
Journal of Neonatology & Clinical Pediatrics Category: Clinical
Type: Review Article
Immunological Aspects of Pregnancy: A Literature Review
*Corresponding Author(s):
Ana Karina Marques SalgeSchool Of Nursing School, Goiás Federal University, UFG Rua 227 Qd 68, S/N - Setor Leste Universitário, CEP: 74605-080, Goiânia, Goiás, Brazil
Tel:+55 (62) 32096280,
Fax:+55 (62) 32096282
Email:anasalge@gmail.com
Received Date: Aug 24, 2016
Accepted Date: Mar 06, 2017
Published Date: Mar 21, 2017
Abstract
Pregnancy is a unique phenomenon for the human body’s immune system and makes a single natural model. The maternal immune system is active and functional during pregnancy. The immunological relationship between the mother and the fetus is a bi-directional communication determined on the one hand by fetal antigen presentation and on the other hand by recognition of and reaction to these antigens by the maternal immune system. Two main theories have been proposed to explain the phenomenon of pregnancy. One theory emphasizes the role of immune suppressive reactions to protect the fetus. A second one, the “immune tropism theory”, works on the importance of the maternal immune response to paternal antigens present in the conceptus. Today it is known the maternal immune system recognizes the embryo without triggering a response against its presence and development in the intrauterine environment. Several mechanisms allow for implantation of the embryo/fetus and pregnancy progresses through a specific immunoregulatory response, known as immune tolerance. Its major events include decreased activity of natural killer cells, expression of regulatory proteins of the complement system, and progesterone action - all acting to maintain a suitable intrauterine environment and promote fetal recognition. Inappropriate immune responses can endanger a pregnancy and may be clinically reflected by repeat abortions and restricted intrauterine growth. Although there have been proposed several theories to explain the different immunological pathways involved in this process, knowledge on major immunological changes occurring during pregnancy is key for guiding clinical practice and maintaining a healthy pregnancy for both the mother-to-be and her fetus.
Keywords
Immunology; Placenta; Pregnancy
INTRODUCTION
IMMUNOLOGICAL ASPECTS OF PREGNANCY
Pregnancy is a unique phenomenon for the human body’s immune system and makes a single natural model. It comprises a highly complex immune state with frequent exacerbation of conditions or preexisting abnormalities [1].
The immunological relationship between the mother and the fetus is a bi-directional communication determined on the one hand by fetal antigen presentation and on the other hand by recognition of and reaction to these antigens by the maternal immune system [2].
There is evidence now that immunological recognition of pregnancy is important for the maintenance of gestation, and that inadequate recognition of fetal antigens might result in failed pregnancy [3].
Two main theories have been proposed to explain the phenomenon of pregnancy. One theory emphasizes the role of immunosuppressive reactions to protect the fetus. A second one, the “immune tropism theory”, works on the importance of the maternal immune response to paternal antigens present in the conceptus [4].
In 1953 it was formulated the hypothesis that the embryo can be considered as a semi-allogeneic graft and thus is vulnerable to rejection and immune tolerance. The mechanisms of adaptive immune response during pregnancy that allows successful placental graft have been investigated since then though not yet fully understood [1].
Today it is known the maternal immune system recognizes the embryo without triggering a response against its presence and development in the intrauterine environment. However, understanding the immunological mechanisms involved in the non-rejection of the placenta by the maternal immune system has been challenging. The placenta, for its embryonic origin, has both maternal and paternal genetic material and can potentially express paternal antigens, which are genetically foreign to the mother and therefore could induce an immune reaction that leads to rejection in a manner similar to that of other grafted tissues [2].
Similar to a neo plastic growth, early pregnancy is strongly characterized by intense cell proliferation (embryo and placenta ion). However, during pregnancy, the maternal immune system does not set of factions to block this proliferation, as would be expected in pathological conditions such as in tumors. There is an environment that favors conceptus development to a state of maturity where the fetus is capable of supporting extra uterine life [5].
This phenomenon, so-called “immune tolerance”, is initiated with the implantation of the conceptus. The main tissue exposed to the maternal immune system consists of trophoblasts, and the maternal-embryonic interface is created. In addition to a differentiated expression of HLA molecules that can identify what is “self” and “non-self” in the human body, these cells have a unique potential of response to local cytokines [1].
A pregnancy-specific immune status can be evidenced by the fact that certain diseases of autoimmune origin may aggravate or improve during pregnancy depending on the immune response pattern of the underlying disease. Thus, autoimmune diseases typically due to exacerbated inflammatory responses improve with gestational homeostasis while those characterized by the action of auto antibodies are significantly aggravated [1].
Like in any other inflammatory immune processes, during pregnancy, the uteroplacental unit initiates and modulates a harmonic interaction between the maternal vascular endothelium, local immune competent cells, and antigenic determinants on the surface of trophoblasts to actively regulate cell adhesion, activation, and migration through changes in local cytokines [1].
As pregnancy progresses anew vascular system that can efficiently provide oxygen and nutrients to the embryo develops. This “new” vascular system (placentation) is created when the trophoblasts cells invade the endometrial wall and reach uterine spiral arteries. Cytotrophoblast cells penetrate into the decidua and destroy the muscular layer of these vessels transforming them into high-capacitance/low resistance vessels. During the process of vascular invasion, the cytotrophoblasts differentiate from an originally epithelial phenotype to an endothelial phenotype covering the endovascular layer of spiral arteries [6].
Placentation allows the uterine artery to grow as pregnancy progresses, preventing shortage of blood flow to the fetal-placental unit. Fetal gene products as well as specific differentiation antigens are exposed locally to maternal immune competent cells [7].
In early stages of implantation of the ovum, when the human body acknowledges the presence of a “semi-allogeneic graft” - the trophoblast, an immune inflammatory response is set up, similar to that in the early of graft rejection. Mononuclear cell columns derived from trophoblasts are formed and invade the uterine decidua and its blood vessels to provide anchorage and blood supply to the developing fetus. Thus, trophoblast antigen presenting at the maternal-fetal interface seems to be an important component of the maternal immune response during pregnancy [1,4].
The immunological relationship between the mother and the fetus is a bi-directional communication determined on the one hand by fetal antigen presentation and on the other hand by recognition of and reaction to these antigens by the maternal immune system [2].
There is evidence now that immunological recognition of pregnancy is important for the maintenance of gestation, and that inadequate recognition of fetal antigens might result in failed pregnancy [3].
Two main theories have been proposed to explain the phenomenon of pregnancy. One theory emphasizes the role of immunosuppressive reactions to protect the fetus. A second one, the “immune tropism theory”, works on the importance of the maternal immune response to paternal antigens present in the conceptus [4].
In 1953 it was formulated the hypothesis that the embryo can be considered as a semi-allogeneic graft and thus is vulnerable to rejection and immune tolerance. The mechanisms of adaptive immune response during pregnancy that allows successful placental graft have been investigated since then though not yet fully understood [1].
Today it is known the maternal immune system recognizes the embryo without triggering a response against its presence and development in the intrauterine environment. However, understanding the immunological mechanisms involved in the non-rejection of the placenta by the maternal immune system has been challenging. The placenta, for its embryonic origin, has both maternal and paternal genetic material and can potentially express paternal antigens, which are genetically foreign to the mother and therefore could induce an immune reaction that leads to rejection in a manner similar to that of other grafted tissues [2].
Similar to a neo plastic growth, early pregnancy is strongly characterized by intense cell proliferation (embryo and placenta ion). However, during pregnancy, the maternal immune system does not set of factions to block this proliferation, as would be expected in pathological conditions such as in tumors. There is an environment that favors conceptus development to a state of maturity where the fetus is capable of supporting extra uterine life [5].
This phenomenon, so-called “immune tolerance”, is initiated with the implantation of the conceptus. The main tissue exposed to the maternal immune system consists of trophoblasts, and the maternal-embryonic interface is created. In addition to a differentiated expression of HLA molecules that can identify what is “self” and “non-self” in the human body, these cells have a unique potential of response to local cytokines [1].
A pregnancy-specific immune status can be evidenced by the fact that certain diseases of autoimmune origin may aggravate or improve during pregnancy depending on the immune response pattern of the underlying disease. Thus, autoimmune diseases typically due to exacerbated inflammatory responses improve with gestational homeostasis while those characterized by the action of auto antibodies are significantly aggravated [1].
Like in any other inflammatory immune processes, during pregnancy, the uteroplacental unit initiates and modulates a harmonic interaction between the maternal vascular endothelium, local immune competent cells, and antigenic determinants on the surface of trophoblasts to actively regulate cell adhesion, activation, and migration through changes in local cytokines [1].
As pregnancy progresses anew vascular system that can efficiently provide oxygen and nutrients to the embryo develops. This “new” vascular system (placentation) is created when the trophoblasts cells invade the endometrial wall and reach uterine spiral arteries. Cytotrophoblast cells penetrate into the decidua and destroy the muscular layer of these vessels transforming them into high-capacitance/low resistance vessels. During the process of vascular invasion, the cytotrophoblasts differentiate from an originally epithelial phenotype to an endothelial phenotype covering the endovascular layer of spiral arteries [6].
Placentation allows the uterine artery to grow as pregnancy progresses, preventing shortage of blood flow to the fetal-placental unit. Fetal gene products as well as specific differentiation antigens are exposed locally to maternal immune competent cells [7].
In early stages of implantation of the ovum, when the human body acknowledges the presence of a “semi-allogeneic graft” - the trophoblast, an immune inflammatory response is set up, similar to that in the early of graft rejection. Mononuclear cell columns derived from trophoblasts are formed and invade the uterine decidua and its blood vessels to provide anchorage and blood supply to the developing fetus. Thus, trophoblast antigen presenting at the maternal-fetal interface seems to be an important component of the maternal immune response during pregnancy [1,4].
FACTORS INVOLVED WITH THE IMMUNE RESPONSE DURING PREGNANCY
Maternal immune response is biased toward humoral immunity and away from cell-mediated immunity that could be harmful to the fetus. Cytokines of maternal origin act on placental development. Antigen expression on the placenta determines maternal cytokine pattern [3].
Several genes are likely regulators of human implantation including Decay Accelerating Factor (DAF), Indoleamine 2,3 Dioxygenase (IDO), IL-15, IL-15Rα, Interferon Regulatory Factor-1 (IRF-1), lymphotoxin, Natural Killer (NK) Associated Transcript2 (NKAT2) and NKG5. The functions of these genes are quite diverse including promotion of NK cell proliferation, chemotaxis, inhibition of NK cell cytolytic activity, inhibition of cell growth (T-cells and pathogens), and inhibition of the classical complement pathway. These diverse functions are likely designed to prepare the endometrium for implantation and particularly to adapt the maternal immune system so that it can accommodate the fetus, an immunological “stranger” [8]. Normal human pregnancy is characterized by low peripheral NK activity, and increased NK activity seems to play a role in spontaneous abortions of unknown etiology. The dynamics of the appearance of uterine NK cells suggest that one of the functions of these cells is control of placentation [3].
Ovulation, mating, and fertilization produce inflammatory immune changes at the mucosal surfaces of the female reproductive tract with direct effects on the blastocyst implantation in the uterus. The very exposure to sperm and its elements produces an immunosuppressive effect on the mucosal surfaces of the female reproductive tract [5].
Steroid hormones produced during ovulation also contribute to this effect. They modulate the activity of Antigen-Presenting Cells (APC) - originated from both myeloid and lymphoid lineage - that are directly involved with the immune responses [9,10].
Progesterone is the female hormone that has the most distinctive activity on the maternal immune system and maternal-fetal interface. Progesterone can suppress the effectors function of T cells and modulate calcium ion and potassium channels of cell membranes with a direct effect on gene expression in these cells [11].
The most important immunoregulatory effect of progesterone is probably its action on T lymphocytes that possess gamma/delta (γδ) T Cell Receptors (TCR). Following allogeneic stimulation by fatally derived antigens; this subset of pregnancy T lymphocytes develops progesterone receptors. At high progesterone concentrations, these lymphocytes produce an immunomodulatory protein, Progesterone-Induced Blocking Factor (PIBF), a mediator that, through changes in cytokine balance, inhibits NK activity and exerts an antiabortive effect in mice [3,12].
It is thus possible that, under certain circumstances, the semi-allogeneic fetus is not able to induce progesterone receptor expression in γδ T cells so that progesterone supplementation is not strong enough to elicit an appropriate alloimmune response and to maintain pregnancy [10]. A successful pregnancy requires the maternal immune system to recognize and tolerate the semiallogeneic fetus, and allow for normal invasion of trophoblasts. Pregnancy complications are considered to be associated with dysfunctional maternal-fetal crosstalk [13].
Syncytiotrophoblast cells are responsible for nutrition of the embryo and production of ?-HCG and progesterone. These cells are originated from the cytotrophoblasts and recover almost all fetal surfaces and do not express HLA molecules. On the other hand, villous, extra villous, endovascular, and interstitial cytotrophoblasts cells express small amounts of HLA class I molecules. Extra villous cytotrophoblast cells express a specific HLA class I molecule, so-called HLA-G. HLA-Gexpressionis restricted to placental andthymic epithelial cells [10].
Decidual leukocytes are composed of a prominent cell population including NK cells, macrophages and T cells. These cells are responsible for alerting the maternal immune system for the presence of alloantigens throughout the entire pregnancy [5].
The decidua and its components function as a specialized immune tissue with several different functions including an essential role in implantation and maintenance of pregnancy. Decidual leukocytes may act directly and indirectly by expressing receptors that potentially mediate the recognition of fetal trophoblasts, trophoblastic invasion, and production of cytokines that regulate and modulate the maternal immune response and vascular function [7,8].
An orchestrated evolutionary maternal adaption toward tolerance of the semiallogeneic fetus is required to ensure decidualization and early embryo development [14]. The mother does not reject the embryo as it develops in an isolated semi permeable environment. Thus, the maternal immune response is modulated and recognizes the fetus, which allows for the development of a successful pregnancy (Figure 1).
Several genes are likely regulators of human implantation including Decay Accelerating Factor (DAF), Indoleamine 2,3 Dioxygenase (IDO), IL-15, IL-15Rα, Interferon Regulatory Factor-1 (IRF-1), lymphotoxin, Natural Killer (NK) Associated Transcript2 (NKAT2) and NKG5. The functions of these genes are quite diverse including promotion of NK cell proliferation, chemotaxis, inhibition of NK cell cytolytic activity, inhibition of cell growth (T-cells and pathogens), and inhibition of the classical complement pathway. These diverse functions are likely designed to prepare the endometrium for implantation and particularly to adapt the maternal immune system so that it can accommodate the fetus, an immunological “stranger” [8]. Normal human pregnancy is characterized by low peripheral NK activity, and increased NK activity seems to play a role in spontaneous abortions of unknown etiology. The dynamics of the appearance of uterine NK cells suggest that one of the functions of these cells is control of placentation [3].
Ovulation, mating, and fertilization produce inflammatory immune changes at the mucosal surfaces of the female reproductive tract with direct effects on the blastocyst implantation in the uterus. The very exposure to sperm and its elements produces an immunosuppressive effect on the mucosal surfaces of the female reproductive tract [5].
Steroid hormones produced during ovulation also contribute to this effect. They modulate the activity of Antigen-Presenting Cells (APC) - originated from both myeloid and lymphoid lineage - that are directly involved with the immune responses [9,10].
Progesterone is the female hormone that has the most distinctive activity on the maternal immune system and maternal-fetal interface. Progesterone can suppress the effectors function of T cells and modulate calcium ion and potassium channels of cell membranes with a direct effect on gene expression in these cells [11].
The most important immunoregulatory effect of progesterone is probably its action on T lymphocytes that possess gamma/delta (γδ) T Cell Receptors (TCR). Following allogeneic stimulation by fatally derived antigens; this subset of pregnancy T lymphocytes develops progesterone receptors. At high progesterone concentrations, these lymphocytes produce an immunomodulatory protein, Progesterone-Induced Blocking Factor (PIBF), a mediator that, through changes in cytokine balance, inhibits NK activity and exerts an antiabortive effect in mice [3,12].
It is thus possible that, under certain circumstances, the semi-allogeneic fetus is not able to induce progesterone receptor expression in γδ T cells so that progesterone supplementation is not strong enough to elicit an appropriate alloimmune response and to maintain pregnancy [10]. A successful pregnancy requires the maternal immune system to recognize and tolerate the semiallogeneic fetus, and allow for normal invasion of trophoblasts. Pregnancy complications are considered to be associated with dysfunctional maternal-fetal crosstalk [13].
Syncytiotrophoblast cells are responsible for nutrition of the embryo and production of ?-HCG and progesterone. These cells are originated from the cytotrophoblasts and recover almost all fetal surfaces and do not express HLA molecules. On the other hand, villous, extra villous, endovascular, and interstitial cytotrophoblasts cells express small amounts of HLA class I molecules. Extra villous cytotrophoblast cells express a specific HLA class I molecule, so-called HLA-G. HLA-Gexpressionis restricted to placental andthymic epithelial cells [10].
Decidual leukocytes are composed of a prominent cell population including NK cells, macrophages and T cells. These cells are responsible for alerting the maternal immune system for the presence of alloantigens throughout the entire pregnancy [5].
The decidua and its components function as a specialized immune tissue with several different functions including an essential role in implantation and maintenance of pregnancy. Decidual leukocytes may act directly and indirectly by expressing receptors that potentially mediate the recognition of fetal trophoblasts, trophoblastic invasion, and production of cytokines that regulate and modulate the maternal immune response and vascular function [7,8].
An orchestrated evolutionary maternal adaption toward tolerance of the semiallogeneic fetus is required to ensure decidualization and early embryo development [14]. The mother does not reject the embryo as it develops in an isolated semi permeable environment. Thus, the maternal immune response is modulated and recognizes the fetus, which allows for the development of a successful pregnancy (Figure 1).
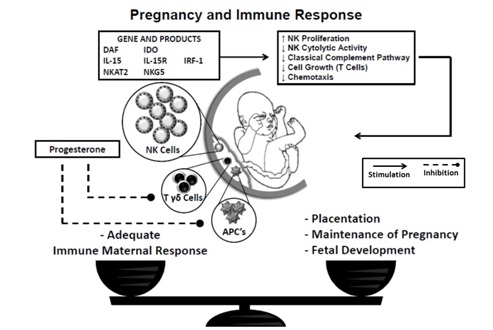
Figure 1: Interaction between maternal and fetal immune response. The placentation, maintenance of pregnancy and fetal development need an adequate immune maternal response.
EPIGENETIC CONTROL OF IMMUNE RESPONSE IN PREGNANCY
A major question in the biology of stress and environmental adaptation concerns the neurobiological basis of how neuroendocrine systems governing physiological regulatory mechanisms essential for life (metabolism, immune response, organ function) become harmful [15]. Maternal nutrition has a considerable impact on fetal metabolic programming and environmental adaptation. Specifically, high-fat diets, micro- and macronutrient deficiencies, and low-protein diet intake during conception through lactation are all deleterious to fetal and infant development [16]. The deleterious effects involve alterations in gene expression and epigenetic programming that converge to induce inflammation, impair the immune system, and cause a series of pathologic conditions at both early and late stages of life. Maternal nutrient intake, maternal infection, obesity, environmental pollution and toxicants, and psychological stress may all influence immune function and inflammation in utero [16].
FINAL CONSIDERATIONS
The maternal immune system is active and functional during pregnancy. Several mechanisms allow for implantation of the embryo/fetus and pregnancy progresses through a specific immunoregulatory response, known as immune tolerance. Its major events include decreased activity of NK cells, expression of regulatory proteins of the complement system, and progesterone action - all acting to maintain a suitable intrauterine environment and promote fetal recognition. Inappropriate immune responses can endanger a pregnancy and may be clinically reflected by repeat abortions and restricted intrauterine growth. Although there have been proposed several theories to explain the different immunological pathways involved in this process, knowledge on major immunological changes occurring during pregnancy is key for guiding clinical practice and maintaining a healthy pregnancy for both the mother-to-be and her fetus.
REFERENCES
- Medawar PB (1953) Some immunological and endocrinological problems raised by evolution of viviparity in vertebrates. Symp Soc Exp Biol 7: 320-328.
- Kumpe BM, Manoussaka MS (2012) Placental immunology and maternal alloimmune responses. Vox Sang 102: 2-12.
- Szekeres-Bartho J (2002) Immunological relationship between the mother and the fetus. Int Rev Immunol 21: 471-495.
- Zhou Y, Fisher SJ, Janatpour M, Genbacev O, Dejana E, et al. (1997) Human cytotrophoblasts adopt a vascular phenotype as they differentiate. A strategy for successful endovascular invasion? J Clin Invest 99: 2139-2151.
- Olding LB, Papadogiannakis N, Barbieri B, Murgita RA (1997) Suppressive cellular and molecular activities in materno fetal immune interactions; supressor cell activity, prostaglandins, and alphafetoproteins. Curr Top Mirobiol Immunol 222: 159-187.
- Cross JC, Werb Z, Fisher SJ (1994) Implantation and the placenta: key pieces of the development puzzle. Science 266: 1508-1518.
- Lobo SC, Huang ST, Germeyer A, Dosiou C, Vo KC, et al. (2004) The immune environment in human endometrium during the window of implantation. Am J Reprod Immunol 52: 244-251.
- Moffett-King A (2002) Natural killer cells and pregnancy. Nat Rev Immunol 2: 656-663.
- Prabhala RH, Wira CR (1995) Sex hormone and IL-6 regulation of antigen presentation in the female reproductive tract mucosal tissues. J Immunol 155: 5566-5573.
- Ehring GR, Kerschbaum HH, Eder C, Neben AL, Fanger CM, et al. (1998) A nongenomic mechanism for progesterone-mediated immunes supression: inhibition of K+ channels, Ca+2 signaling, and gene expression in T lymphocytes. J Exped Med 188: 1593-1602.
- Szekeres-Bartho J, Polgar B, Kozma N, Miko E, Par G, et al. (2005) Progesterone-dependent immunomodulation. Chem Immunol Allergy 89: 118-125.
- Check JH, Arwitz M, Gross J, Peymer M, Szekeres-Bartho J (1997) Lymphocyte immunotherapy (LI) increases serum levels of progesterone induced blocking factor (PIBF). Am J Reprod Immunol 37: 17-20.
- Xu YY, Wang SC, Li DJ, Du MR (2017) Co-Signaling Molecules in Maternal-Fetal Immunity. Trends Mol Med 23: 46-58.
- Fu B, Wei H (2016) Decidual natural killer cells and the immune microenvironment at the maternal-fetal interface. Sci China Life Sci 59: 1224-1231.
- Claycombe KJ, Brissette CA, Ghribi O (2015) Epigenetics of inflammation, maternal infection, and nutrition. J Nutr 145: 1109-1115.
- Weaver IC (2009) Shaping adult phenotypes through early life environments. Birth Defects Res C Embryo Today 87: 314-326.
Citation: Salge AKM, Siqueira KM, Castral TC, Guimarães JV, Rocha AA, et al. (2017) Immunological aspects of pregnancy: a literature review. J Neonatol Clin Pediatr 4: 016.
Copyright: © 2017 Ana Karina Marques Salge, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
© 2026, Copyrights Herald Scholarly Open Access. All Rights Reserved!

