Nanomedicine to Tumor by Enzymosomes
*Corresponding Author(s):
Basavaraj K NanjwadeDepartment Of Pharmaceutics, Faculty Of Pharmacy, Omer Al-Mukhtar University, Tobruk, PO Box 919, Libyan Arab Jamahiriya
Tel:+218 944828793,
Email:nanjwadebk@gmail.com
Abstract
Keywords
INTRODUCTION
Enzymosomes are the part of vesicular drug delivery system, enzymo means enzymes and somes means cell-like, these are liposomal constructs engineered to provide a mini bioenvironmental in which enzymes are covalently immobilized or coupled to the surface of liposomes [2]. Most of the catalytic enzymes are useful in enhancing the potent activity of antitumor drugs, such enzymes are alkaline phosphatase, carboxypeptidase, β-glucosidase, β-lactamase. The enzyme reaction specificity provides the prodrug activation at the tumour site, through prior enzyme targeting by using liposomes or via enzyme expressing gene delivery in to the tumour cells [3].
Present study, delivery methods for an enzyme/prodrug strategy can be divided into two major classes: (a) delivery of genes that encode prodrug-activating enzymes into tumour tissues (Gene-Directed Enzyme Prodrug Therapy (GDEPT), Virus Directed Enzyme Prodrug Therapy (VDEPT), etc.,); and (b) delivery of active enzymes onto tumour tissues (Antibody-Directed Enzyme Prodrug Therapy (ADEPT) [4].
For the targeting of cytotoxic and antiviral agents to specific cell types is the prodrug activation by antibody enzyme conjugates (known as prodrug activation therapy, ADEPT or Antibody-Directed Catalysis (ADC) [5]. The cancer chemotherapy is limited by lack of specificity toward the tumour cells. This problem has been solved by ADEPT also it is known as an alternative for conventional cancer therapy [6]. In this approach an antibody linked by an enzyme that binds to an antigen preferentially expressed on tumour cells (Figure 1). Subsequent administration of a prodrug results in appropriate conversion of the prodrug into the parent cytotoxic drug at the tumour site. The converted prodrug should kill not only the tumour cells to which the conjugate shave bound but also neighbouring tumour cells [7]. The prodrug is designed to be a substrate for the chosen enzyme (Figure 2).
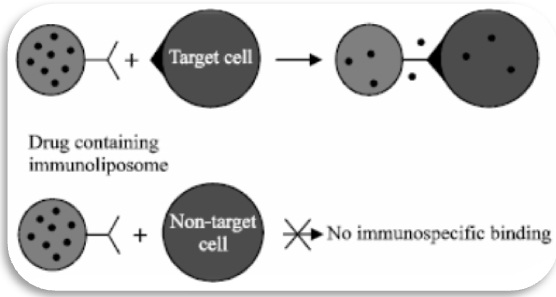
Figure 1: Enzyme linked drugs targeting tumour cells.
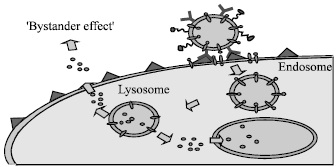
Figure 2: Bystander killing effect.
The enzyme-activating prodrug therapy in a two-step approach. First step, a drug-activating enzyme is targeted and expressed in tumours. In the second step nontoxic prodrug called as a substrate of the exogenous enzyme is now expressed in tumours and it is administered by parenteral route [2].
The prodrug activation generates a very small toxic species which is not affected by the neutralising action of serum enzymes and may have a half-life long enough to have a by stander killing effect. This is the cyanide generation system devised by Rowlinson-Busza in 1992 (antibody guided enzyme nitrile therapy) [8].
In tumour therapeutics dun the various body immune components are recognised for attack of disease i.e., tumour antigens (vaccine), antibodies, Monoclonal Antibodies (MAb), cytokinase and immunotoxins etc.
To avoid heterogeneity problem in antigen expression the MAb-guided therapy is useful by using antibodies to carry enzymes to tumour cells. There is huge number of different types of enzymes used in the conversion of prodrugs into respective active drug by coupling with antigen/antibody. Such enzymes are Carboxypeptidase G2, ?-lactamase, ?-glucuronidase, ?-galactosidase, Alkaline Phosphatase, Penicillin V-amidase, Cytosine Deaminase, Nitroreductase, Carboxypeptidase A and catalytic antibodies. Followed by the activity of enzymes is the release of active drug required at the targeted tumour site. Lastly, heterogeneity in antigen expression will result in escape of antigen-negative cells from treatment [2].
The main objective is to develop and optimize enzymosomes, with long circulation times in the blood in order to accumulate at target tumour site, while maintaining enzymatic activity in its intact form. Therefore, the present review work is designed to study of enzymosomes for which enzyme release from the lipidic particles is not needed and simultaneously being able to exert therapeutic effect even when the liposomes remain intact within the pathological site of action. Currently enzymes used for prodrug activation systems with antigen/antibody showing in (Table1) [9].
| Antibody/Antigen | Enzyme | Prodrug | Active drug |
| MAbs W14A and SBIO/chorionic onadotropin | Carboxypeptidase G2 | Benzoic acid mustards-glutamic acid | Benzoic acid mustards |
| No antibody in study | β-Lactamase | Vinca-cephalosporin | Vinca alkaloid |
| MAb L6 (against a carbohydrate antigen on human carcinomas) | Penicillin amidase |
Doxorubicin-phenoxyacetamide Melphalan-phenoxyacetamide |
Doxorubicin Melphalan |
| MAb BW431/26/Carcinoembryonic Antigen (CEA) | Alkaline phosphatase | Etoposide phosphate | Etoposide |
| MAb 323/A3 (against a pan-carcinoma membrane glycoprotein) | β-GIucuronidase | Epirubicin-glucoronide | Epirubicin |
| MAb KS1/4/UCLA-P3 human lung adenocarcinoma | Carboxypeptidase A | Methotrexate-alanine | Methotrexate |
Table 1: Currently used prodrug activation systems with antigen/antibody.
Different strategies can be used to improve carrier mediated delivery of therapeutic proteins as: its incorporation into polymeric carriers, into aqueous space of lipid, detergent or lipid-detergent vesicles or incorporation of hydrophobized ones into lipid bilayer of vesicles. A strategy not usually used for therapeutic enzymes. It is attachment to the outer surface of liposomes and using technologies developed for antibodies. Some publications report the construction of carriers with surface attached enzymes in contrast with a huge number of publications with antibodies attached to carriers concept widely used for the active targeting of nanocarriers. Superoxide Dismutase (SOD) is used as a therapeutic agent for oxidative stress related diseases such as Rheumatoid Arthritis and Ischaemia/Reperfusion situations. Our objective was to develop and optimize SOD enzymosomes, with long circulation times in the blood in order to accumulate at inflamed target sites, while maintaining enzymatic activity in its intact form [10].
Enzymes are promising therapeutic agents due to their specific substrate selectivity and unparalleled reaction efficiency. The intrinsic shortcomings of enzymes, relatively low enzymatic activity under physiological conditions and short circulation life (Figure 3). An inherent immunogenicity and might be overcome by encapsulating them into certain biocompatible and biodegradable drug carriers, such as lipid vesicles [11].
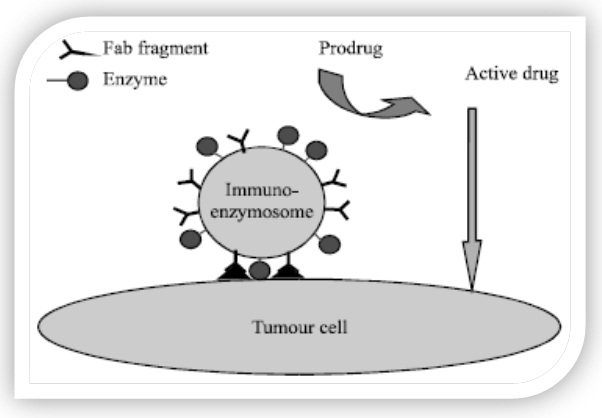
Figure 3: Prodrug converted in to active drug by enzymes.
ADVANTAGES OF ANTIBODY DIRECTED ENZYME PRODRUG THERAPY
1. A single enzyme can convert a large number of prodrugs to the cytotoxic agents; therefore, the drug potency should not be a problem.
2. The antibody-enzyme complex and prodrug are relatively non-toxic unlike the radionuclide and they should not cause damage when they are in the systemic circulation.
3. Like radionuclide can also overcome the problems of antigenic heterogeneity and tumor accessibility [9].
DISADVANTAGES OF ANTIBODY DIRECTED ENZYME PRODRUG THERAPY
ADEPT will result in toxicity in normal cells, if:
a) The body itself has enzymes that can convert the prodrug to the cytotoxic agent.
b) A residual amount of enzyme-antibody conjugates remains in the systemic circulation
c) The produced cytotoxic agent at the tumor site gets back to the systemic circulation.
d) The tumor specific antibody has cross reactivity with the cell surface antigens of normal cells and delivers the enzyme to the normal tissue [12].
Genotype/Phenotype correlation
Monoallelic mutations in FANCD1/BRCA2, FANCJ/BRIP1, FANCN/PALB2, FANCO/RAD51C, FANCP/SLX4 and FANCQ/XPF are related with ovarian and breast cancer, but molecular defects in these genes are also identified in the non-FA population [33,42-46].
LIPOSOME PREPARATION
Film hydration method, pH 5.6
FREEZE-THAWING METHOD, PH 5.6
Dehydration and rehydration method, pH 5.6
CONJUGATION OF ENZYME SOD TO LIPOSOMES FOR IN VIVO STUDIES
LIPOSOME CHARACTERIZATION
PHYSICOCHEMICAL CHARACTERIZATION
Size and zeta potential
Photon Correlation Spectroscopy (PCS)
Laser diffraction with sub-micron equipment
Asymmetrical Flow Field-Flow Fraction (A4F)
Zeta potential
PHARMACOKINETICS AND PHARMACODYNAMICS (PK/PD)
STABILITY STUDIES
REGULATORY ASPECTS
FUTURE PROSPECTIVE
| Vesicular System | Description | Description |
| Enzymosomes | Liposomal construct engineered to provide a mini bio environment in which the enzyme covalently immobilized to the surface of liposomes. | Targeted delivery to tumor cell. |
| Virosomes | Liposomes spiked with virus glycoprotein’s, incorporated in the liposomal bilayer based on retrovirus based lipids. | Immunological adjuvants. |
| Ufasomes | Vesicles enclosed by fatty acids obtained by long chain fatty acids by mechanical agitation of evaporated film in the presence of buffer solution. | Ligand mediated drug targeting. |
| Cryptosomes | Lipid vesicle with surface coat composed of PC and of suitable polyoxyethylene derivative of phosphatidyl ethanolamine. | Ligand mediated drug delivery. |
| Emulsomes | Nanosized lipid particles consisted of lipid assembly and a polar group. | Parenteral delivery of poorly water soluble drugs. |
| Discosomes | Niosomes coupled with non-ionic surfactants. | Ligand mediated drug targeting. |
| Aquasomes | These are spherical 60-300 nm particles used for drug and antigen delivery. The particle core is composed of non crystalline calcium phosphate or ceramic diamond and is covered by a polyhydroxyloligomeric film. | Specific targeting, molecular shielding. |
| Collidosomes | Are solid microcapsules formed by the self assembly of colloidal particles at the interface of emulsion droplets? “Colloidosomes” are hollow, elastic shells whose permeability and elasticity can be precisely controlled. | Drug targeting. |
| Genosomes | Artificial macromolecular complex for functional gene transfer. | Cell specific gene transfer. |
| Photosomes | Photolyase encapsulated in liposomes, which release the contents by photo triggered charges in membrane permeability characteristics. | Photodynamic therapy. |
| Erythrosomes | Liposomal system in which chemically cross-linked human erythrocytes cytoskeletons are used as to which a lipid bilayer is coated. | Targeting of macromolecular drugs. |
| Hemosomes | Heamoglobin containing liposomes engineered by immobilizing heamoglobin with polymerizable phospholipids. | High capacity oxygen carrying system. |
| Protosomes | High molecular weight multi subunit enzyme complexes with catalytic activity. | Better catalytic activity turnover than non associated enzymes. |
| Vesosomes | Nested bilayer composed of bilayer. | Multiple compartments of vesosomes give better protection to the interior content of serum. |
| Archaeosomes | Vesicles composed of gylcerolipids of archaea with potent adjuvant activity. | Poor adjuvant activity. |
| Cubosomes | Are bicontinuous cubic phases, consisting of two separate, continuous, but non intersecting hydrophilic regions divided by a lipid layer that is contorted into a periodic minimal surface with zero average curvature. | Drug targeting. |
Table 2: Comparative overview of enzymosome with current emerging vesicular system.
CONCLUSION
REFERENCES
- Bhowmik D, Chiranjib, Chandira RM, Tripathi KK, Kumar KPS (2010) Nanomedicine-An overview. International Journal of PharmTech Research 2: 2134-2151.
- Kumar R, Kumar S, Jha SS, Jha AK (2011) Vesicular System-Carrier for Drug Delivery. Pelagia Research Library 2:192-202.
- Vyas SP, Khar RK (2012) Immunotherapy of tumour. Targeted & Controlled Drug Delivery: Novel Carrier Systems. CBS, India. Pg: 521-523.
- Deonarain MP, Epenetos AA (1994) Targeting enzymes for cancer therapy: old enzymes in new roles. Br J Cancer 70: 786-794.
- Fonseca MJ, Jagtenberg JC, Haisma HJ, Storm G (2003) Liposome-mediated targeting of enzymes to cancer cells for site-specific activation of prodrugs: comparison with the corresponding antibody-enzyme conjugate. Pharm Res 20: 423-428.
- Vingerhoeds MH, Haisma HJ, van Muijen M, van de Rijt RB, Crommelin DJ, et al. (1993) A new application for liposomes in cancer therapy. Immunoliposomes bearing enzymes (immuno-enzymosomes) for site-specific activation of prodrugs. FEBS Lett 336: 485-490.
- Xu G, McLeod HL (2001) Strategies for enzyme/prodrug cancer therapy. Clin Cancer Res 7: 3314-3324.
- Haisma HJ, Boven E, van Muijen M, de Jong J, van der Vijgh WJ, et al. (1992) A monoclonal antibody-beta-glucuronidase conjugate as activator of the prodrug epirubicin-glucuronide for specific treatment of cancer. Br J Cancer 66: 474-478.
- Foldvari M, Jaafari MR, Mezei M, Mezei C (1998) Targeting of liposomes through immunoglobulin superfamily domains: P0 protein as a model. Drug Deliv 5: 183-195.
- Vale CA, Corvo ML, Martins LCD, Marques CR, G Storm, et al. (2006) Construction of Enzymosomes: Optimization of Coupling Parameters. NSTI-Nanotech 2: 396-397.
- Tan QY, Zhang JQ, Wang N, Yang H, Li X, et al. (2012) Improved biological properties and hypouricemic effects of uricase from candida utilis loaded in novel alkaline enzymosomes. Int J Nanomedicine 7: 3929-3938.
- Corvo ML, Jorge JC, van't Hof R, Cruz ME, Crommelin DJ, et al. (2002) Superoxide dismutase entrapped in long-circulating liposomes: formulation design and therapeutic activity in rat adjuvant arthritis. Biochim Biophys Acta 1564: 227-236.
- Biel S, Simon J, Gross R, Ruiz T, Ruitenberg M, et al. (2002) Reconstitution of coupled fumarate respiration in liposomes by incorporating the electron transport enzymes isolated from Wolinella succinogenes. Eur J Biochem 269: 1974-1983.
- Riaz M (1996) Liposomes preparation methods. Pak J Pharm Sci 9: 65-77.
- Traïkia M, Warschawski DE, Recouvreur M, Cartaud J, Devaux PF (2000) Formation of unilamellar vesicles by repetitive freeze-thaw cycles: characterization by electron microscopy and 31P-nuclear magnetic resonance. Eur Biophys J 29: 184-195.
- Gaspar MM, Boerman OC, Laverman P, Corvo ML, Storm G, et al. (2007) Enzymosomes with surface-exposed superoxide dismutase: In Vivo behaviour and therapeutic activity in a model of adjuvant arthritis. Journal of Control Release 117: 186-195.
- Ruozi B, Belletti D, Tombesi A, Tosi G, Bondioli L, et al. (2011) AFM, ESEM, TEM, and CLSM in liposomal characterization: a comparative study. Int J Nanomedicine 6: 557-563.
- Kuntsche J, Mader K (2010) Solid Lipid Nanoparticles (SLN) for Drug Delivery. In: Torchilin V, Amiji MM (eds.). Handbook of Materials for Nanomedicine. Pan Stanford Publishing, United Kingdom. Pg: 403-408.
- http://ncore.web.unc.edu/in-vivo-pkpd-studies/
- Yoshimoto M (2011) Stabilization of enzymes through encapsulation in liposomes. Methods Mol Biol 679: 9-18.
- Gaspar RS, Florindo HF, Silva LC, Videira MA, Corvo ML, et al. (2014) Regulatory Aspects of Oncologicals: Nanosystems Main Challenges. In: Alonso MJ, Garcia-Fuentes M (eds.). Nano-Oncologicals. Springer International Publishing, Switzerland, Pg: 425-452.
- Rathore P, Swami G (2012) Planterosomes: a potential phyto-phospholipid carriers for the bioavailability enhancement of herbal extracts. IJPSR 3: 737-755.
Citation: Hundekar YR, Nanjwade BK, Mohamied AS, Idris NF, Srichana T, et al. (2015) Nanomedicine to Tumor by Enzymosomes. J Nanotechnol Nanomed Nanobiotechnol 2: 004.
Copyright: © 2015 Basavaraj K Nanjwade, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.