
Journal of Angiology & Vascular Surgery Category: Medical
Type: Case Report
Endovascular Treatment of Common Carotid Artery Aneurysm - Case Report and Review of Current Literature
*Corresponding Author(s):
Bistra BonevaDepartment Of Vascular Surgery, National Heart Hospital, Sofia, Bulgaria
Tel:+359 889304112,
Email:bistra.pb@gmail.com
Received Date: Aug 07, 2019
Accepted Date: Aug 29, 2019
Published Date: Sep 05, 2019
Abstract
Objective
Extracranial Carotid Artery Aneurysms (ECAAs) are rare but potentially life-threatening pathology. Left untreated ECAAs may cause stroke, Transient Ischemic Attacks (TIA) or cranial nerve dysfunction. Open surgical repair is a first line of treatment but still related to high mortality and morbidity rates. In the modern endovascular era, an endovascular approach is a viable option in the management of ECAAs. However, no evidence-based guidelines have been established.
Case report
A 64-year-old male presented with a painful, pulsatile mass in the neck. The patient’s concomitant diseases include arterial hypertension and diabetes. Due to the clinical, US and CT findings and patient's refusal for conventional surgery an endovascular treatment was chosen (ECAA of the right Common Carotid Artery (CCA) 68.9/62.1 mm, massive thrombosis and blood flow in the sac, significant communication of 7.6 mm between the CCA and the aneurysm sac). A stent-graft covering CCA to its origin was deployed. Later follow-up showed complete exclusion of the aneurysm sac with no endoleak.
Conclusion
The main goal of this treatment is to prevent thromboembolic complications, compression and rupture of the aneurysm. It is achieved by excluding the aneurysm and restoring the blood flow. The treatment of choice should be based on the size, type and location of the aneurysm, the underlying cause for its development and the general condition of the patient.
The endovascular approach could be the preferred therapeutic option in select patients. However, a larger series and longer follow-up are necessary to evaluate the safety and effectiveness of these procedures.
Extracranial Carotid Artery Aneurysms (ECAAs) are rare but potentially life-threatening pathology. Left untreated ECAAs may cause stroke, Transient Ischemic Attacks (TIA) or cranial nerve dysfunction. Open surgical repair is a first line of treatment but still related to high mortality and morbidity rates. In the modern endovascular era, an endovascular approach is a viable option in the management of ECAAs. However, no evidence-based guidelines have been established.
Case report
A 64-year-old male presented with a painful, pulsatile mass in the neck. The patient’s concomitant diseases include arterial hypertension and diabetes. Due to the clinical, US and CT findings and patient's refusal for conventional surgery an endovascular treatment was chosen (ECAA of the right Common Carotid Artery (CCA) 68.9/62.1 mm, massive thrombosis and blood flow in the sac, significant communication of 7.6 mm between the CCA and the aneurysm sac). A stent-graft covering CCA to its origin was deployed. Later follow-up showed complete exclusion of the aneurysm sac with no endoleak.
Conclusion
The main goal of this treatment is to prevent thromboembolic complications, compression and rupture of the aneurysm. It is achieved by excluding the aneurysm and restoring the blood flow. The treatment of choice should be based on the size, type and location of the aneurysm, the underlying cause for its development and the general condition of the patient.
The endovascular approach could be the preferred therapeutic option in select patients. However, a larger series and longer follow-up are necessary to evaluate the safety and effectiveness of these procedures.
Keywords
Endovascular treatment; Extracranial carotid artery aneurysm; Stentgraft
INTRODUCTION
Extracranial Carotid Artery Aneurysms (ECAAs) is rare (less than 1% of all carotid diseases) but potentially life-threatening pathology [1,2]. They could be a result of diverse causes - atherosclerotic degeneration, traumatic injury, dissection, local infection, fibromuscular dysplasia, connective tissue disease, or complication after Carotid Endarterectomy (CEA) [2]. According to a meta-analysis, the majority of ECAAs are pseudoaneurysms and true ECAAs are less than 10% [2,3]. Although the natural course of ECAAs is still unknown, they may cause a stroke, Transient Ischemic Attacks (TIA) or cranial nerve dysfunction if left untreated with a mortality rate of 60% to 70% [2,4]. Conservative management has been reported to lower the risk of ischemic events but it is not effective at resolving lesions, so the risk for embolism remains high [2,5]. Although the open surgical repair is considered as first line of treatment, it is still related to high mortality and morbidity rates (mortality and major stroke rate of 9% in one of the largest series) [3]. In the modern endovascular era, there are an increasing number of literature reports, showing that an endovascular approach is a viable option in the management of ECAAs [1-6]. However, no evidence-based guidelines have been established mainly because of the small number of cases in each center [6].
We present a case of large, atherosclerotic saccular ECCA treated with endovascular approach in the light of recent literature data. This report is compliant with the SCARE Guidelines.
We present a case of large, atherosclerotic saccular ECCA treated with endovascular approach in the light of recent literature data. This report is compliant with the SCARE Guidelines.
CASE REPORT
A 64-year-old male patient was admitted in the vascular surgery department with a history of a painful, pulsatile mass in the right supraclavicular area for several years. Clinical examination showed tumour approximately 5 cm in size. The overlying skin was normalwith no signs of inflammation. All laboratory tests were in referent ranges as these included also biomarkers for infection. Patient’s concomitant diseases included arterial hypertension, medicated with Bisoprolol fumarate 5mg, and diabetes treated with combined drug therapy (insulin and Metformin hydrochloride). The latter manifested with peripheral vascular complications - diabetic retinopathy and lower extremity peripheral artery disease. The right toe had been amputated a few years ago. Nine years ago, due to urinary bladder carcinoma, a cystectomy and urethrostomy on the abdominal wall were performed.
Neurological examination found no signs of cranial nerve compression or injury. The Ultrasound (US) examination revealed a saccular aneurysm of the right common carotid artery with a maximum size of 61/55 mm, with massive thrombosis and pulsatile blood flow within the sac. Subsequent contrast Computer Tomography (CT) confirmed an aneurysmal formation of the right common carotid artery above the clavicle -maximal diameter 68.9 mm and length 62.1 mm with massive eccentric thrombosis and blood flow in the sac (Figure 1A). A hemodynamically significant communication of 7.6 mm between the Common Carotid Artery (CCA) and the aneurysm sac was found. The communication was 49.4 mm away from the origin of the right Internal Carotid Artery (ICA) which was dislocated medially and compressed.
Due to the clinical, US and CT findings and most importantly - refusal for open surgery, endovascular treatment was performed. 10 FR long 90 cm sheath was introduced from the right common femoral artery. Initial angiography showed a large aneurysm with extensive communication between the sac and right CCA (Figure 1B). A stent-graft (Fluency Plus 9/60, Bard) covering CCA to its origin was deployed. Final angiography showed complete closure of the defect without any sign of endoleak (Figure 1C). Protection devices were not necessary due to the saccular morphology of the aneurysm (no interference with the thrombus in the sac during cannulation and deployment) and also relatively free of disease carotid bifurcation.
We offered the patient decompression surgery (excision of aneurysm) which he absolutely refused. He was discharged on the second postoperative day without any complications especially neurological ones. The follow-up ultrasound demonstrated a patent reconstruction without blood flow in the aneurysmal sac and no signs of endoleak. Dual Antiplatelet Therapy (DAPT) was prescribed (ASA 100 mg and Clopidogrel 75mg), combined with proton pump inhibitor 20mg twice a day and statin (Rosuvastatin 20mg) therapy.
Follow-up with ultrasound on the 1st and 6th month after the procedure and a CT scan on 12th month were performed. They showed complete exclusion of the aneurysm sac with no endoleak or size enlargement (Figure 1D).
Neurological examination found no signs of cranial nerve compression or injury. The Ultrasound (US) examination revealed a saccular aneurysm of the right common carotid artery with a maximum size of 61/55 mm, with massive thrombosis and pulsatile blood flow within the sac. Subsequent contrast Computer Tomography (CT) confirmed an aneurysmal formation of the right common carotid artery above the clavicle -maximal diameter 68.9 mm and length 62.1 mm with massive eccentric thrombosis and blood flow in the sac (Figure 1A). A hemodynamically significant communication of 7.6 mm between the Common Carotid Artery (CCA) and the aneurysm sac was found. The communication was 49.4 mm away from the origin of the right Internal Carotid Artery (ICA) which was dislocated medially and compressed.
Due to the clinical, US and CT findings and most importantly - refusal for open surgery, endovascular treatment was performed. 10 FR long 90 cm sheath was introduced from the right common femoral artery. Initial angiography showed a large aneurysm with extensive communication between the sac and right CCA (Figure 1B). A stent-graft (Fluency Plus 9/60, Bard) covering CCA to its origin was deployed. Final angiography showed complete closure of the defect without any sign of endoleak (Figure 1C). Protection devices were not necessary due to the saccular morphology of the aneurysm (no interference with the thrombus in the sac during cannulation and deployment) and also relatively free of disease carotid bifurcation.
We offered the patient decompression surgery (excision of aneurysm) which he absolutely refused. He was discharged on the second postoperative day without any complications especially neurological ones. The follow-up ultrasound demonstrated a patent reconstruction without blood flow in the aneurysmal sac and no signs of endoleak. Dual Antiplatelet Therapy (DAPT) was prescribed (ASA 100 mg and Clopidogrel 75mg), combined with proton pump inhibitor 20mg twice a day and statin (Rosuvastatin 20mg) therapy.
Follow-up with ultrasound on the 1st and 6th month after the procedure and a CT scan on 12th month were performed. They showed complete exclusion of the aneurysm sac with no endoleak or size enlargement (Figure 1D).
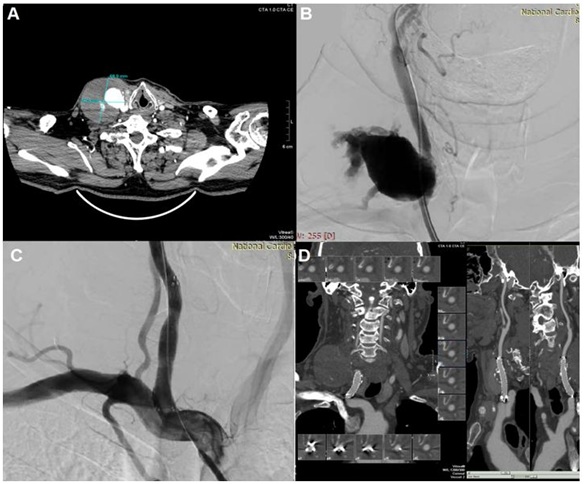 Figure 1(A-D): (A) Preoperative CT scan (B) Intraoperative angiography (C) Intraoperative result after stentgraft deployment (D) First year follow up-CT scan showing no endoleak.
Figure 1(A-D): (A) Preoperative CT scan (B) Intraoperative angiography (C) Intraoperative result after stentgraft deployment (D) First year follow up-CT scan showing no endoleak.DISCUSSION
Due to the extremely low prevalence of ECAAs, there is a lack of adequate experience and guidelines in terms of choices of treatment and treatment optimization [6]. Invasive treatment of ECAAs pertains to only 0.6-3.8% of all extracranial carotid interventions, 0.6-2% of all carotid endarterectomies and 0.4-2% of all extracranial arterial aneurysm repairs [5]. The golden standard for management remains open repair but this is related to more frequent postoperative complications [2-6]. The review by Welleweerd et al., which included 39 articles covering a total of 1,239 patients, concluded that surgical treatment could reduce the risk of stroke and death in patients with ECAAs [5]. However, a cranial nerve injury, which is the most common complications, occurs in 2.2-44% [3]. Further, surgical repair is technically difficult when the aneurysms are located near the skull base, requiring a very extensive and invasive exposure of the lesions. The open-access surgery could be tricky in patients with hostile neck due to previous neck surgery and/or radiation [2,3]. All these factors may partially explain the increasing popularity of endovascular stenting for ECAAs, after its successful application in many other areas. Li et al., published a systematic review of 113 studies involving 224 endovascular patients treated for ECCA and found a 92.8% procedure success with 93.2% stent-graft patency during the follow-up period. The overall in-hospital mortality, stroke, and cranial nerve injury were 4.1%, 1.8%, and 0.5%, respectively [3].
The main goal of the treatment is to prevent the thromboembolic complications of the aneurysm. It is achieved by excluding the aneurysm and restoring the blood flow. The treatment of choice should be based on size, type and location of the aneurysm, the underlying cause for its development, as well as the general condition of the patient [2]. Regarding the aneurysmal type, some authors found that long-term outcomes after the endovascular procedures were better in the treatment of pseudoaneurysms than true aneurysms [2,7]. This could be a result from the fact that most of the pseudoaneurysms are believed to be self-limited and not likely to reoccur after endovascular therapy, whereas the causes of true aneurysms, such as atherosclerosis, are ongoing and could lead to further aneurysmal degeneration [2]. In addition, true aneurysms often have a significant amount of thrombus inside the sac and frequently cause cerebral embolic events during the endovascular procedure [2,8]. Ni L et al., demonstrated good results in both types of aneurysms, as the procedure was successful in our case with saccular ECCA [2]. However, the limited follow-up and the number of patients do not allow a firm conclusion to be drawn about that.
According to the Attigah classification about aneurysm location, high type I and low type V aneurysms might be particularly suitable for an endovascular approach [9]. The reasons are related to the fact that type II endoleak from the patent ECA can be avoided when the aneurysms are far from the bifurcation of the CCA as it is in our case. Another reason is that these types of aneurysms allow stent-grafts to be deployed because the diameters of the proximal and distal landing zones are similar [2]. The only reasonable contraindication for endovascular treatment ishostile aortic arch (and its branches) anatomy mainly because of the large size of the delivery system of stentgrafts as well as its stiffness.
Endovascular techniques include the implantation of stents with or without embolization of the aneurysmal sac, stents coated with autogenous venous grafts, endovascular embolization or balloon occlusion, and placement of stent-grafts [5]. The meta-analysis of Li et al., showed a slight decrease in postoperative endoleak, a significantly lower rate of reintervention, overall late complications and stent-graft stenosis and a significant increase in thrombosis of the aneurysmal sac can be found in the patients treated with covered stents, which may support the covered stent as a better choice for ECAA [3]. However, the greater profile of covered stent makes its flexibility inferior to that of the bare stent.
Endovascular treatment also can lead to different complications [2,3,5,6,10]. Alaraj et al., found that immediate complications were recorded in 17 (9.1%) of 164 procedures, which included arterial dissection, arterial occlusion, intracerebral hemorrhage and embolic stroke [10]. Postprocedural TIA has been reported in 6 cases [6,11,12]. Subacute thrombosis and intimal hyperplasia may lead to delayed stenosis within the covered stents [6]. Dolmatch et al., reported a slower endothelialization and a thicker neointima using a covered stent compared to a bare-metal stent in an animal experiment [13]. Taha et al., reported a serious complication of immediate occlusion of the CCA after stopping an antiplatelet agent 3 months following the placement of a covered stent [14]. However, no antithrombotic therapy protocol about the duration and type of antiplatelet agent has been established on the use of a covered stent for ECAAs [6,10]. Based on the above, our patient received DAPT for 12 months and aspirin lifelong.
The main goal of the treatment is to prevent the thromboembolic complications of the aneurysm. It is achieved by excluding the aneurysm and restoring the blood flow. The treatment of choice should be based on size, type and location of the aneurysm, the underlying cause for its development, as well as the general condition of the patient [2]. Regarding the aneurysmal type, some authors found that long-term outcomes after the endovascular procedures were better in the treatment of pseudoaneurysms than true aneurysms [2,7]. This could be a result from the fact that most of the pseudoaneurysms are believed to be self-limited and not likely to reoccur after endovascular therapy, whereas the causes of true aneurysms, such as atherosclerosis, are ongoing and could lead to further aneurysmal degeneration [2]. In addition, true aneurysms often have a significant amount of thrombus inside the sac and frequently cause cerebral embolic events during the endovascular procedure [2,8]. Ni L et al., demonstrated good results in both types of aneurysms, as the procedure was successful in our case with saccular ECCA [2]. However, the limited follow-up and the number of patients do not allow a firm conclusion to be drawn about that.
According to the Attigah classification about aneurysm location, high type I and low type V aneurysms might be particularly suitable for an endovascular approach [9]. The reasons are related to the fact that type II endoleak from the patent ECA can be avoided when the aneurysms are far from the bifurcation of the CCA as it is in our case. Another reason is that these types of aneurysms allow stent-grafts to be deployed because the diameters of the proximal and distal landing zones are similar [2]. The only reasonable contraindication for endovascular treatment ishostile aortic arch (and its branches) anatomy mainly because of the large size of the delivery system of stentgrafts as well as its stiffness.
Endovascular techniques include the implantation of stents with or without embolization of the aneurysmal sac, stents coated with autogenous venous grafts, endovascular embolization or balloon occlusion, and placement of stent-grafts [5]. The meta-analysis of Li et al., showed a slight decrease in postoperative endoleak, a significantly lower rate of reintervention, overall late complications and stent-graft stenosis and a significant increase in thrombosis of the aneurysmal sac can be found in the patients treated with covered stents, which may support the covered stent as a better choice for ECAA [3]. However, the greater profile of covered stent makes its flexibility inferior to that of the bare stent.
Endovascular treatment also can lead to different complications [2,3,5,6,10]. Alaraj et al., found that immediate complications were recorded in 17 (9.1%) of 164 procedures, which included arterial dissection, arterial occlusion, intracerebral hemorrhage and embolic stroke [10]. Postprocedural TIA has been reported in 6 cases [6,11,12]. Subacute thrombosis and intimal hyperplasia may lead to delayed stenosis within the covered stents [6]. Dolmatch et al., reported a slower endothelialization and a thicker neointima using a covered stent compared to a bare-metal stent in an animal experiment [13]. Taha et al., reported a serious complication of immediate occlusion of the CCA after stopping an antiplatelet agent 3 months following the placement of a covered stent [14]. However, no antithrombotic therapy protocol about the duration and type of antiplatelet agent has been established on the use of a covered stent for ECAAs [6,10]. Based on the above, our patient received DAPT for 12 months and aspirin lifelong.
CONCLUSION
ECCAs are rare but represent a serious clinical condition in the light of potentially life-threatening complications. The endovascular approach could be the preferred therapeutic option in selected patients. However, a larger data-set and a longer follow-up are necessary to evaluate the safety and effectiveness of these procedures.
CONFLICT OF INTEREST
The authors hereby declare that they have no conflict of interest.
REFERENCES
- Miksi? K, Flis V, Kosir G, Pavlovic M, Tetickovic E (1997) Surgical aspects of fusiform and saccular extracranial carotid artery aneurysms. Cardiovasc Surg 5: 190-195.
- Ni L, Pu Z, Zeng R, Zhang R, Zheng YH, et al. (2016) Endovascular stenting for extracranial carotid artery aneurysms: Experiences and mid-term results. Medicine 95: 5442.
- Li Z, Chang G, Yao C, Guo L, Liu Y, et al. (2011) Endovascular stenting of extracranial carotid artery aneurysm: a systematic review. Eur J Vasc Endovasc Surg 42: 419-426.
- Welleweerd JC, Moll FL, de Borst GJ (2012) Technical options for the treatment of extracranial carotid aneurysms. Expert Rev Cardiovasc Ther 10: 925-931.
- Welleweerd JC, den Ruijter HM, Nelissen BG, Bots ML, Kappelle LJ, et al. (2015) Management of extracranial carotid artery aneurysm. Eur J Vasc Endovasc Surg 50: 141-147.
- Yamamoto S, Akioka N, Kashiwazaki D, Koh M, Kuwayama N, et al. (2017) Surgical and Endovascular Treatments of Extracranial Carotid Artery Aneurysms-Report of Six Cases. J Stroke Cerebrovasc Dis 26: 1481-1486.
- Fankhauser GT, Stone WM, Fowl RJ, O'Donnell ME, Bower TC, et al. (2015) Surgical and medical management of extracranial carotid artery aneurysms. J Vasc Surg 61: 389-393.
- Pulli R, Dorigo W, Alessi Innocenti A, Pratesi G, Fargion A, et al. (2013) A 20-year experience with surgical management of true and false internal carotid artery aneurysms. Eur J Vasc Endovasc Surg 45:1-6.
- Attigah N, Külkens S, Zausig N, Hansmann J, Ringleb P, et al. (2009) Surgical therapy of extracranial carotid artery aneurysms: long-term results over a 24-year period. Eur J Vasc Endovasc Surg 37: 127-133.
- Alaraj A, Wallace A, Amin-Hanjani S, Charbel FT, Aletich V (2011) Endovascular implantation of covered stents in the extracranial carotid and vertebral arteries: case series and review of the literature. Surg Neurol Int 2: 67.
- Assadian A, Senekowitsch C, Rotter R, Zölß C, Strassegger J, et al. (2004) Long-term results of covered stent repair of internal carotid artery dissections. J Vasc Surg 40: 484-487.
- Felber S, Henkes H, Weber W, Miloslavski E, Brew S, et al. (2004) Treatment of extracranial and intracranial aneurysms and arteriovenous fistulae using stent grafts. Neurosurgery 55: 631-638.
- Dolmatch BL, Tio FO, Li XD, Dong YH (1996) Patency and tissue response related to two types of polytetrafluoroethylene-covered stents in the dog. J Vasc Interv Radiol 7: 641-649.
- Taha MM, Nakahara I, Higashi T, Iwamuro Y, Watanabe Y, et al. (2010) Interventional neuroradiological techniques for the treatment of aneurysms of the supra-aortic extracranial arteries. Neurol Med Chir (Tokyo) 50: 275-280.
Citation: Boneva B, Nikolov N, Stoyanova B, Dimova M, Alexandov A, et al. (2019) Endovascular Treatment of Common Carotid Artery Aneurysm - Case Report and Review of Current Literature. J Angiol Vasc Surg 4: 027.
Copyright: © 2019 Bistra Boneva, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

Journal Highlights
© 2026, Copyrights Herald Scholarly Open Access. All Rights Reserved!
