
Expression of miRNA-33 and miRNA-155 in Symptomatic and Asymptomatic Carotid Artery Stenosis
*Corresponding Author(s):
Noemi CifaniSapienza Universita Di Roma, Facoltà Di Medicina E Psicologia, Dipartimento Di Medicina Clinica E Molecolare, UOC Medicina Interna, Ospedale Sant’Andrea, Roma, Italy
Tel:+39 633777710,
Email:noemi.cifani@uniroma1.it
Abstract
Background: The aim of this study was to evaluate miRNA-33 and miRNA-155 expression in Peripheral Blood Mononuclear Cells (PBMC) and carotid specimens of patients affected by Critical Carotid Artery Stenosis (CAS).
Material and Methods: We selected 17 asymptomatic (CAS-A group) and 10 symptomatic (CAS-S group) patients with CAS. Ten patients with traditional cardiovascular risk factors (RF group), matched for age and sex, were used as control group.
Results: A significant increase in miRNA-33 expression was observed both in peripheral blood and in carotid specimens of CAS-A patients (p 0.04) in comparison with CAS-S and RF, whereas no significant difference were found among the groups regarding miRNA-155 expression both in peripheral blood and in carotid specimens.
Conclusions: This is to our knowledge the first report on miRNAs expression in human PBMCs from CAS patients. Results of this study suggest that miRNA-33 in involved in the process underling plaque formation and growth, but not in plaque instability and ischemic brain damage, whereas miRNA-155 is expressed during all the phases of atherosclerotic disease.
Keywords
Carotid artery stenosis; Immunity; Inflammation; miRNAs
INTRODUCTION
MicroRNAs (miRNAs) are a class of small non-coding RNAs that regulate gene expression post transcriptionally by facilitating mRNA degradation and translation repression [1]. MiRNAs have emerged as critical endogenous regulators of several biological processes, such as cell proliferation development and death [2], angiogenesis and cholesterol metabolism [1,3]. As miRNAs participate in all of the main biological processes, deregulation of their functions may be responsible for several disorders, including Atherosclerosis (ATS) [4,5]. MiRNAs, indeed, are able to regulate macrophage activities as well as vascular smooth muscle cell and endothelial cell functions [4,5]. Atherosclerotic Carotid Artery Stenosis (CAS) leading to cerebrovascular disease, represents one of the main causes of morbidity and mortality around the world [6]. Clinical features related to CAS depend on entity of stenosis and speed of occlusion and range from asymptomatic to chronic cerebrovascular disease and acute ischemic brain damage [7].
In the field of ATS, MiRNAs play a relevant role in plaque growth and instability, by regulating cell adhesion molecule expression on endothelial cells, cholesterol metabolism, angiotensin 1 receptor expression and immune cell activation [5]. In particular, miRNA-33 has been related to lipid metabolism and diabetes mellitus [3], whereas miRNA-155 has been demonstrated to play multifunctional pro-atherogenic actions by controlling monocyte/macrophage responses, arterial blood pressure, vascular inflammation and lipid uptake [8].
Based on such premises, the aim of this study was to evaluate the expression of miRNA-33 and miRNA-155 in the peripheral blood and inside atherosclerotic plaques of patients affected by symptomatic and asymptomatic critical CAS.
MATERIAL AND METHODS
The present was an observational study. The population included in this study was composed of 27 patients with critical CAS selected in a period ranging from May 2016 to May 2017 among those undergoing to carotid Thromboendoarteriectomy (TEA) at the Department of Vascular Surgery, Sant’Andrea Hospital, “Sapienza” University of Rome. Patients were selected from about 200 subjects on the basis of the following inclusion criteria: (i) critical carotid stenosis, defined as a narrowing of the carotid lumen >70% [9]; (ii) no cardiac causes of stroke; (iii) no history of neoplasm, autoimmune or inflammatory systemic diseases; (iv) no familiar or personal history of aneurysms/dissection. All patients underwent to physical and neurological examinations, carotid artery ultrasound and angiography by Magnetic Resonance Imaging (MRI) or Contrast Tomography (CT). Brain CT scan or MRI were also peformed. Based on neurological symptoms and brain imaging, CAS patients were further divided into 2 subgroups: (a) group CAS-S (symptomatic CAS), and (b) group CAS-A (asymptomatic CAS). CAS-S included 10 patients with evidence of cerebro-vascular symptoms such as stroke or Transient Ischemic Attack (TIA) [10]. Patients were selected based on the following exclusion criteria: (i) doubtful neurological symptoms; (ii) negative brain imaging; (iii) symptoms onset >1month. All patients were included in the study in a period ranging from 2 weeks to 1 month from the onset of neurological symptoms; blood and tissue samples were collected at time of surgery. CAS-A comprised 17 patients who did not experienced any neurological symptoms within the previous 6 months, and their brain MRI was consistent with the diagnosis of chronic cerebrovascular disease.
Ten patients with traditional cardiovascular risk factors, matched for age and sex, attending to the Department of Atherosclerosis and Dyslipidemia, Sant’Andrea Hospital, “Sapienza” University of Rome were selected as control group (RF group). Patients were enrolled on the basis of the following criteria: (i) no acute cerebro-vascular symptoms or history of cardiovascular disease; (ii) no carotid stenosis >20%; (iii) no familiar or personal history of aneurysms/dissection. Physical and neurological examinations and carotid artery ultrasound were performed in all the controls.
A venous blood sample was withdrawn from each patient (just before surgery) and from each control, in order to isolate Peripheral Blood Mononuclear Cells (PBMCs) [11].
Carotid specimens were collected from all CAS patients and were fixed with 10% formalin immediately after surgery in order to extract miRNAs. For each carotid specimens have been taken two sections: the first one on the site of lesion (+) and the second one away from site of lesion that serves as negative control (-).
The study was performed according to the principles of the Declaration of Helsinki and was approved by the Ethics Committee of the Faculty of Medicine. Written informed consent was obtained from each patient or from an authorized family member.
PBMCs isolation
Blood samples were collected in tubes containing 0.2 ml sodium heparin and PBMCs were isolated by density gradient centrifugation (LympholyteVR , Cedarlane, Hornby, CA) as previously described [11]. PBMCs were used to extract miRNAs.
RNA extraction
MiRNAs were extracted from human carotid plaques using the High Pure miRNA Isolation Kit (Roche) according to the manufacturer’s instructions.
Total RNA, including miRNAs, was extracted from PBMCs using Trizol Reagent (Invitrogen) reagents according to the manufacturer’s instructions.
The RNA yield and concentrations were determined by NanoDrop spectrophotometer and all RNA samples were stored at -80°C until use.
MiRNA expression
MiRNA expression was analyzed using the two-step protocol of TaqMan MicroRNA Assays (Applied Biosystems), according to the manufacturer’s instructions.
Briefly, 8-10 ng of total RNA were reverse transcribed using miRNA-specific primers for human miRNA-155 (Applied Biosystems) and human miRNA-33 (Applied Biosystems) combined with reagents (dNTPs, reverse transcription buffer, RNase inhibitor, reverse transcriptase, and H2O nuclease free) from the Taqman miRNA Reverse Transcription kit (Applied Biosystems). The small nucleolar RNAU6B (Applied Biosystems) was used as internal control. The reactions were performed for 30 minutes at 16°C, 30 minutes at 42°C, 5 minutes at 85°C, and then stored at 4°C.
Real-Time quantitative Polymerase Chain Reaction (RT-qPCR) was subsequently conducted using specific TaqMan probes of the TaqMan microRNA Assay kits (Applied Biosystems) to quantify miRNA expression [12]. The reactions were incubated in a 96-well plate at the following thermal cycling conditions: 2 minutes at 50°C, 10 minutes at 95°C, (15 seconds at 95°C and 60 seconds at 60°C) for 40 cycles.
Data analysis
Relative quantification of miRNA expression was calculated with the 2- ΔΔCt method [13]. Moreover, each symptomatic and asymptomatic Patient (P) was randomly assigned to an RF patient (R) to analyze data, and values were normalized to an endogenous control as follow 2_(Ct[microRNA-155/33] -Ct [miRNA-U6B])P- (Ct[microRNA-115/33] _ Ct[miRNA-U6B]) RF. Results were represented as histograms reflecting the fold changes over the expression levels of the miRNAs.
Statistical analysis
Results for continuous variables were expressed as median and mean ± Standard Deviation (SD) and compared with the non-parametric Mann–Whitney test. Categorical variables were expressed in percentage and compared with the Chi square test. Values of p <0.05 was considered significant.
RESULTS
Patient characteristics
Demographic and baseline characteristics of patients included in the study are reported in table 1. No significant differences regarding age, sex, diabetes, dyslipidaemia and smoke were observed between the groups, with the exception for dyslipidaemia that was present in 100% of RF patients.
However, all RF patients were treated with statins. All hypertensive patients were treated with ace inhibitors or sartanic, with or not calcium antagonists or beta-blockers or diuretics. All diabetic patients were taking oral blood glucose lowering drugs and/or subcutaneous insulin.
|
|
Group CAS-A (n=17) |
M |
Group CAS-S (n=10) |
M |
Group RF (n=10) |
M |
|
Age (years) |
70.69±7.61 |
71.00 |
72.86±7.60 |
70.00 |
63.88±3.75 |
63.50 |
|
Female Sex (%) |
5/16 (31.25%) |
3/8(37,50%) |
6/10 (60.00%) |
|||
|
Diabetes |
5/16 (31.25%) |
4/8 (50.00%) |
3/10 (30.00%) |
|||
|
Dyslipidemia |
13/16 (81.25%) |
5/8 (52.50%) |
10/10 (100%) |
|||
|
Hypertension |
15/16 (100.00%) |
4/8 (50.00%) |
3/10 (30.00%) |
|||
|
Smoke |
11/16 (68.75%) |
|
4/8 (50.00%) |
|
1/10 (10.00%) |
|
Table 1: Demographic and baseline characteristics of patients.
MiRNA expression in PBMCs
Relative quantification revealed significantly higher level of miRNA-33 in CAS-A patients compared to both CAS-S (p=0.0396) and RF-patients (p=0.0036), whereas any significant difference occur between CAS-S and RF patients (Figure1A, Table 2).
Results were confirmed also when by random comparison of each symptomatic and asymptomatic
patient with a RF patient (CAS-A versus CAS-S p=0.0290) (Figure 1B, Table 3).
The expression of miRNA-155 progressively increase from RF to CAS-A group, despite no significant differences were observed (Figures 1, Tables 2 & 3).
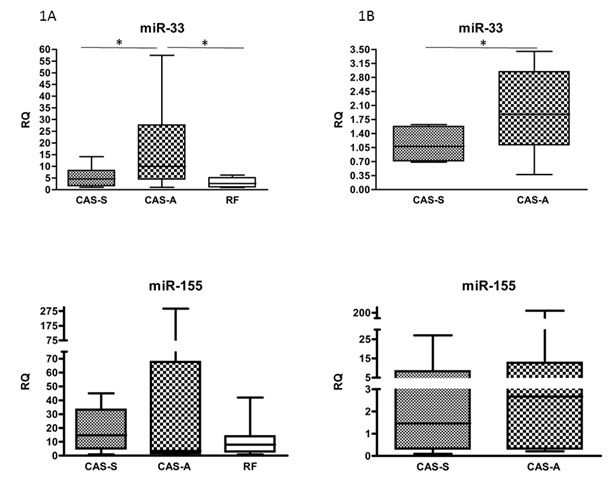 Figure 1: Expression of microRNAs in PBMCs of CAS-A, CAS-S and RF patients. Data are expressed as median and mean ± SD.
Figure 1: Expression of microRNAs in PBMCs of CAS-A, CAS-S and RF patients. Data are expressed as median and mean ± SD.
|
Mean ± SD, median |
CAS-S |
CAS-A |
RF |
|
miRNA-155 |
18.96±16.63, 14.67 |
45.20±86.34, 3.42 |
11.05±12.58, 7.90 |
|
miRNA-33 |
5.31±4.21, 4.65 |
15.23±15.10, 10.01 |
3.04±1.95, 2.63 |
Table 2: RQ values of microRNAs in PBMCs of CAS-A, CAS, S and RF patients. Data are expressed as Mean ± SD, median.
|
Mean ± SD, median |
CAS-S |
CAS-A |
|
miRNA-155 |
5.92±9.41, 1.46 |
24.38±59.78, 2.67 |
|
miRNA-33 |
1.31±0.39, 1.08 |
2.01±1.02, 1.88 |
Table 3: RQ values of microRNAs in PBMCs of CAS-A, CAS, S and RF patients. Data are expressed as Mean ± SD, median.
MiRNA expression in carotid specimens
RT- qPCR showed that miRNA-33 was expressed in 14 out of 17 (82.35%) carotid specimens from CAS-A patients and in 10 out of 10 (100%) from CAS-S, both in positive and negative sections. A significantly increased expression of miRNA-33 was observed in CAS-A+ versus CAS-S+ specimens (p=0.04) (Figure 2, Table 4).
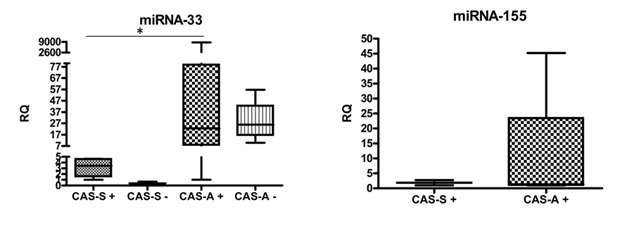 Figure 2: Expression of microRNAs in carotid specimens of CAS-A and CAS-S patients. Data are expressed as median and mean ± SD.
Figure 2: Expression of microRNAs in carotid specimens of CAS-A and CAS-S patients. Data are expressed as median and mean ± SD.
|
Mean ± SD, median |
CAS-S + |
CAS-S - |
CAS-A + |
CAS-A - |
|
miRNA-155 |
1.86±0.85, 1.86 |
/ |
12,23±22,00, 1.36 |
/ |
|
miRNA-33 |
3.15±1.59, 3.45 |
0.39±0.42, 0.39 |
893.2±2731.00, 22.70 |
29.58±19.68, 25.86 |
Table 4: RQ values of microRNAs in carotid specimens of CAS-A, CAS, S and RF patients. Data are expressed as Mean ± SD, median.
MiRNA-155 expression was never detected in negative controls, whereas was observed in 6 of the 17 (35.29%) CAS-A and in 4 of the 10 (40.00%) CAS-S positive carotid specimens analyzed. No significant difference were observed regarding miR-155 expresson, despite relative quantification revealed an higher level of miR-155 in CAS-A patients compared to CAS-S (Figure 2, Table 4).
DISCUSSION
This is to our knowledge the first report on miRNAs expression in human PBMCs from CAS patients. Results of this study demonstrated a different and peculiar expression of miRNA-33 in peripheral blood and carotid specimens of patients affected by asymptomatic and symptomatic CAS, suggesting that miRNA-33 is not related to plaque instability and to acute ischemic brain damage, but its expression increases in patients with chronic disease. Considering the role of MiRNA-33 on lipid metabolism, it is not surprisingly that such MiRNA is involved in severe atherosclerotic carotid artery disease [3]. Cholesterol homeostasis, indeed, is strongly related to ATS risk and its progression [10]. MiRNA-33 acts as a post-transcriptional regulator of lipid metabolism by targeting ATP-binding cassette transporter A1. Inhibition of miRNA-33, therefore, raises plasma High Density Lipoprotein (HDL) Levels, Reduces Very Low Density Lipoprotein (VLDL) triglycerides and promotes reverse cholesterol transport, exerting protective effects against ATS [3,10]. This has been repeatedly confirmed in mice apoE-/E- in whom inhibition of MiRNA-33 ameliorated lipid profile and prevent progression of atherosclerotic lesions [3,10].
However, the role of MiRNA-33 in ATS is more complex than expected. Its effects on inflammatory mediators, indeed, seem to be paradoxical. MiRNA-33 expression has been inversely related to with pro-inflammatory cytokine levels, therefore inhibition of MiRNA-33 promotes macrophage differentiation and pro-inflammatory cytokine release [10]. Thus, suggesting that MiRNA-33 expression can exert pleiotropic actions in the different stages of ATS disease also by affecting other risk factors such as obesity, glucose metabolism and inflammation [10]. This can partly explain our results, dyslipidemia, indeed, represents a key event in plaque formation and growth, whereas inflammation works during all the phases of ATS and represents the main factor leading to plaque instability [14].
Further confirming the complexity of the actions exerted by MiRNA in the different phases of ATS, accordingly with the literature, we found a low expression of MiRNA-33 in RF patients who were all affected by dyslipidemia and all under treatment with statins and/or ezetimibe and did not show any sign of carotid ATS [10]. Actually, statin effects on lipid profile have also been associated with a significant decrease of miRNA-33 expression. Results obtained from miRNA-155, confirm that it is expressed in all the phases of ATS, from subclinical stages to critical occlusion, being deeply related to the different steps of the immune-inflammatory process underlying plaque formation, growth and complications. This miRNA, indeed, plays a major role in OxLDL uptake, monocyte/macrophage activation and oxLDL stimulated inflammatory responses that are key events in early phase of ATS [11,15,16]. Moreover, it mediates intraparietal interaction between dendridic cell and T lymphocytes, promotes T lymphocyte differentiation and macrophage release of pro-inflammatory cytokine, being implicated also in plaque growth and destabilization [17]. In agreement, we did not find any significant difference in MiRNA-155 expression among RF, CAS-A and CAS-S patients. However, miRNA-155 has also implicated in lowering inflammation [18], suggesting that it plays a role in regulating immune-inflammatory process underling to the different stages of parietal remodeling.
This is, to our knowledge, the first report on expression of MiRNAs in human PBMCs from CAS patients. Our results suggest that symptomatic and asymptomatic patients affected by CAS display a different expression of miRNA-33, suggesting that this miRNA plays a role in plaque formation and progression, but not in instability, whereas miRNA-155 is expressed in all the phases of ATS process.
AUTHOR CONTRIBUTION
DF and NC wrote the main manuscript test, PM and LM prepared figures and all authors reviewed the manuscript.
ADDITIONAL INFORMATION
Competing interest statement
The authors have no actual or potential conflict of interest to declare, including any financial and non-financial, personal or other relationships with other people or organizations that could inappropriately influence, or be perceived to influence, their work.
Sources of support
Università di Roma ‘La Sapienza’, Progetto di Ricerca di Università n C26A15FL92, funded this study. The Authors have not any pharmaceutical and industry founding support, that require acknowledgment to declare.
Ethical approval
The study was performed according to the principles of the Declaration of Helsinki and was approved by the Ethics Committee of the Faculty of Medicine. Written informed consent was obtained from each patient or from 5.
ACKNOWLEDGMENT
The authors thank Andrea Micaloni for technical support with carotid specimens collection and processing.
REFERENCES
- Menghini R, Stohr R, Federici M (2014) MicroRNAs in vascular aging and atherosclerosis. Aging Res Rev. 17: 68-78.
- Hwang HW, Mendell JT (2006) MicroRNAs in cell proliferation, cell death, and tumorigenesis. Br J Cancer. 94: 776-80.
- Rayner KJ, Suarez Y, Davalos A, Parathath S, Fitzgerald ML, et al. (2010) MiR-33 contributes to the regulation of cholesterol homeostasis. Science 328: 1570-1573.
- Egea V, Schober A, Weber C (2012) Circulating miRNAs: messengers on the move in cardiovascular disease. Thromb Haemost 108: 590-151.
- Andreou I, Sun X, Stone PH, Edelman ER, Feinberg MW, et al. (2015) MiRNAs in atherosclerotic plaque initiation, progression, and rupture. Trends Mol Med 21: 307-318.
- Lopez AD, Mathers CD, Ezzati M, Jamiston DT, Murray CJ (2006) Global and regional burden of disease and risk factors, 2001: systematic analysis of population health data Lancet 367: 1747-1757.
- Muluk SC, Muluk VS, Sugimoto H, Rhee RY, Trachtenberg J, et al. (1999) Progression of asymptomatic carotid stenosis: A natural history study in 1004 patients. J Vasc Surg 29: 208-214.
- Ma X, Ma C, Zheng X (2013) MicroRNA-155 in the pathogenesis of atheroclerosis: A conflicting role? Heart Lung Circ 22: 811-818.
- Raman G, Moorthy D, Hadar N, Dahabreh IJ, O'Donnell TF, et al. (2013) Management strategies for asymptomatic carotid stenosis: a systematic review and meta-analysis. Ann Intern Med 158: 676-685.
- Koyama S, Horie T, Nishino T, et al. (2019) Identification of differential roles of MicroRNA-33 a and -33b during atherosclerosis progression with genetically modified mices. J Am Heart Assoc 8.
- Del Porto F, Cifani N, Proietta M, Perrotta S, Dito R, et al. (2017) Regulatory T CD4 + CD25+ lymphocytes increase in symptomatic carotid artery stenosis. Ann Med 49: 283-290 .
- Maitrias P, Metzinger-Le Meuth V, Massy ZA, M'Baya-Moutoula E, Reix T, et al. (2015) MicroRNA deregulation in symptomatic carotid plaque. J Vasc Surg 62: 1245-1250.
- 13. Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)), Method. Methods. 25: 402-408.
- Rayner KJ, Moore KJ (2012) The plaque “micro” environment: microRNAs control the risk and the development of atherosclerosis. Curr Atheroscler Rep 14: 413-421.
- Nazari-Jahantigh M, Wei Y, Noels H, Akhtar S, Zhou Z, et al. MicroRNA-155 promotes atherosclerosis by repressing Bcl6 in macrophages. J Clin Invest 122: 4190-202.
- Del Porto F, Cifani N, Proietta M, Toni D, Taurino M (2017) MMP-12 and TIMP Behavior in Symptomatic and Asymptomatic Critical Carotid Artery Stenosis. J Stroke Cerebrovasc Dis 26: 334-338.
- Donners MM, Wolfs IM, Stöger LJ, van der Vorst EPC, Pöttgens CCH, et al. Hematopoietic miR155 deficiency enhances atherosclerosis and decreases plaque stability in hyperlipidemic mice. PLoS One 7: 35877.
- Zhang F, Zhao J, Sun D, Wei N (2018) MiR-155 inhibits transformation of macrophages into foam cells via regulating CEH expression. Biomed Pharmacother 104: 645-651.
Citation: Cifani N, Proietta M, Dezi T, Luciani M, Raffa S, et al. (2020) Expression of miRNA-33 and miRNA-155 in Symptomatic and Asymptomatic Carotid Artery Stenosis. J Angiol Vasc Surg 5: 047.
Copyright: © 2020 Noemi Cifani, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

