
Management of Hyperhidrosis: A Proposal Protocol for Trainees
*Corresponding Author(s):
Ashok HandaNuffield Department Of Surgical Sciences, University Of Oxford, Oxford, United Kingdom
Tel:+44 1865231067,
Email:ashok.handa@nds.ox.ac.uk
Abstract
Hyperhidrosis is a chronic condition characterised by excessive sweating [1], and can effect up to 3% of the population [2]. It can be classed as generalised; involving all the body’s sweat glands, or localized; affecting specific areas of the body only e.g., palms, soles of feet, groin [3].
Keywords
INTRODUCTION
The condition is seriously debilitating due to both emotional and social embarrassment. Some patients may also endure physical or occupational disability, hindering their activities of daily living and negatively impacting on their employability [4]. With this in mind it is important to treat patients with successful therapies and ensure adequate follow up is sought after the procedure.
METHODS
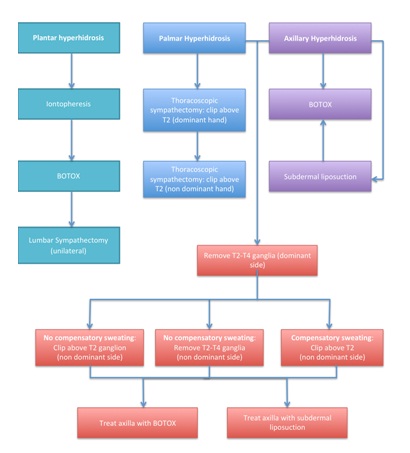 Figure 1: Management of Hyperhidrosis.
Figure 1: Management of Hyperhidrosis.RESULTS
Plantar hyperhidrosis
Palmar hyperhidrosis
The pitfalls to this procedure are;
1) Failure; due to clipping the wrong ganglion, or to reoccurrence,
2) Compensatory sweating, particularly around the abdomen [13].
In the case of unilateral sympathectomy, compensatory sweating is rarely a problem, however if a patient requests a sympathectomy on the contralateral side, compensatory sweating of the back, abdomen and feet should be discussed prior to procedure.
Axillary hyperhidrosis
We have treated a total of 168 patients from 2007-2015, on a self referral basis. Some patients require injections every 4 months, whilst others only re-present after 2 years. General patient satisfaction is high and outcomes are reputable.
The second pathway involves referral to a plastic surgeon for subdermal liposuction as primary therapy. Although this treatment has a risk of seroma formation [16], it can be extremely effective in the long term and the seromas tend to settle with time. If this is unsuccessful, botulinum toxin injections could be considered.
Palmar and axillary hyperhidrosis
Our unit has demonstrated reliable results following removal of the T2-T4 ganglia; however studies have shown similar long term outcomes following T3-T4 sympathectomy, and possible reduction in compensatory sweating [19].
If the patient wishes to have further treatment, there are several options available:
1) If no compensatory sweating has occurred, the T2-T4 (or T3-T4) ganglion can be removed from the contralateral side. An alternative option would be to clip above the T2 ganglion only on the contralateral side and treat the axilla with either botulinum toxin or subdermal liposuction, reducing the risk of compensatory sweating.
2) If compensatory sweating has occurred following treatment of the dominant side, the second option of clipping above the T2 ganglion and treating the axilla separately is advised.
If however the patient fails to have reduced axillary sweating after treatment, they can either be treated with botulinum toxin or subdermal liposuction bilaterally.
Our unit operates on approximately 6-8 patients per year for palmar and axillary hyperhidrosis. All have had adequate outcomes post procedure, with around 60% requesting further sympathectomy on their non dominant side.
ALTERNATIVE TREATMENTS
Anticholinergic medications can be used either topically or orally. Multiple studies have evaluated their outcomes, and although their efficacy (topically and orally) is in the range of 60-75% [22], the side effects are marked and they are contraindicated in a wide range of patients [23].
CONCLUSION
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
The authors declare no conflict of interest in preparing this article.
REFERENCES
- Lakraj AA, Moghimi N, Jabbari B (2013) Hyperhidrosis: anatomy, pathophysiology and treatment with emphasis on the role of botulinum toxins. Toxins (Basel) 5: 821-840.
- Strutton DR, Kowalski JW, Glaser DA, Stang PE (2004) US prevalence of hyperhidrosis and impact on individuals with axillary hyperhidrosis: results from a national survey. J Am Acad Dermatol 51: 241-248.
- Stashak AB, Brewer JD (2014) Management of hyperhidrosis. Clin Cosmet Investig Dermatol 7: 285-299.
- Naumann MK, Hamm H, Lowe NJ, Botox Hyperhidrosis Clinical Study Group (2002) Effect of botulinum toxin type A on quality of life measures in patients with excessive axillary sweating: a randomized controlled trial. Br J Dermatol 147: 1218-1226.
- Daghfous M, Zghal M, Zghal A, Cherif F, Kamoun MR (1993) [Treatment of palmar and plantar hyperhidrosis by ++ionophoresis]. Tunis Med 71: 387-389.
- McAleer MA, Collins P (2014) A study investigating patients’ experience of hospital and home iontophoresis for hyperhidrosis. J Dermatolog Treat 25: 342-344.
- Dressler D, Adib Saberi F (2013) Towards a dose optimisation of botulinum toxin therapy for axillary hyperhidrosis: comparison of different Botox(®) doses. J Neural Transm 120: 1565-1567.
- Naumann M, Lowe NJ (2001) Botulinum toxin type A in treatment of bilateral primary axillary hyperhidrosis: randomised, parallel group, double blind, placebo controlled trial. BMJ 323: 596-599.
- Takats G, Helfrich L (1941) Sterility of the males after sympathectomies. JAMA 117: 20-21.
- Loureiro Mde P, de Campos JR, Kauffman P, Jatene FB, Weigmann S, et al. (2008) Endoscopic lumbar sympathectomy for women: effect on compensatory sweat. Clinics (Sao Paulo) 63: 189-196.
- Solish N, Bertucci V, Dansereau A, Hong HC, Lynde C, et al. (2007) A comprehensive approach to the recognition, diagnosis, and severity-based treatment of focal hyperhidrosis: recommendations of the Canadian Hyperhidrosis Advisory Committee. Dermatol Surg 33: 908-923.
- Fox AD, Hands L, Collin J (1999) The results of thoracoscopic sympathetic trunk transection for palmar hyperhidrosis and sympathetic ganglionectomy for axillary hyperhidrosis. Eur J Vasc Endovasc Surg 17: 343-346.
- Licht PB, Pilegaard HK (2004) Severity of compensatory sweating after thoracoscopic sympathectomy. Ann Thorac Surg 78: 427-431.
- Scamoni S, Valdatta L, Frigo C, Maggiulli F, Cherubino M (2012) Treatment of Primary Axillary Hyperhidrosis with Botulinum Toxin Type A: Our Experience in 50 Patients from 2007 to 2010. J ISRN Dermatol.
- Naumann M, Lowe NJ, Kumar CR, Hamm H, Hyperhidrosis Clinical Investigators Group (2003) Botulinum toxin type A is a safe and effective treatment for axillary hyperhidrosis over 16 months: a prospective study. Arch Dermatol 139: 731-736.
- Dixit VV, Wagh MS (2013) Unfavourable outcomes of liposuction and their management. Indian J Plast Surg 46: 377-392.
- Smidfelt K, Drott C (2011) Late results of endoscopic thoracic sympathectomy for hyperhidrosis and facial blushing. Br J Surg 98: 1719-1724.
- Glaser DA, Hebert AA, Pariser DM, Solish N (2007) Palmar and plantar hyperhidrosis: best practice recommendations and special considerations. Cutis 79: 18-28.
- Scognamillo F, Serventi F, Attene F, Torre C, Paliogiannis P, et al. (2011) T2-T4 sympathectomy versus T3-T4 sympathicotomy for palmar and axillary hyperhidrosis. Clin Auton Res 21: 97-102.
- Stolman LP (2003) Treatment of hyperhidrosis. J Drugs Dermatol 2: 521-527.
- Rayner CR, Ritchie ID, Stark GP (1980) Axillary hyperhidrosis, 20% aluminum chloride hexahydrate, and surgery. Br Med J 280: 1168.
- Wolosker N, de Campos JR, Kauffman P, Yazbek G, Neves S, et al. (2013) Use of oxybutynin for treating plantar hyperhidrosis. Int J Dermatol 52: 620-623.
- Wolosker N, de Campos JR, Kauffman P, Puech-Leão P (2012) A randomized placebo-controlled trial of oxybutynin for the initial treatment of palmar and axillary hyperhidrosis. J Vasc Surg 55: 1696-1700.
Citation: Hurst KV, Handa A, Gordon A (2015) Management of Hyperhidrosis: A Proposal Protocol for Trainees. J Angiol Vasc Surg 1: 001.
Copyright: © 2015 Katherine V Hurst, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

