Morphofunctional Changes of Blood Supply and the Glycogen Content in the Liver during Acute Strangulational Small Bowel Obstruction
*Corresponding Author(s):
Vladimir Efimovich MilyukovFederal State Autonomous Educational Institution Of Higher Education Im Sechenov, First Moscow State Medical University Of The Ministry Of Health Of The Russian Federation (Sechenov University), Moscow, Russian Federation
Tel:+7 9161564127,
Email:Milyucov@mail.ru
Abstract
Summary: Changes in the morphofunctional organization of the liver in acute small bowel obstruction, leading to the development of acute hepatic insufficiency, have not been adequately studied. The aim of the work is to identify the patterns of the dynamics of changes in the morphofunctional organization of the liver vasculary bed and their relationship to changes in the energy metabolism level of the liver at different times of acute strangulation small bowel obstruction development.
Materials and Methods: The study was carried out on 23 adult mongrel dogs of both sexes weighing 17 - 20 kg. The study of the normal liver morphology was performed on 3(three) dogs, which served as the first control group. Modelling of strangulational bowel obstruction was performed on the animals of the second group (12 animals) with excision of liver pieces for observation periods of 3, 6, 12 and 24 hours.
The third control group was consisted of 8 animals, whose liver morphology was studied after laparotomy without intervention on the vessels and organs of the abdominal cavity. The sampling of the material for the study was carried out over 3, 6, 12 and 24 hours following the laparotomy. Comparison of the study results in the second and third groups allows revealing changes in the liver caused by acute small bowel obstruction, but not the consequences of laparotomy when modeling the disease.
A morphometric method for estimating the change in the volume of liver’s vascular bed was used for departments (arterial part of the liver’s Hemomicrocirculatory Bed (HMCB), capillary and venous part), the samples were stained with hematoxylin and eosin. To assess the change in the energy metabolism level, a method was used to quantify the glycogen content by the results of the Schiff (PAS) reaction.
Results: We revealed patterns of changes in the morphofunctional organization of liver’s vascular bed and the correlation between changes in the area (and consequently, the volume) of the vascular bed and changes in the energy consumption level in the dynamics of development of acute strangulational small bowel obstruction.
Conclusion: The changes in the liver hemocirculatory bed revealed by us are characterized by the development of compensatory vasodilatation of the arterial and venular link of the HMCB with a decrease in the glycogen level at the initial time (3 hours) and at the end of the experiment (24 hours). This is due, in our opinion, to an increase in the venous blood supply through the portal vein saturated with toxic products, which requires an increase in energy consumption for the performance of detoxifying function. With a decrease in the area of interlobular veins and a decrease in toxic effect, the glycogen level was increasing. The arterial blood supply remained elevated at all times of the experiment, due to the high demand of the liver for oxygen. We have revealed a strong inverse correlation between the change in the vascular bed area and the energy metabolism level.
Keywords
INTRODUCTION
Acute intestinal obstruction incidence is 1.2 - 14.2% of cases of all acute surgical diseases of the abdominal cavity [1-4]. In case of ASBO complicated with peritonitis, the mortality is 25 - 70%. In the terminal stage of peritonitis, with the development of Toxic Septic Shock (TSS) and syndrome of multiple organ failure, the mortality reaches 100% [3,5,6]. It is known that the liver is one of the main organs of life support in the human body [7,8]. The most important function of the liver is detoxifying, which is also called barrier [9-11]. The liver is the main organ in which poisons and toxins as a result of metabolic processes are continuously metabolized [9,10]. Specialized liver macrophages, stellate endotheliocytes (reticuloendoteliocyti stellatum) – “stellate Kupffer cells”, cells of the reticuloendothelial system, are located on the inner surface of the sinusoids of the liver. These cells are involved in phagocytosis and protect the liver cells from many toxic effects. Due to their numerous functions, they are a unique barrier that performs the protectional function not only from bacteria and toxic agents, but also from a number of other factors, including immunological [8].
In the case of ASBO, the liver appears to be the first target organ, which takes the main impact of toxemia in the form of a massive supply of blood with biologically active substances and toxic products resulting from ischemia and/or tissue necrosis of the pathologically altered small intestine through portal vein [4,12]. Toxins exert a direct damaging effect on liver cells, damage the membrane structures of hepatocytes and disrupt the transmembrane transport of hydrogen and other substrates, which in turn leads to discoordination of metabolic processes and subsequently with the progression of the disease to cell death. The result of this process, according to the literature, is the hepatic insufficiency development of varying severity [10,13,14]. There are quite a few studies of the morphofunctional organization of the liver in a healthy person. However, in the literature published over the last decade, unfortunately, there are unjustifiably few studies devoted to morphofunctional changes in the liver in acute small bowel obstruction.
Due to the high mortality among people of young and working age and the insufficient effectiveness of the existing methods of treating ASBO, we found it necessary to conduct an experimental morphological analysis of the nature and scale of changes in the hepatic lobules in the dynamics of development of ASBO [15].
In our study, we used a morphometric method to assess the change in the volume of liver vascular bed (the arterial part of liver hemomicrocirculatory bed, the capillary and venous part) on samples stained with hematoxylin and eosin.
The most important chemical compounds that ensure the vital activity of the human body are carbohydrates [16,17]. Their role is extremely important in providing the energy component of intracellular metabolism, providing it up to 60% [16]. The liver plays a central role in numerous carbohydrate metabolism reactions, it is the organ that maintains a normal blood glucose level due to the processes of glycogenesis - the conversion of glucose to glycogen by means of the hormone insulin. A characteristic feature of hepatocytes is their ability to store and dispose of glycogen as the major metabolite of energy [17]. Depot of glycogen in the liver can reach 1/5 of body weight. With the development of hypoxic conditions, intensive mobilization of carbohydrates from the liver takes place to compensate for the energy needs of the organism [18]. We studied the level of glycogen in the liver at different stages of the strangulational ASBO.
Thus, the purpose of our work was to identify the patterns of changes in the morphofunctional organization of the vascular bed of the liver and their relationship to changes in the level of energy metabolism in the liver at different stages of development of acute strangulational small intestine obstruction. Studying these changes in the liver with acute strangulational small bowel obstruction on the clinical material, performing a biopsy in the dynamics of the pathological process, is not possible. In this regard, the basis of our work was the experimental modeling of this disease.
MATERIALS AND METHODS
The subject of our work were dogs. When choosing the subject for our study we were guided by the fact that the anatomy, physiology and morphological structure of the liver of the dog and human are the closest to each other [19-21]. Structure and functions of the digestive tract, the main sources of blood circulation and blood outflow, the structure of the arterial and venous bed and consequently, the hemomicrocirculatory bed of the gastrointestinal tract in dogs and humans are similar [19]. A sufficiently high position in the phylogenetic raw of the dog and human determines the uniformity of the course of reactions.
The study was performed on 23 adult mongrel dogs of both sexes weighing 17 - 20 kg. On 3(three) dogs which served as first control group, we performed the study of normal liver morphology. On animals of the second group (12 animals), modeling of strangulational small bowel obstruction was performed. Simulation of the disease was carried out under anesthesia, for which dogs were administered 2.5% solution of sodium thiopental at a rate of 0.4 - 0.5 ml per 1 kg of body weight of the animal. After antiseptic treatment of the skin the peritoneal cavity was opened with a median incision and ligature was applied with a capron thread No. 5 on the loop of the small intestine for 15 cm long at a distance of 80 cm from the ileocecal transition. After 3, 6, 12 and 24 hours following the creation of acute strangulational small bowel obstruction model for the preparation of histological samples the sections of liver tissue was excised from the anterior margin of the right lobe size 2 × 3 cm. The pieces were fixed in a Cornois liquid and then in a 12% neutral formalin and after the appropriate washing, was poured with paraffin. Slices of 5 - 7 microns thick were prepared from the material embedded in paraffin. Samples were stained with hematoxylin and eosin and using the Schiff (PAS) reaction method.
The third control group, consisted of 8 animals, in whom the study of liver morphology after laparotomy without intervention on the vessels and organs of the abdominal cavity was performed. The sampling of the material for the study was carried out 3, 6, 12 and 24 hours after laparotomy. Comparison of the results of the second and third groups allows to reveal changes in the liver caused by acute small bowel obstruction, but not the consequences of laparotomy for the modeling of the disease.
The choice of observation periods is based on the peculiarities of the clinical manifestations of ASBO. The manifestation of clinical symptoms of acute strangulational small bowel obstruction occurs almost immediately after the formation of strangulation, as the pathological process involves mesentery of the small bowel, which contains a large number of blood vessels and nerve endings.
METHODS OF RESEARCH INCLUDED
1 - Methods of setting experiments - modeling of acute strangulational small bowel obstruction
2 - Methods for the study of liver morphology
Methods for studying liver morphology under normal conditions and after modeling of small bowel obstruction were the same. A morphometric method was used to assess the change in the volume of the vascular bed in terms of the degree of change in the area occupied by the departments (arterial part of the hemomicrocirculatory bed of the liver, capillary and venous) in a histological sample on a 600 × 103m2, samples were stained with hematoxylin and eosin. The ratio of the volume of the vascular system of the portal system, the system of the hepatic artery and the system of hepatic veins (central veins of liver lobules) is calculated on the basis of a comparison of the calculated value of the total radius of the vascular bed by applying formula;
S=πr2
where S - the area of the vascular bed in the selected standard area of the sample.
The calculated value of the total radius of the vascular bed of interlobular veins (interlobular arteries, central veins of the lobules) of the liver is based on the generally accepted conclusion about the identical structure and organization of vascular branching of portal vein system and the system of the hepatic artery, which are the vessel-satellites, respectively having the same length and branches but a different diameter (radius) of the vessels clearence, consequently, a different volume of the vascular bed (basin).
Glycogen content estimation in the liver was determined by the results of the quantitative evaluation of products of Schiff (PAS) reaction. The area occupied by the cytochemical reaction products having a specific color intensity per unit area of 7500 µm2 was estimated. Morphometry was performed using the program “Adobe Photoshop CS3 Extended RUS”, “Image Pro”, “Image 3.0”. The last group of methods was a statistical analysis of the data. Statistical processing of the results was carried out using the methods of variational statistics in the programs “Statistica 6.0”. (StatSoft Inc., USA), AtteStat. The results were considered statistically significant at p < 0.05 (95% accuracy).
RESULTS
Normally the area of interlobular veins is 33.6 times larger than the area of interlobular arteries, the area of the central veins (the system of the inferior vena cava) is 37.2 times larger than the interlobular artery, which corresponds to the calculated ratio of the volume of the portal vein system and the hepatic artery system 5.8:1, the inferior vena cava system and the hepatic artery system 6:1. Consequently, the main role in ensuring blood supply to the liver belongs to the vessels of the portal system and the volume of the central veins of the lobe - the vessels of the basin of the inferior vena cava, equals to total blood flow volume both through the portal and along the arterial course. The estimated ratio of the volume of the portal vein system and the hepatic artery system, as well as the ratio of the volume of the inferior vena cava system and the hepatic artery system in the dynamics of modeling of acute strangulational small bowel obstruction is presented in table 1.
|
Test Parameters |
Experiment |
|||
|
After 3 hours |
After 6 hours |
After 12 hours |
After 24 hours |
|
|
1. The estimated ratio of the volume of the portal vein system and the hepatic artery system |
4, 39:1 |
2, 82:1 |
4, 45:1 |
4, 22:1 |
|
2. The calculated ratio of the volume of the inferior vena cava system and the hepatic artery system |
5, 95:1 |
1, 85:1 |
6, 08:1 |
5, 15:1 |
Table 1: The estimated ratio of the volume of the portal vein system and the hepatic artery system (1), as well as the ratio of the volume of the inferior vena cava system and the hepatic artery system (2) in the dynamics of acute strangulational small bowel obstruction modeling.
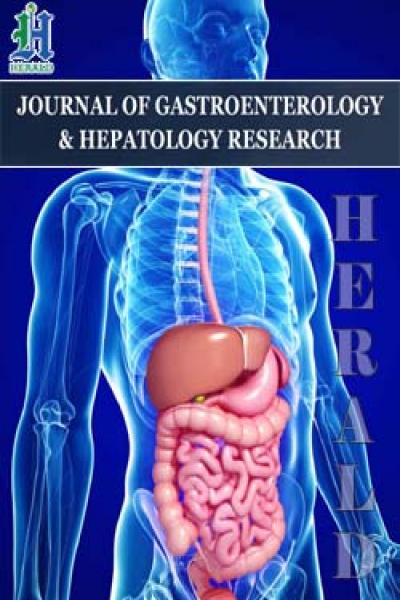
Three hours after the modeling of ASBO, there is a statistically significant increase in the area of interlobular veins 1.62 times and interlobular arteries 2.82 times in comparison with the norm. At this timeframe of the experiment, the area of the central veins increases statistically significantly in comparison with the norm by 2.68 times. In 3 hours after the formation of strangulation ASBO, a statistically significant increase in the total vascular bed volume was observed 2.28 times (Figures 1 - 4).
 Figures 1&2: The parameters of the total area of the vascular bed (Figure 1), The area of interlobular arteries (Figure 2) per unit area of 600 × 103 µm2 with acute strangulational small bowel obstruction.
Figures 1&2: The parameters of the total area of the vascular bed (Figure 1), The area of interlobular arteries (Figure 2) per unit area of 600 × 103 µm2 with acute strangulational small bowel obstruction.
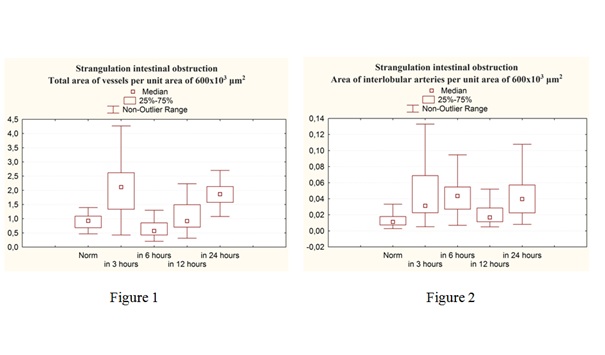
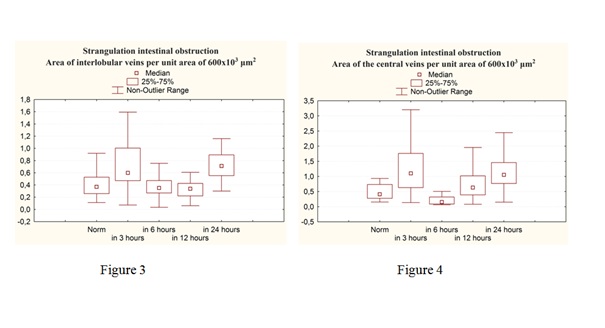 Figures 3&4: The indices of the area of interlobular veins (Figure 3), the area of the central veins (Figure 4) per unit area of 600 x 103 µm2 in acute strangulational small bowel obstruction.
Figures 3&4: The indices of the area of interlobular veins (Figure 3), the area of the central veins (Figure 4) per unit area of 600 x 103 µm2 in acute strangulational small bowel obstruction.
At the same period of the experiment, we detected a decrease in the level of glycogen in the hepatocytes both in the central and peripheral parts of the lobules (Figures 5,6).
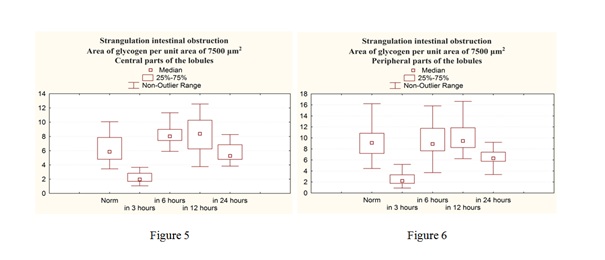 Figures 5&6: Indices of glycogen area per unit area of 7500 mkm2 for acute strangulational small bowel obstruction in the central parts of the lobules (Figure 5), in the peripheral parts of the lobules (Figure 6).
Figures 5&6: Indices of glycogen area per unit area of 7500 mkm2 for acute strangulational small bowel obstruction in the central parts of the lobules (Figure 5), in the peripheral parts of the lobules (Figure 6).
In the central sections of the hepatic lobules, a statistically significant decrease in the glycogen level was observed 2.98 times as compared with the norm. In the peripheral parts of the lobules, a statistically significant decrease in the glycogen level in 4.16 times is also noted in comparison with the norm.
After 6 hours from the beginning of the experiment, vasodilation of the arterial link of the microvascular bed was observed, a statistically significant increase in the area of interlobular arteries was observed 4 times when compared with the norm. At the same time, a statistically insignificant decrease in the area of interlobular veins was observed 1.06 times in comparison with the norm and a statistically significant decrease in the area of interlobular veins in 1.71 times in comparison with the previous period of the experiment (Figure 3).
The area of the central veins was statistically significantly decreased by 2.73 times compared with the norm and by 7.33 times in comparison with the previous period of the experiment (Figure 4).
The total area of the vessels decreases 1.63 times in comparison with the norm and 3.72 times in comparison with the previous period of the experiment, mainly due to deterioration of the blood supply through the portal system (Figure 1).
After 6 hours from the beginning of the experiment, a statistically significant increase in the glycogen level in the central sections was observed in 1.37 times, compared with the norm and a statistically significant increase in the glycogen level in the central sections of the lobules in 4.09 times compared with the previous period of the experiment (Figure 5). The level of glycogen in the peripheral parts of the lobules did not change statistically significantly as compared with the norm, but in comparison with the previous period of the experiment statistically significantly increased 4.07 times, practically reaching the norm (Figure 6).
12 hours after the formation of strangulational acute intestinal obstruction, the area of interlobular arteries statistically significantly increases by 1.55 times in comparison with the norm, and when compared with the previous period of the experiment the area of interlobular arteries decreases 2.59 times, which indicates a generalized spasm of vessels of a great circle of blood circulation, with spreading to the vessels of the celiac trunk (Figure 2). The area of interlobular veins at this time of the experiment decreases 1.09 times in comparison with the norm, it does not change statistically significantly in comparison with the previous period (Figure 3).
At this time of the experiment, the area of the central veins, in comparison with the norm, increases statistically significantly by 1.54 times, and in comparison with the previous period of the experiment it increases by 4.2 times (Figure 4).
There is a statistically significant gradual increase in the total share of the vascular bed in 1.61 times compared with the previous period of the experiment, practically reaching the norm (Figure 1) due to increased arterial blood flow.
12 hours after the formation of the strangulational ASBO, the level of glycogen in the central sections of the lobules increases 1.43 times when compared with the norm, it does not change statistically significantly when compared with the previous period of the experiment, and reaches the norm in the peripheral parts of the lobules and, when compared with the previous period of the experiment, statistically does not change significantly (Figures 5,6).
24 hours after the formation of the strangulational ASBO the area of interlobular arteries, when compared with the norm, increases statistically significantly 3.63 times, and compared with the previous period of the experiment, the area of interlobular arteries increases 2.35 times (Figure 2). The area of interlobular veins at this time of the experiment, as compared with the norm, increases statistically significantly by 1.92 times, statistically significantly increases by 2.09 times in comparison with the previous period (Figure 3).The area of the central veins, as compared to the norm, increases statistically significantly by 2.59 times, and in comparison with the previous period of the experiment it increases by 1.68 times (Figure 4).
In this case, the total area of the vessels increases by 2.01 times in comparison with the norm and by 2.03 times in comparison with the previous period of the experiment (Figure 1).
After 24 hours, a statistically significant decrease in the level of glycogen in the central parts of the lobules is observed when compared with the norm of 1.11 times, and when compared with the previous period of the experiment, a statistically significant decrease by 1.59 times. In the peripheral parts of the lobules a statistically significant decrease in the glycogen content is observed when compared with the norm by 1.45 times, and when compared with the previous period of the experiment a statistically significant decrease in 1.50 times (Figures 5,6).
The correlation coefficient of the area of the HMCB (the total area of the vascular bed, the area of interlobular veins, interlobular arteries and central veins) of the liver with the level of glycogen in the central and peripheral parts of the lobules is determined by the Spearman correlation coefficient (r) (Table 2).
|
Evidence |
Total area of vessels |
Area of interlobular arteries |
The area of interlobular veins |
The area of the central veins |
|
|
Glycogen central sections of the lobules |
-0.9 (?<0.05) |
-0.1 (?<0.05) |
-0.9 (?<0.05) |
-0.7 (?<0.05) |
|
|
Glycogen peripheral parts of the lobules |
-0.7 (?<0.05) |
-0.5 (?<0.05) |
-0.8 (?<0.05) |
-0.6 (?<0.05) |
|
Table 2: Spearman correlation coefficient (r) for assessing the relationship between the change in the area of the HMCB (the total area of the vascular bed, the area of interlobular veins, interlobular arteries, central veins) of the liver with the level of glycogen in the central and peripheral parts of the lobules.
1. Dependence of signs is statistically significant (p < 0.05)
2. Communication between the characteristics being investigated (if the “+” is a direct link, if the “-“ - connection is inverse)
3. Tightness (strength) of the bond: less than 0.3 - weak bond, from 0.31 to 0.5 - moderate; from 0.51 to 0.7 -significant, from 0.71 to 0.9 close; from 0.91 to 1 very close.
DISCUSSION
The results of our study coincide with the conclusions presented in cases [3,4] and confirm that the liver is the first target organ on which the main impact of toxemia occurs in the form of a massive supply of biologically active substances and toxic products via the portal vein with blood, formed during ischemia and/or necrosis of tissues of the pathologically altered portion of the small intestine.
The result of our study is the determination of the timing of the maximum changes in the hemocirculatory bed and the energy metabolism correlated with it in the liver in the dynamics of the development of acute strangulational small bowel obstruction.
We have revealed a strong inverse correlation between the change in the area of the vascular bed and the energy metabolism. The increase in the total area of the vascular bed (reactive hyperemia of the organ) after 3 hours and 24 hours after the onset of the formation of acute strangulational small bowel obstruction, which occurs mainly due to an increase in the area of interlobular veins and central veins of the liver lobules, was accompanied by a decrease in glycogen levels during these periods of the experiment. We believe that the glycogen level decreased due to a change in the distribution of incoming blood, the predominance of venous blood flow through the portal system saturated with toxic products and biologically active substances resulting from ischemia and/or necrosis of tissues of the pathologically altered site of the small intestine, which, in its in turn, required an increase in energy costs for the performance of a detoxifying function. The area of interlobular arteries at all times of the experiment exceeded the norm, which, in our opinion, is due to the increase in the body's oxygen demand. After 6 hours and 12 hours from the onset of the formation of the spinal CT, we observed a decrease in the blood supply of the liver through the portal system, the toxic effect on hepatocytes decreased and the level of glycogen increased. It is likely that the decrease in the flow of venous blood through the portal system is due to a decrease in the influx of arterial blood to the intestine due to the reactive spasm of the vessels of the great circle of blood circulation, observed under stress.
Paralytic vasodilation, which develops at the maximum time of observation in the experiment leads to an increase in the flow of toxins into the blood from the area of pathological changes in the small intestine. Toxins, in turn, have a direct damaging effect on liver cells, damage the membrane structures of hepatocytes and disrupt the transmembrane transport of hydrogen and other substrates, which in turn leads to discoordination of metabolic processes and subsequently, with the progression of the disease, to death cells. The result of this process is the development of hepatic insufficiency of varying severity, which is confirmed by the results of studies [13,14].
Reduction in the level of glycogen in liver at the end of the experiment indicates a violation of energy metabolism at the organ level. These changes in turn can lead to disruption of carbohydrate metabolism and energy processes in other organs and tissues, since the liver is one of the main organs for storing glycogen. Insufficiency of energy metabolism in the liver leads to a lack of protein synthetics, detoxification, reparative, plastic and other liver functions, since all processes in the liver are associated with the consumption of glycogen, which is the main source of energy in metabolic processes.
The changes in the microcirculatory bed and energy metabolism revealed by us during the modeling of acute strangulational small bowel obstruction characterize a marked decrease in functional and reparative capacities of the liver tissue. Energy deficiency leads to a decrease in metabolic, detoxifying, excretory and other functions of the liver.
Dysfunction of the liver and violation of its barrier function, in turn, is the basis of systemic intoxication, contributes to the defeat of other organs and body systems, the violation of protein, lipid, enzyme, mineral, water metabolism, discoordination of protective and compensatory reactions at the body level, i.?., a vicious pathogenetic circle is formed, the result of which is the development of the syndrome of multiple organ failure. Thus our study also confirms the conclusions [12] about the need for early corrective therapy in patients with strangulational ASBO.
REFERENCES
- Petrovic B, Nikolaidi P, Hammond N, Grant TH, Miller FH (2006) Identification of adhesions on CT in small-bowel obstruction. Emerg Radio 12: 88-95.
- Jackson PG, Raiji MT (2011) Evaluation and Management of Intestinal Obstruction. Am Fam Physician 83: 159-165.
- Samell S, Keesel M, Kleczka M, Lanig S, Gretz N, et al. (2002) Microscopy of bacterial translocation during small bowel obstruction and ischemia in vivo-a new animal model. BMC Surg 13: 2-6.
- Costa RIDD, Rasslan R, Koike MK, Utiyama EM, Montero EFS (2017) Bacterial translocation and mortality on rat model of intestinal ischemia and obstruction. Acta Cir Bras 32: 641-647.
- Akçay MN, Capan MY, Gündogdu C, Polat M, Oren D (1996) Bacterial translocation in experimental intestinal obstruction. J Int Med Res 24: 17-26.
- Taylor MR, Lalani N (2013) Adult small bowel obstruction. Acad Emerg Med 20: 528-544.
- Cerniy V, Turek Z, Parizkova R (2009) In situ assessment of the liver microcirculation in mechanically ventilated rats using sidestream dark-field imaging. Physiol Res 58: 49-55.
- Sakamoto S, Okanoue T, Itoh Y, Nakagawa Y, Nakamura H, et al. (2002) Involvement of Kupffer cells in the interaction between neutrophils and sinusoidal endothelial cells in rats. Shock 18: 152-157.
- Maronne G, Shah VH, Gracia-Sancho J (2016) Sinusoidal communication in liver fibrosis and regeneration. J Hepat 65: 608-617.
- Wohlleber D, Knolle PA (2016) The role of liver sinusoidal cells in local hepatic immune surveillance. Clin Transl Immunology 5: 117.
- Nolan JP (2010) The role of intestinal endotoxin in liver injury: a long and evolving history. Hepatology 52: 1829-18
- Skrypko VD, Klymenko AO, Honchar MH, Klymenko IuA (2014) Correction of early liver dysfunction in patients with acute small bowel obstruction. Klin Khir 15-1
- Bernal W, Wendon J (2013) Acute Liver Failure. N Engl J Med 369: 2525-2534.
- O’Grady JG (2005) Acute liver failure. Postgrad Med J 81: 148-154.
- Adhikari S, Hossein MZ, Das A, Mitra N, Ray U (2010) Etiology and outcome of acute intestinal obstruction: A review of 367 patients in Eastern India. Saudi J Gastroenterol 16: 285–287.
- Vakaraeva MM (2016) Mechanisms of regulation of sugar levels in the human body. Intel potents XXI veka: stup pozn 36: 7-11.
- Lebedeva EI, Myader OD (2015) Histochemistry of glycogen in the liver of rats with experimental cirrhosis. Dostizheniya fund, klin med i farm 2015; 198-199.
- Engin A, Ferahkö?e Z, Ozdemir E, Altan N (1988) Liver glycogen in gangrenous intestinal obstruction. Res Exp Med (Berl) 188: 299-303.
- Zapadnyuk IP, Zapadnyuk VI, Zakharnya EA (1983) Laboratory animals: their breeding, keeping and usage in experimental researches. Vyishcha shkola, Kiev, Ukraine.
- Kopaladze RA (2004) The experiments with laboratory animals from a bioethical point of view--history, modern time, perspectives. Usp Fiziol Nauk 35: 92-109.
- Chadayev VYe (2012) Ethical principles when working with laboratory animals. Vestnik Probl Biol I Med 1: 113-115.
Citation: Milyukov VE, Sharifova KM (2018) Morphofunctional Changes of Blood Supply and the Glycogen Content in the Liver during Acute Strangulational Small Bowel Obstruction. J Gastroenterol Hepatology Res 3: 012.
Copyright: © 2018 Vladimir Efimovich Milyukov, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.