
Hepatic Portal Venous Gas in a Patient Submitted to Percutaneous Gastrostomy: Case Report and Literature Review
*Corresponding Author(s):
Luiz Carlos De Araújo SouzaResearcher Of The Department Of Cytopathology And Pathological Anatomy Of The Base Institute Of The , St. Hospital Medical South - Asa Sul, Brasilia, Brazil
Tel:+55 61999232232,
Email:luiz_carlos5@hotmail.com
Abstract
Introduction
Hepatic Portal Venous Gas (HPVG) is a rare condition associated with several pathological conditions that can evolve with high morbidity.
Presentation of case
A 89-year-old female patient who needed hospitalization for pneumonia and intestinal constipation. The patient used antibiotic therapy with piperacillin and tazobactam for pneumonia but evolved with Enterecoccus cystitis. Due to the difficulty of motor rehabilitation and deglutition, they chose to perform a percutaneous endoscopic gastrostomy. After two days, the patient presented diffuse abdominal pain, abdominal distension, nausea, vomiting, tachycardia, hypotension, without signs of peritonitis. Laboratory tests showed leukocytosis (15.570 leukocytes /mm3), C-reactive protein (166 mg/dl) and worsening of renal function (1.4 mg/dl). Computed Tomography (CT) revealed abdominal HPVG and small pneumoperitoneum near the area of gastrostomy. After clinical deterioration with septic shock, they opted for an exploratory laparotomy that showed cloudy ascites and the gastrostomy was well positioned, with no signs of leakage into the cavity. Despite the intensive support, the patient evolved with multiple organ failure and death.
Discussion and conclusion
HPVG is a rare radiological signal associated with severe underlying pathological conditions in need of early intervention. With the ease of CT abdomen, the diagnosis of HPVG is increasing, so it is up to the surgeon to indicate the operative procedure when this is necessary, especially when there is evidence of intestinal ischemia.
Keywords
INTRODUCTION
Hepatic Portal Venous Gas (HPVG) is a rare condition, first described by Wolfe and Nevins (1955) in neonates with Necrotizing Enterocolitis (NEC). Scientific work has shown that this pathology is associated with several pathological conditions of high morbidity, requiring rapid and effective treatment [1,2].
Liebman et al., in describing the diseases most commonly associated with HPVG determined that intestinal necrosis (72%), followed by ulcerative colitis (8%), intra abdominal abscess (6%), small bowel obstruction and gastric ulcer (3%) are the most associated, explaining the high mortality rate (56%-90%) reported in association with HPVG [3-5].
The physiopathology for the onset of HPVG is not well elucidated. The main factors that predispose the portal venous system to gas accumulation include: The escape of gas produced by gas-forming organisms in the intestinal lumen or in an abscess circulating in the liver or the presence of gas-forming organisms in the venous system with gas passage to the circulation [3].
One of the main methods for the diagnostic elucidation of HPVG is simple abdominal radiography, ultrasonography, color Doppler flow or Computed Tomography (CT). In this case, one of the main characteristic findings on abdominal plain is a branching radiolucency extending to within 2cm beneath the liver capsule [6].
With the advent of CT, the identification of intestinal pneumatosis became an important sign in the diagnosis of patients with intestinal ischemia justifying HPVG [7]. CT as well as other imaging tests help in the decision making regarding the need for a surgical approach when associated with clinical evaluation data. As most cases are associated with intestinal ischemia, surgical exploration is often necessary, especially in the presence of intestinal pneumatosis [7].
In this work we present a case report of HPVG in a patient undergoing percutaneous gastrostomy.
PRESENTATION OF CASE
A 89-year-old female patient admitted to the ward of neurology due to a cerebrovascular accident in the middle cerebral artery. As background, the patient had intermittent controlled atrial fibrillation, heart failure and left internal carotid artery stenosis. During hospitalization, the Patient presented pneumonia on day 7 and intestinal constipation resolved with glycerin clister. At the end of the antibiotic Therapy with piperacillin and tazobactam for pneumonia on the 14th day of hospitalization. On the 17th day of hospitalization, the Patient presented Vancomycin-sensitive Enterecoccuscy stitis. On the 21st day of hospital stay, after resolution of the pulmonary infection, the patient presented good general condition and due to the difficulty of motor rehabilitation and swallowing, chosen for percutaneous endoscopic gastrostomy.
Two days after the gastrostomy, the Patient had abdominal distention, nausea and vomiting. At the evaluation of the surgery team, the Patient complained of diffuse abdominal pain, was tachycardic and hypotensive (Blood pressure: 96x60 mmhg). At physical examination, diffuse abdominal pain, worse in the hypogastroregion, without signs of peritonitis. Laboratory tests showed leukocytosis (15.570 leukocytes /mm3), C-reactive protein: 166 mg/dl (previous 16 mg/dl) and worsening of renal function (acute renal failure, creatinine of 0.81 mg/dl initial evolving to 1.4 mg/dl).
After performing abdominal CT that showed HPVG and small pneumoperitoneum near the area of gastrostomy. Considering CT findings (Figure 1) and clinical deterioration with septic shock, we chose exploratory laparotomy in view of possible intestinal ischemia/peritonitis. The exploratory laparotomy was found to be turbidascitic fluid in small amount that was sent to culture. The gastrostomy was well positioned, with no signs of leakage into the cavity. No lesions were found in intestinal loops or lesions of abominable viscera. In the postoperative follow-up, there was a progressive worsening of the septic shock with need for vasoactive drugs in the first post operative day.
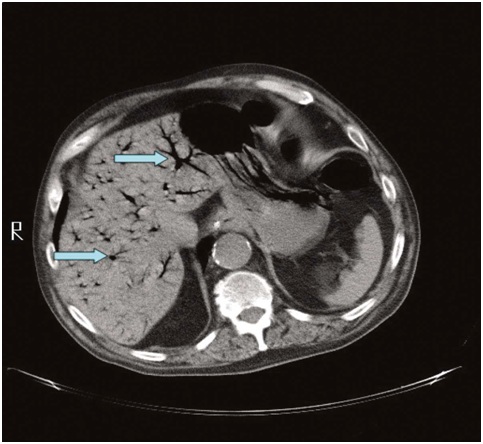 Figure 1: Computed tomography of the abdomen, the blue colored arrows identify and shows the hepatic portal venousgas.
Figure 1: Computed tomography of the abdomen, the blue colored arrows identify and shows the hepatic portal venousgas.
Maintain edonmechanical ventilation and anti biotic therapy with meropenem and vancomycin since the surgical procedure.
After isolating cultures from the onset of the carbapenem sensitive Proteus mirabiliss epticum and the ascitic fluid culture collected at the operative procedure, it isolated vancomycin-sensitive Staphylococcus aureus. On the 6th post operative day, the patient did not receive surgical indications for surgical re-placement, since the gastrostomy was well positioned, with no signs of cavity leakage and no lesion was found in intestinal loops or lesions of abominable viscera. On the 6th postoperative day, the patient evolved with multiple organ failure and death, despite the intensive support.
DISCUSSION
In relation to the patho physiological mechanisms of HPVG, there are two main mechanisms demonstrated in animal model: The first consists of the passage of gas under pressure from the lumen of the intestines to the portal venous circulation, the second consists of the passage of gas into the portal circulation by changes in intestinal mucosa, which could justify the passage of air to the portal circulation in cases of gastric ulcers [7]. A third theory would be the production of gas by bacteria associated or not with pileflebite [6,7].
HPVG occurs mainly in cases of intestinal ischemia but may also be due to sepsis, inflammatory bowel disease, diverticulitis, gastric dilatation, abdominal trauma, catheter insertion for jejunostomy, laparoscopic percutaneous gastrostomy, esophageal variceal ligation, pancreatitis acute [7]. Several other associated conditions have been described and, according to Abboud et al., any of these conditions can lead to intestinal hypo perfusion and subsequent ischemia [6]. Adynamic ileus would also play an important role, as it would facilitate bacterial proliferation and loosening of the loops. Increased intra-luminal pressure of the intestinal loops would allow the passage of gas and bacterial translocation to the portal system through lesions of the intestinal mucosa [6].
In the only case report of HPVG after endoscopic gastrostomy previously published, the author attributed the presence of air in the portal circulation to gastric mucosal lesion that associated with gastro paresis, ileus and distension of loops secondary to leakage of gastrostomy to peritoneal cavity allowed bacterial translocation to the portal system [7].
In our case, we did not show signs of gastrostomy leakage, intestinal ischemia, or even distensión of loops in exploratory laparotomy. We believe that the HPVG picture may be secondary to a set of factors and not only to the endoscopic procedure. The presence of multiple comorbidities, the state of splanchnic and mesenteric hypoperfusion associated with pulmonary and urinary infectious pictures, even in antibiotic therapy, would lead to the state of immunosuppression. The percutaneous gastrostomy and the gastric distensión resulting from the endoscopic procedure would have led to a portal bacterial translocation that would contribute to the worsening of the septic condition in a patient already debilitated by the previous conditions, determining the poor prognosis.
Even in the absence of signs of intestinal ischemia to the surgical procedure, it is probable that there was already relative intestinal ischemia. Since the Patient had evidence of macrovascular at herosclerotic disease in the carotid and coronary territories, it is probable that she already had mesenteric arteriopathy. The hypoperfusion profile secondary to infectious events could lead to non-obvious intestinal ischemia and surgical evaluation, since initially the ischemic lesión occurs at the mucosal level.
Abboud et al., emphasizes the importance of the clinical events underlying HPVG and the coexistence of chronic diseases that could lead to immunological dysfunction and changes in the intestinal microbiota [6]. In the past, the presence of HPVG was almost always associated with conditions of high mortality and in need of surgical treatment in the context of mesenteric ischemia [3]. However, the advancement of imaging studies has allowed the diagnosis of HPVG in increasingly early stages of intestinal ischemia and in conditions in which surgical treatment is not necessary [6].
It is evident that the presence of HPVG is related to serious conditions, which in spite of the necessity of the operative treatment must be known to the surgeon. The surgeon should keep in mind that the presence of air in the port system does not necessarily determine the poor prognosis. However, in the presence of HPVG, the underlying pathological conditions should be part of the diagnostic reasoning and clinical support is often the difference in the success of the treatment of these patients.
CONCLUSION
HPVG is a radiological signal that should always lead the surgeon to seek serious underlying pathological conditions in need of early intervention. The surgeon should indicate an operative procedure when this is necessary, especially when there is evidence of intestinal ischemia. However, the surgeon should know that the presence of HPVG alone does not indicate a worse prognosis. The careful evaluation of the underlying conditions Will díctate the best treatment, surgical or not.
DISCLOSURES AND ETHICS
The case report followed the ethical precepts, analyzed the data in a confidential way by the research team and the results will not allow the identification of study participants.
SOURCE OF FINANCING
The author(s) received no financial support for the research, authorship, and/or publication of this article.
CONFLICT OF INTERESTS
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
REFERENCES
- Thompson AM, Bizzarro MJ (2008) Necrotizing enterocolitis in newborns: Pathogenesis, prevention and management. Drugs 68: 1227-1238.
- Merritt CR, Goldsmith JP, Sharp MJ (1984) Sonographic detection of portal venous gas in infants with necrotizing enterocolitis. AJR Am J Roentgenol 143: 1059-1062.
- Liebman PR, Patten MT, Manny J, Benfeld JR, Hechtman HB (1978) Hepatic--portal venous gas in adults: Etiology, pathophysiology and clinical significance. Ann Surg 187: 281-287.
- Sellner F, Sobhian B, Baur M, Sellner S, Horvath B, et al. (2007) Intermittent hepatic portal vein gas complicating diverticulitis--a case report and literature review. Int J Colorectal Dis 22: 1395-1399.
- Muscari F, Suc B, Lagarrigue J (1999) [Hepatic portal venous gas: Is it always a sign of severity and surgical emergency?] Chirurgie 124: 69-72.
- Abboud B, Hachem JE, Yazbeck T, Doumit C (2009) Hepatic portal venous gas: Physiopathology, etiology, prognosis and treatment. World J Gastroenterol 15: 3585-3590.
- Peloponissios N, Halkic N, Pugnale M, Jornod P, Nordback P, et al. (2003) Hepatic Portal Gas in Adults: Review of the Literature and Presentation of a Consecutive Series of 11 Cases. Arch Surg 138: 1367-1370.
Citation: Souza LCA, Silva CHW, Claros OR, Silva RFA (2019) Hepatic Portal Venous Gas in a Patient Submittedto Percutaneous Gastrostomy: Case Report and Literature Review. J Surg Curr Trend Innov 3: 010.
Copyright: © 2019 Luiz Carlos de Araújo Souza, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

