and neck patients are followed for 5 years. A South Island-wide clinical information system makes review of distant follow-up easy online. No patient was lost to follow-up.
An audit of ECLA patients was conducted and local control, disease-specific survival and overall survival examined. Late toxicity was assessed by survey mailed to long-term survivors with the Sydney Swallow Questionnaire (SSQ) and the Pai Xerostomia score [8,9]. The SSQ is a validated series of 17 questions with a 10cm visual analog scale where the patient places their decision about the difficulty or ease of the topic. The higher the score, the further to the right of each question’s scale the respondent marked, the worst score being 1700. The Pai xerostomia questionnaire has similar 10cm visual analog scales for 8 questions specifically about mouth dryness, the top and worst score being 800.
Hospital records of each patient were examined to assess admissions for chest infections or respiratory specialist care which would indicate long-term pharyngeal constrictor dysfunction.
RESULTS
From 2006 to 2015 94 patients were treated with the ECLA regimen and had a median follow-up of 48 months and at least 24 months. Sites were oropharynx 58, oral cavity 10, hypopharynx 7, larynx 17 and nasal vestibule 2.Eight had local recurrence and 11 died of pulmonary and 1 of hepatic metastases. Six patients had ECLA after neck dissection. Table 1 shows the T and N stage of all patients.
|
|
T1 |
T2 |
T3 |
T4 |
Total |
|
N0 |
|
8 |
13 |
4 |
25 |
|
N1 |
|
7 |
11 |
1 |
19 |
|
N2a |
2 |
3 |
2 |
1 |
8 |
|
N2b |
6 |
9 |
9 |
4 |
28 |
|
N2c |
2 |
2 |
1 |
2 |
7 |
|
N3a |
1 |
1 |
1 |
3 |
6 |
|
N3c |
|
|
1 |
|
1 |
|
|
11 |
30 |
38 |
15 |
94 |
Table 1: Tumour characteristics.
Local control at 5 years for all sites is 86.8%. Follow-up generally stopped at 60 months, and there are 35 patients still alive and free of recurrence beyond 5 years. T-stage and N-stage affected the local control rate to a surprisingly small amount (Figure 1).
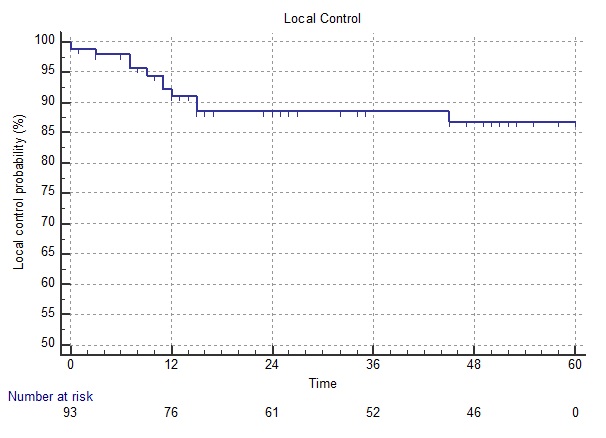 Figure 1a: Local control for whole group treated with ECLA.
Figure 1a: Local control for whole group treated with ECLA.
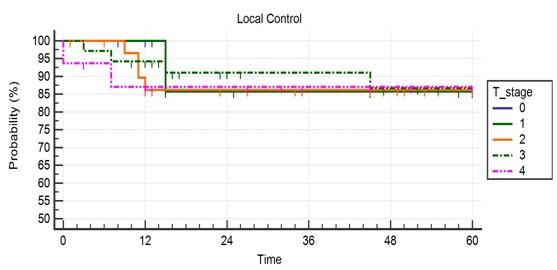 Figure 1b: Local control according to T stage.
Figure 1b: Local control according to T stage.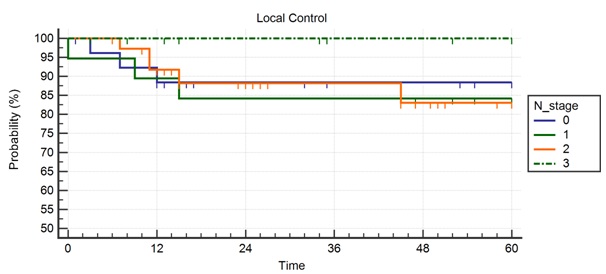 Figure 1c: Local control according to N-stage.
Figure 1c: Local control according to N-stage.
Figures 1a,1b, and 1c. Kaplan-Meier curves showing overall local control and effect of T and N stage on local control in patients treated with the ECLA regimen.
Disease-specific survival at 5 years is 69.6% (Figure 2). The size of nodes affects DSS as N3 patients are more likely to die of systemic disease than are N1 patients.
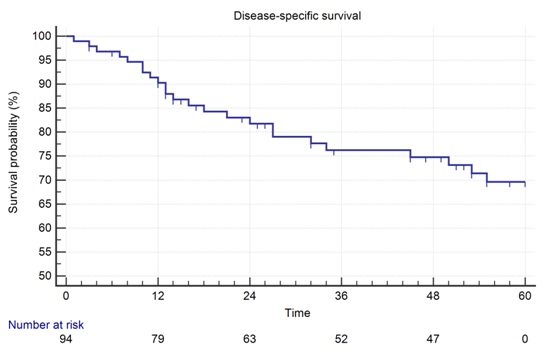 Figure 2a: Disease specific survival.
Figure 2a: Disease specific survival.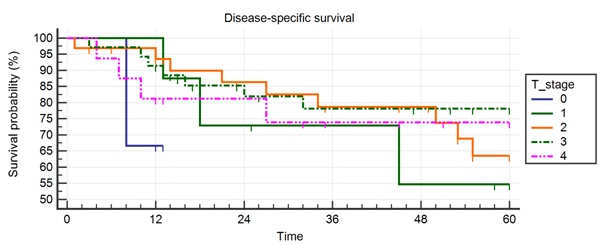 Figure 2b: Disease-specific survival according to T-stage.
Figure 2b: Disease-specific survival according to T-stage.
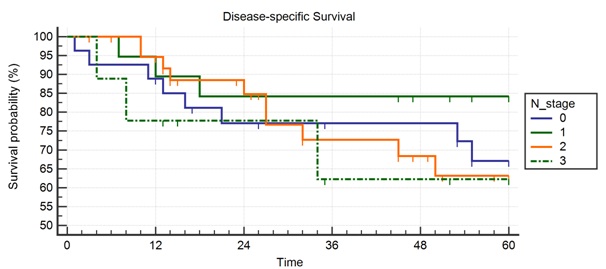 Figure 2c: Disease-specific survival according to N-stage.
Figure 2c: Disease-specific survival according to N-stage.
Figure 2a,b,c. Disease-specific survival is 69.6% at 5 years with a mean of 48.9 months. The effect of T stage is very minor but the hazard ratio for N3 relative to N1 is 3.05 (0.59-15.59).
Overall survival also includes incidental causes of death such as myocardial infarction (n=2) and other cancers (n=3) and is 63.1% at 5 years.
Oropharyngeal SCC had local control of 92.2% and disease-specific survival of 81.6% at 5 years.
There were 3 treatment related deaths. During treatment two men died of aspiration pneumonia, one with supra-glottic SCC and one with oropharyngeal SCC and asbestosis complicating a chest infection. One man died of aspiration pneumonia 50 months after completing treatment of supraglottic SCC, with rheumatoid disease contributing.
Mean weight loss during the course and after was 8kg.
SSQ and Pai xerostomia questionnaires were sent out to 45 people and responses received from 37. From the SSQ, there was no discernible pattern to swallowing difficulty with time, site or stage. Twenty-two of 37 respondents (59%) had scores greater than 234, indicating dysphagia. Eleven of the 37 (30%) had scores more than 500 and only one patient scored more than 850. Clinic letters contain very little information about dysphagia.
Likewise xerostomia was highly individually determined and no trend could be detected. Ten patients (27%) had scores greater than 400. Correlation between the SSQ and Pai xerostomia scores was high at 0.7540 (95% confidence interval 0.569 - 0.8664) and p<0.0001.
There were no admissions for chest infections in hospital records of all patients. One patient required a PEG for 2 years before recovering.
DISCUSSION
The results of this audit are very good both regarding local control and long-term effects on normal tissues. Level 1 evidence shows compared to conventional radiotherapy both Accelerated Fractionation (AF) and synchronous Chemotherapy-Radiotherapy (CRT) improve local control and survival [4,6]. There are multiple different schedules combining AF and CRT [10]. This schedule has distinct theoretical advantages as outlined in the introduction and confirmed in this audit.
The comparison of regimens is very difficult as results depend on multiple factors such as stage of presentation, prevalence of continued tobacco use, HPV status, availability of support services, etc, as well as the particular regimen used. There is a range of local control reports in the literature ranging from 80% at 3 years [11]- to local failure at 3 years of 39.4% for HPV+ cancers or 45.8% for HPV- cancers or 19.9% at 3yrs [12,13]. The DAHANCA regimen of 6 fractions per week with weekly Cisplatin and the hypoxic cell sensitizer nimorazole achieved 80% local control and 67% disease-specific survival at 5 years [14]. The ECLA regimen achieved 86.8% and 69.6% respectively at 5 years.
Since Withers, Taylor and Macejewski, et al., described accelerated repopulation after 4 weeks, there has been concern about gaps in radiotherapy [15]. These have usually been towards the end of treatment when the patient needed to recover from mucosal side-effects. This is a different scenario from an early gap during which shrinkage and re-oxygenation can occur. Several early combinations had gaps and better results than other contemporaneous regimens [16-19]. RTOG97-03 had as one arm a 13 week protocol treating alternate weeks with daily fluoro-uracil and hydroxyurea with equivalent results to conventional schedules [20]. Neo-adjuvant chemotherapy regimens constitute an early gap in anti-cancer treatment have shown results equivalent to synchronous regimens [21]. A dogmatic condemnation of early gaps is unwarranted.
Concern about duration of treatment led to very fast treatments with associated toxicity negating gains of chemo [22]. If total duration of treatment is less than 46 days, there doesn’t seem to be a negative effect [3]. The ECLA regimen takes 47 days and results suggest the effect of early shrinkage and re-oxygenation overcomes any negative effect of duration. The short duration of accelerated radiotherapy (50Gy in weeks 5-7) may be advantageous.
Oropharyngeal SCC is more likely to be HPV-induced and therefore have better prognosis. Unfortunately not all oropharyngeal SCCs in this series were tested for p16, a surrogate genetic marker for HPV. Results however, show for stages III and IV a 92.2% LC and DSS of 81.6%, equal to those of the MD Anderson [23] where at 5 years oropharyngeal SCC had LC 90% and DSS 82%. Even if all in our study were HPV+, results are better than reported for such a group [12].
Although results are very good, the question of morbidity arises.
Acute morbidity is moderate. Rades, et al., show that fractionated cisplatin (20mg/m2 x5) is the least morbid way of giving cisplatin and is possibly more effective than other regimens [7]. Other studies report using this in weeks 1 and 5 but Murthy, et al., used it alternate weeks up to 7 times [16,24,25]. Patients having ECLA treatment develop minor nausea on day 4 or 5 of each chemo week. This clears by the middle of the following rest week. Patients are able to eat normally until late week 6 or 7. Analgesia with low dose slow-release morphine and topical local anaesthetic is usually adequate. About half require naso-gastric feeding to supplement oral intake for 2 to 4 weeks. Koyfman and Adelstein describe a need for enteral feeding in about half and median naso-gastric duration of 8 weeks [26]. Only one patient required a PEG for ongoing dysphagia after ECLA treatment. Two patients died during treatment, both of chest infections with compromised lungs.
Long-term morbidity is best assessed by the incidence of chest infections from damaged constrictor muscles and xerostomia.
On the SSQ, 22 patients (59%) had scores greater than 234 out of 1700, indicating dysphagia. This poor result is a surprise as clinic letters contain very little information about problems. It is suspected that the scores are personality and mood dependant and this is a factor worth further study. Similar findings of the results of radiotherapy alone were made by Szsczesniak, et al., who also found 59% had scores more than 234, not correlated with site, stage or time from treatment and most reported no problem at follow-up visits [27]. It is to be expected that a chemo-radiotherapy regimen would have worse results but that is not the case with the ECLA regimen.
We found no patient treated with the ECLA regimen was seen by respiratory specialists or was admitted for chest infections after ECLA treatment. All deaths had a defined cause and there was only one death from aspiration pneumonia at 50 months. Szczesniak, et al., found aspiration pneumonia was responsible for 7 of 37 (19%) non-cancer-related deaths [27]. Despite the detail of the SSQ, it may be that we simply did not detect aspiration or stricture formation as the reported incidence of such late toxicities is very high [28]. No long-term PEG dependence occurred.
Those with high dysphagia scores also tend to have high xerostomia scores (Figure 3). This correlation of dysphagia and xerostomia is well-known [29]. Reducing the dose on the superior pharyngeal constrictor muscle is important in reducing long-term dysphagia but is very difficult with a T2, 3 or 4 carcinoma without protecting the primary cancer and local lymphatics. VMAT allows greater normal structure protection but it is a delicate balancing act to protect constrictors without raising the risk of a marginal miss.
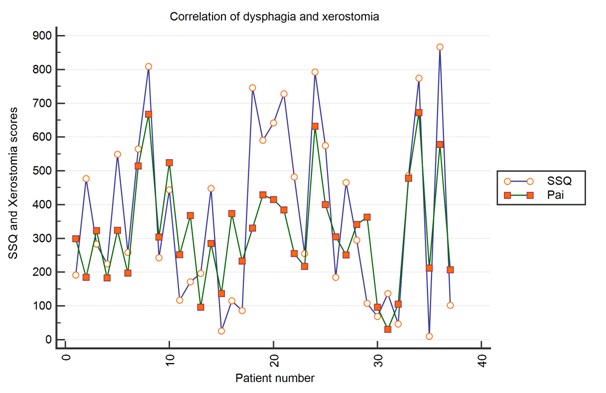 Figure 3: Responses to the Sydney Swallow Questionnaire and to the Pai Xerostomia questionnaire correlated with a correlation coefficient of 0.7540 (95% confidence interval 0.569 to 0.8664) and p<0.0001.
Figure 3: Responses to the Sydney Swallow Questionnaire and to the Pai Xerostomia questionnaire correlated with a correlation coefficient of 0.7540 (95% confidence interval 0.569 to 0.8664) and p<0.0001.
SUMMARY
The early-chemo late acceleration protocol is at least as effective as other published protocols and is shown to have reasonable long-term morbidity. It is resource-intensive requiring ten chemotherapy and up to 40 radiotherapy sessions. But the long-term advantage of optimal cure rates and safety from aspiration make this worthwhile. While further investigation is warranted of the reasons for variability of ongoing dysphagia, it is a concern that the attempt to protect pharyngeal constrictors will lead to more local recurrences.
CONFLICT OF INTEREST STATEMENT
No author had a conflict of interest in the preparation of this study. No external source of funding was required for this study.
REFERENCES
- Machtay M, Moughan J, Trotti A, Garden AS, Weber RS, et al. (2008) Factors associated with severe late toxicity after concurrent chemoradiation for locally advanced head and neck cancer: An RTOG Analysis. J Clin Oncol 26: 3582-3589.
- Nordsmark M and Overgaard J (2000) A confirmatory prognostic study on oxygenation status and loco-regional control in advanced head and neck squamous cell carcinoma treated by radiation therapy. Radiother Oncol 57: 39-43.
- Platek ME, McCloskey SA, Cruz M, Burke MS, Reid ME, et al., (2013) Quantification of the effect of treatment duration on local-regional failure after definitive concurrent chemotherapy and intensity-modulated radiation therapy for squamous cell carcinoma of the head and neck. Head Neck 35: 684-688.
- Bourhis J, Overgaard J, Audry H, Ang KK, Saunders M, et al. (2006) Hyperfractionated or accelerated radiotherapy in head and neck cancer: a meta-analysis. Lancet 368: 843-854.
- Poulsen M, Denham J, Spry N, Lamb D, Peters L, et al. (1999) Acute toxicity and cost analysis of a phase III randomized trial of accelerated and conventional radiotherapy for squamous carcinoma of the head and neck: A Trans-Tasman Radiation Oncology Group study. Australas Radiol 43: 487-494.
- Blanchard P, Baujat B, Holostenco V, Bourredjem A, Baey C, et al. (2011) Meta-analysis of chemotherapy in head and neck cancer (MACH-NC): A comprehensive analysis by tumour site. Radiother Oncol 100: 33-40.
- Rades D, Kronemann S, Meyners T, Bohlen G, Tribius S, et al (2011) Comparison Of Four Cisplatin-Based Radiochemotherapy Regimens for Nonmetastatic Stage iii/iv Squamous Cell Carcinoma of The Head And Neck. Int J Radiat Oncol Biol Phys 80: 1037-1044.
- Dwivedi RC, St Rose S, Roe JW, Khan AS, Pepper C, et al. (2010) Validation of the Sydney Swallow Questionnaire (SSQ) in a cohort of head and neck cancer patients. Oral Oncol 46: 10-14.
- Pai S, Ghezzi E and Ship J (2001) Development of a Visual Analogue Scale questionnaire for subjective assessment of salivary dysfunction. Oral Surg Oral Med Oral Pathol Oral RadiolEndod 91: 311-316.
- Prior D, Solomon B, and Porceddu S (2011) The emerging era of personalized therapy in squamous cell carcinoma of the head and neck. Asia Pac J Clin Oncol 7: 236-251.
- Kader HA, Mydin AR, Wilson M, Alexander C, Shahi J, et al. (2011) Treatment Outcomes Of Locally advanced Oropharyngeal Cancer: A Comparison Between Combined Modality Radio-Chemotherapy And Two Variants Of Single Modality Altered Fractionation Radiotherapy. Int J Radiat Oncol Biol Phys 80: 1030-1036.
- Ang KK, Harris J, Wheeler R, Weber R, Rosenthal DI, et al. (2010) Human papillomavirus and survival of patients with oropharyngeal cancer. N Engl J Med 363: 24-35.
- Ang KK, Zhang Q, Rosenthal DI, Nguyen-Tan PF, Sherman EJ, et al. (2014) Randomized Phase III Trial of Concurrent Accelerated Radiation Plus Cisplatin With or Without Cetuximab for Stage III to IV Head and Neck Carcinoma: RTOG 0522. J Clin Onc 32: 2940-2950.
- Bentzen J, Toustrup K, Eriksen JG, Primdahl H, Andersen LJ, et al. (2015) Locally advanced head and neck cancer treated with accelerated radiotherapy, the hypoxic modifier nimorazole and weekly cisplatin. Results from the DAHANCA 18 phase II study. Acta Oncologica 54: 1001-1007.
- Withers R, Taylor J and Maciejewski B (1988) The hazard of accelerated tumor clonogen repopulation during radiotherapy. Acta Oncologica 27: 131-46.
- Murthy AK, Taylor SG 4th, Showel J, Caldarelli DD, Hutchinson JC Jr, et al. (1987) Treatment of advanced head and neck cancer with concomitant radiation and chemotherapy. Int J Radiat Oncol Biol Phys 13: 1807-1813.
- Olmi P, Crispino S, Fallai C, Torri V, Rossi F, et al. (2003) Locoregionally advanced carcinoma of the oropharynx: conventional radiotherapy vs accelerated hyperfractionated radiotherapy vs. concomitant radiotherapy and chemotherapy -- a multicentre randomized trial. Int J Radiat Oncol Biol Phys 55: 78-92.
- Wendt TG, Hartenstein RC, Wustrow TP, Lissner J (1989) Cisplatin, fluorouracil with leukovorin calcium enhancement, and synchronous accelerated radiotherapy in the management of locally advanced head and neck cancer: A Phase II study. Journal of Clin Oncol 7: 471-476.
- Merlano M, Benasso M, Corvò R, Rosso R, Vitale V, et al. (1996) Five-year update of a randomized trial of alternating radiotherapy and chemotherapy compared with radiotherapy alone in treatment of unresectable squamous cell carcinoma of the head and neck. J Natl Cancer Inst 88: 583-589.
- Garden AS, Harris J, Vokes EE, Forastiere AA, Ridge JA, et al. (2007) Results of Radiation Therapy Oncology Group 97-03-A Randomized Phase II Trial of Concurrent Radiation and Chemotherapy for Advanced Squamous Cell Carcinomas of the Head and Neck: Long-term Results and Late Toxicities. Int J Radiat Oncol 69: 140.
- Vermorken JB, Remenár E, Herpen CV, Gorlia T, Mesia R, et al. (2007) Cisplatin, Fluorouracil, and Docetaxel in Unresectable Head and Neck Cancer. N Engl J Med 357: 1695-1704.
- Bourhis J, Lapeyre M, Tortochaux J, Lusinchi A, Etessami A, et al. (2011) Accelerated radiotherapy and concomitant high dose chemotherapy in nonresectable stage IV locally advanced HNSCC: Results of a GORTEC randomized trial. Radiother Oncol 100: 56-61.
- Garden AS, Dong L, Morrison WH, Stugis EM, Glisson BS, et al. (2013) Patterns of disease recurrence following treatement of oropharyngeal cancer with intensity modulated radiation therapy. Int J Radiat Oncol Biol Phys 85: 941-947.
- Lau H, Brar S, Hao D, MacKinnon J, Yee D, Gluck S, et al. (2006) Concomitant low-dose cisplatin and three-dimensional conformal radiotherapy for locally advanced squamous cell carcinoma of the head and neck: analysis of survival and toxicity. Head Neck 28: 189-96.
- Huguenin P, Beer KT, Allal A, Rufibach K, Friedli C, et al. (2004) Concomitant Cisplatin Significantly Improves Locoregional Control in Advanced Head and Neck Cancers Treated with Hyperfractionated Radiotherapy. J Clin Oncol 22: 4665-4673.
- Koyfman SA, Adelstein D (2012) Enteral feeding tubes in patients undergoing definitive chemoradiation therapy for head-and-neck cancer: A critical review. Int J Radiat Oncol Biol Phys 84: 581-589.
- Szczesniak MM, Maclean J, Zhang T, Graham PH, Cook IJ (2014) Persistent Dysphagia after Head and Neck Radiotherapy: A Common and Under-reported Complication with Significant Effect on Non-cancer-related Mortality. Clin Oncol (R Coll Radiol) 26: 697-703.
- Rancati T, Schwarz M, Allen AM, Feng F, Popovtzer A, et al. (2010) Radiation dose-volume effects in the larynx and pharynx. Int J Radiat Oncol Biol Phys 76: 64-69.
- Mortensen H, Jensen K, Aksglaede K, Behrens M and Grau C (2013) Late Dysphagia after IMRT for head and neck cancer and correlation with dose-volume parameters. Radiother Oncol 107: 288-294.
Citation: Kelly LE, Hollows K, Pepping B, Mannion A, Tuaine J, et al. (2019) Effectiveness and Tolerability of the Early Chemo & Late Acceleration Protocol for Stage III and IV Head and Neck Cancer. J Clin Stud Med Case Rep 6: 066.
Copyright: © 2019 Lyndell E Kelly, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

