
Focal Nerve Compression by Tenosynovitis as a Concomitant Cause of Neuropathic Pain in Guillain-Barré Syndrome: a Case Report
*Corresponding Author(s):
Rui PeresPhysical And Rehabilitation Medicine Department, ULS Amadora E Sintra, Amadora, Portugal
Email:ruipedroperes@gmail.com
Abstract
Pain is a common symptom in the acute and sub-acute phase of Guillain-Barré syndrome (GBS), with several possible etiologies. It tends to be disabling, difficult to diagnose and a challenge to treat. Usual pain relief medication is sometimes ineffective and there is no consensus to the treatment approach. The diagnosis workup is hindered when there are concomitant pathologies contributing to different pain mechanisms from those described in the literature. The authors report a case of a 55-year-old man with focal neuropathic pain secondary to tenosynovitis of the extensor digitorum longus muscle after an acute onset of GBS in a patient with Sjogren’s syndrome. Diagnostic workup included ultrasound assessment and electromyographic sensory nerve conduction tests. Complete symptom relief was obtained after extensor digitorum longus tendon sheath effusion aspiration and corticoanaesthetic ultrasound-guided injection. Focal nerve compression secondary to tendon inflammation as a cause of neuropathic pain in a GBS setting should be considered, especially if the patient has other auto-immune diseases.
Keywords
Guillain-Barré syndrome; Sjogren syndrome; Neuropathic pain; Tenosynovitis; Ultrasound; Corticoanaesthetic injection.
Introduction
Guillain-Barré syndrome (GBS) is an acute immune-mediated polyradiculoneuropathy characterized by acute progressive motor weakness and areflexia [1]. GBS is most commonly a post-infectious disorder, with about two-thirds of patients having symptoms of an infection 3 weeks before the onset of weakness. 1 Apart from the motor impairment, sensory symptoms associated with GBS, especially pain, have great impact on quality of life. Although several types of pain have been described in GBS, ranging from radicular pain, meningism, dysesthesias, muscle pain to arthralgia, the pathophysiology is still poorly understood [2].
Sjogren’s syndrome (SS) is an autoimmune disorder characterized by lymphocyte infiltration of salivary and lachrymal glands [3]. Although the main manifestation is sicca complaints, involvement of the musculoskeletal system (targeting bones, joints and muscles) and of the central and peripheral nervous system may also occur [4,5]. These systemic manifestations may present with different types of pain, ranging from inflammatory, due to arthritic and muscle involvement, to neuropathic from small vessel vasculitis [5].
The literature available on neuropathic pain (frequency, character and intensity) in sub-acute GBS is limited. The correct identification and characterization of pain becomes more problematic when it occurs concomitantly with inflammatory pathologies with different pain mechanisms. Without clinical suspicion and the correct diagnosis, management and treatment of these patients is a challenge. This report presents a case of focal neuropathic pain secondary to tenosynovitis of the extensor digitorum longus after an acute onset of GBS in a patient with SS.
Case Report
A 55-year-old male, with a medical history of essential hypertension, presented to the emergency department with fever associated with headache, fatigue and erythematous pruritic rash in the lower limbs. Physical examination revealed no abnormalities except hepatomegaly. Laboratory findings were normal except for an elevated C-reactive protein level (27.91 mg/dl, normally 9/L leucocytes, normal range 4.5 to 11.0 × 109/L; platelets count 140,000/mcl, normal range 150,000 to 450,000/mcl), and an hepatic cytolysis pattern (glutamic-oxalacetic transaminase (TGO) 128 U/L, normally 5 to 40 U/L; glutamic pyruvic transaminase (TGP) 108 U/L, normally 7 to 56 U/L). Blood cultures and viral serologies were negative except for Epstein-Barr virus viral capsid antigen (IgG and IgM positive). Abdominal ultrasound confirmed hepatomegaly and splenomegaly. Serum serologies showed positive anti-SSA antibodies and salivary gland biopsy showed chronic sialoadenitis. The patient was diagnosed with macrophage activation syndrome secondary to Epstein-Barr virus infection and primary Sjogren's syndrome. Clinical course was favorable with supportive therapy and the patient was discharged after 15 days.
Two days after hospital discharge the patient presented again to the emergency department with facial biparesis, dysarthria, gait imbalance and low back pain radiating to the lower limbs. On examination he showed wide-based gait with positive Romberg sign, lower limb areflexia and bilateral peripheral facial nerve palsy (PFP) with asymmetry at rest and incomplete closure of the left eye (80% of palpebral excursion, with Bell's sign). No other abnormalities were detected on neurological examination.
Cranioencephalic magnetic resonance imaging (MRI) documented a slight anomalous enhancement of the facial nerve bilaterally; lumbar puncture showed an albuminocytological dissociation (white blood cell count: 10 cells/µL; protein level: 82.2 mg/dL) and electromyographic (EMG) study showed moderate axonal damage to the facial nerve bilaterally. Spine MRI and laboratory testing showed no abnormalities.
A diagnosis of a regional variant of GBS was established and the patient started therapy with intravenous immunoglobulin and was referred to Physical Medicine and Rehabilitation (PRM). Good motor improvement under neuromuscular reeducation with progressive achievement of facial movement symmetry and efficient gait balance. However, during hospitalization, the patient began to refer pain in the left lower limb, with implication in the progression of the rehabilitation program. On assessment, the patient presented pain with mixed characteristics with a neuropathic component, located on the dorsum of the foot in a well-circumscribed area, compatible with superficial peroneal nerve dermatome (figure 1).
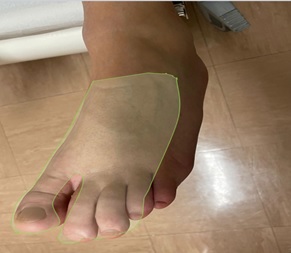 Figure 1: Marking of the neuropathic painfull area in the dorsum of the left foot.
Figure 1: Marking of the neuropathic painfull area in the dorsum of the left foot.
He reported a pain intensity of 7/10 on the numerical rating scale (NRS), which worsened with plantar flexion (NRS 9/10). Neuropathic Pain Questionnaire (DN4) was performed, scoring 5/10: burning sensation and electric shocks, tingling and pins/needles and increased pain with brushing. Tenderness over the extensor digitorum longus was also present. A bedside ultrasound evaluation was performed and documented exuberant extensor digitorum longus tenosynovitis with frank effusion (figure 2).
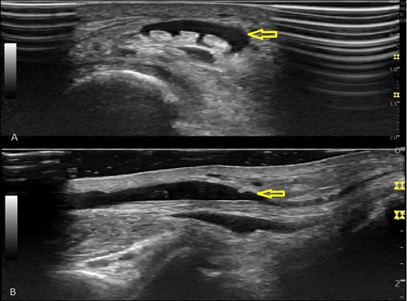 Figure 2: Ultrasound image of the extensor digitorum longus with effusion, in transverse (A) and longitudinal (B) planes.
Figure 2: Ultrasound image of the extensor digitorum longus with effusion, in transverse (A) and longitudinal (B) planes.
Since the pain presented mixed characteristics (nociceptive and neuropathic), a provisional diagnosis was made of extensor digitorum longus tenosynovitis (in the likely context of a systemic inflammatory syndrome of multiple causes - macrophage activation syndrome, SS and GBS) and of neuropathic pain of unidentified cause (in the likely context of GBS and spontaneous activity of myelinated sensory afferent nerves). Conservative treatment was attempted, with local cryotherapy and non-steroidal anti-inflammatory drugs for 5 days. Discrete reduction of local pain was documented (NRS 6/10), but exuberant extensor digitorum longus effusion on ultrasound evaluation was still present. Treatment with minor opioids and gabapentin was started, although with little therapeutic response. As pain persisted despite optimization of analgesic treatment and significant neurological recovery, an electromyogram of the lower limb was performed on suspicion of local compression of the superficial peroneal nerve due to tendon effusion. The sensory nerve conduction study documented an immeasurable action potential of the left superficial peroneal nerve, confirming the clinical suspicion (figure 3).
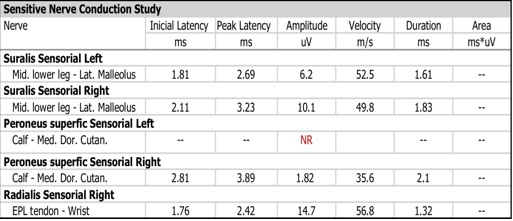 Figure 3: Electromyographic sensory nerve conduction study with an immeasurable action potential of the left superficial peroneal nerve.
Figure 3: Electromyographic sensory nerve conduction study with an immeasurable action potential of the left superficial peroneal nerve.
In order to reduce local effusion and reduce compression of the superficial peroneal nerve, a corticoanaesthetic ultrasound-guided injection was performed on the extensor digitorum longus with 1 cc of methylprednisolone 40mg/ml + 1 cc of lidocaine 1% (figure 4). There were no complications arising from the intervention and the patient was discharged.
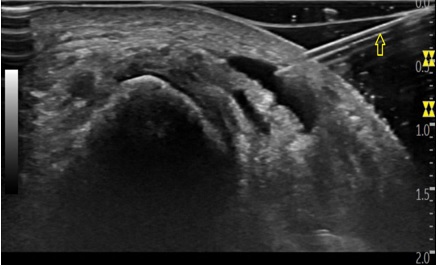 Figure 4: Ultrasound image of the ultrasound-guided intervention on the extensor digitorum longus with corticoanaesthetic injection.
Figure 4: Ultrasound image of the ultrasound-guided intervention on the extensor digitorum longus with corticoanaesthetic injection.
Re-evaluation 2 weeks after the intervention showed reduction of effusion and tendon thickness, with slight pain reduction (NRS 4/10), although neuropathic descriptors were still present. At Follow-up after 4 weeks, persistent discomfort and worsening of tenosynovitis and effusion was documented. A new intervention was performed with aspiration of the effusion (drainage of 0.75 cc of citric yellow liquid) and a new corticoanaesthetic injection with 1 cc of methylprednisolone 40mg/ml + 1 cc of lidocaine 1% under ultrasound guidance. Re-evaluation 4 weeks after the second intervention showed total resolution of pain, no neuropathic descriptors and no tenderness on examination. Ultrasonography showed a reduction of inflammatory activity and effusion (figure 5).
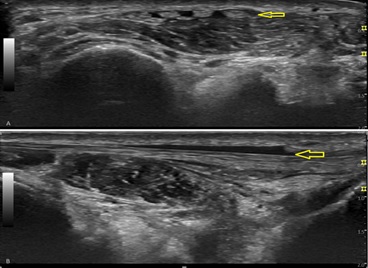
Figure 5: Ultrasound image of the extensor digitorum longus 4 weeks after intervention, in transverse (A) and longitudinal (B) planes.
Discussion
In this paper we describe a case of focal neuropathic pain secondary to tenosynovitis of the extensor digitorum longus after an acute onset of GBS in a patient with SS. Diagnosis and management was difficult due to the patient presenting pain with mixed characteristics: neuropathic in the acute context of GBS, primarily assumed to be secondary to GBS and spontaneous activity of myelinated sensory afferent nerves, and nociceptive by extensor digitorum longus tenosynovitis in the context of SS. The lack of response to conservative treatment prompted a new diagnostic workup. The use of ultrasound with dynamic assessment made it possible to identify tenosynovitis with significant effusion and raise the suspicion of focal peroneal nerve compression. Targeted treatment with effusion aspiration and corticoanaesthetic ultrasound-guided injection in the extensor digitorum longus tendon sheath proved effective and safe in this patient.
There have been no case reports published regarding focal nerve compression by inflammatory tendon pathology in an acute GBS setting. In our case, the concurrent diagnosis of SS may be a possible explanation of an increased risk of tendinopathy due to a pro-inflammatory environment.
Pain is a common symptom in the acute phase and during the later stages of GBS, being described in up to 89% of patients [6,7]. Painful paresthesiae and radicular, muscle and articular pain are common complaints in the extremities [2]. Although the pathophysiology of pain in GBS is largely unknown, several possible mechanisms have been described. In the acute phase of GBS pain might be of nociceptive origin due to nerve inflammation. In contrast, pain in the chronic phase may result from degeneration or even regeneration of sensory nerve fibers with non-nociceptive pain [8]. Neuropathic pain secondary to inflammation or local nerve damage with abnormal activity from large myelinated sensory afferents may explain the occurrence of radicular pain [2]. The same mechanism has been described as a cause of muscle aches and cramps, with no apparent relation with mechanical irritation of the small unmyelinated motor nerve terminals as previously described by Robert B. Layzer, but by affection of sensory afferents [9,10]. Other mechanisms proposed have advocated inflammatory reactions via nervi nervorum and damage to small nerve fibers with a reduction of intraepidermal nerve fiber density [11].
Pain treatment in GBS is often far from optimal, with reported moderate success of systemic corticosteroids, gabapentin, carbamazepine, and opioids [12-14]. Because of limited study results there is insufficient evidence to support the use of any pharmacological intervention in patients with pain in GBS in the latest Cochrane systematic review [15].
The prevalence of peripheral neuropathy in SS patients has been described to be up to 27% [16]. Although there have been several mechanisms proposed to explain peripheral neuropathy in SS, the most accepted theory is a vasculitis pathogenesis for axonal sensory-motor neuropathies, presenting altered conduction velocity values similar to a compression neuropathy [5]. However, no publication has been done describing the same electromyographic alterations due to a true focal compression.
In our case, response to ultrasound guided effusion aspiration and corticosteroid injection of the extensor digitorum longus tendon sheath, with resolution of the neuropathic pain presented by the patient, suggests the focal compression of a branch of the superficial peroneal nerve by the tendon effusion secondary to SS as the cause of this neuropathic pain rather than true nerve involvement. However, it has to be considered that intravenous corticosteroid administration in high doses, around 120 mg/day for 3 days with a 21 day tapper, has been described to be effective in neuropathic pain in an acute GBS setting, without documented focal nerve compression [13].
Thus, published literature is insufficient to attribute focal nerve compression secondary to tendon inflammation as a cause of neuropathic pain in a GBS setting, apart from this case, leading to a need of additional studies to establish this relationship.
Conclusion
We describe a case of GBS and SS with focal neuropathic pain arising from extensor digitorum longus tenosynovitis. Pain management in an acute GBS setting is challenging. In this case, the persistent neuropathic pain in the superficial peroneal nerve dermatome, with no response to medical treatment of GBS or pain medication, led to a further analysis and examination of the patient. The correct identification, diagnosis and treatment are of vital importance to the prognosis of the patient and to ensure an optimization of physical and motor recovery. Focal nerve compression should be considered as a cause of neuropathic pain especially if the patient has other auto-immune diseases.
Funding
Statement of conflicts of interest and source of funding: none declared
References
- Kountouras J, Deretzi G, Grigoriadis N, Zavos C, Boziki M, et al. (2008) Guillain-Barré syndrome. Lancet Neurol 7: 1080-1081.
- Ruts L, Drenthen J, Jongen JL, Hop WC, Visser GH, et al. (2010) Pain in Guillain-Barre syndrome: a long-term follow-up study. Neurology 19: 1439-1447.
- Nair JJ, Singh TP (2017) Sjogren’s syndrome: Review of the aetiology, Pathophysiology & Potential therapeutic interventions. J. Clin. Exp. Dent 9: 584-589.
- Rozis M, Vlamis J, Vasiliadis E, Mavragani C, Pneumaticos S, et al. (2021) Musculoskeletal Manifestations in Sjogren’s Syndrome: An Orthopedic Point of View. J. Clin. Med 10: 1574.
- Fauchais AL, Magy L, Vidal E (2012) Central and peripheral neurological complications of primary Sjogren’s syndrome. Presse Med 41: 485-493.
- Forsberg A, Press R, Einarsson U, de Pedro-Cuesta J, Widén Holmqvist L, et al. (2004) Impairment in Guillain-Barré syndrome during the first 2 years after onset: a prospective study. J Neurol Sci 227: 131-138.
- Moulin DE, Hagen N, Feasby TE, Amireh R, Hahn A (1997) Pain in Guillain-Barré syndrome. Neurology 48: 328-331.
- van Doorn PA, Ruts L, Jacobs BC (2008) Clinical features, pathogenesis, and treatment of Guillain-Barre´ syndrome. Lancet Neurol 7: 939-950.
- Layzer RB (1994) The origin of muscle fasciculations and cramps. Muscle Nerve 17: 1243-1249.
- Bernsen RA, Jager AE, Schmitz PI, van der Meché FG (2001) Long-term sensory deficit after Guillain-Barré syndrome. J Neurol 248: 483-486.
- Pan CL, Tseng TJ, Lin YH, Chiang MC, Lin WM, et al. (2003) Cutaneous innervation in Guillain-Barré syndrome: pathology and clinical correlations. Brain 126: 386-397.
- Connelly M, Shagrin J, Warfield C (1990) Epidural opioids for the management of pain in a patient with the Guillain-Barré syndrome. Anesthesiology 72: 381-383.
- Kabore R, Magy L, Boukhris S, Mabrouk T, Lacoste M, et al. (2004) Contribution of corticosteroid to the treatment of pain in the acute phase of Guillain-Barré syndrome [in French]. Rev Neurol 160: 821-823.
- Pandey CK, Raza M, Tripathi M, Navkar DV, Kumar A, et al. (2005) The comparative evaluation of gabapentin and carbamazepine for pain management in Guillain-Barré syndrome patients in the intensive care unit. Anesth Analg 101: 220-225.
- Liu J, Wang LN, McNicol ED (2015) Pharmacological treatment for pain in Guillain-Barré syndrome. Cochrane Database Syst Rev 9: 2015.
- Gøransson LG, Herigstad A, Tjensvoll AB, Harboe E, Mellgren SI, et al. (2006) Peripheral neuropathy in primary sjogren syndrome: A population-based study. Arch. Neurol 63: 1612-1615.
Citation: Peres R, Ferreira EF, Nunes R, Flores S, Matos C (2025) Focal Nerve Compression by Tenosynovitis as a Concomitant Cause of Neuropathic Pain in Guillain-Barré Syndrome: a Case Report. J Phys Med Rehabil Disabil 11: 097.
Copyright: © 2025 Rui Peres, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

