
Neurotologic Complications Following Microvascular Decompression: A Retrospective Study
*Corresponding Author(s):
John LeonettiDepartment Of Neurological Surgery, Loyola University Medical Center, Illinois, United States
Tel:+1 7082161676,
Email:jleonet@lumc.edu
Abstract
Microvascular Decompression (MVD) offers effective, long-term symptom relief for patients with pathologies related to neurovascular conflicts of cranial nerves in the posterior fossa. We aimed to determine the frequency of neurotologic complications following this surgery. This is a retrospective case review in a tertiary care referral center. A total of 215 consecutive MVD operations by a single surgeon between March 1996 and May 2016 were reviewed with 192 surgeries on 183 patients meeting inclusion criteria. The 52 males and 131 females had a mean age of 58.52 years (range 28-92 years). Indications for MVD were trigeminal neuralgia (n=162), hemifacial spasm (n=23), glossopharyngeal neuralgia (n=13), vagal palsy (n=1), and tinnitus (n=1). The outcomes examined were postoperative hearing loss, tinnitus, vertigo, and hemifacial paresis. At least one of these complications was present in 17.7% of patients. There were 4.17% with permanent hearing loss, 6.77% with transient hearing loss, 5.21% with tinnitus, 5.73% with vertigo, and 0.52% with hemifacial paresis. There was no significant difference in complication rates based on surgical indication. Complication rates in this study are consistent with those in the literature. We recommend perioperative audiometry in all patients undergoing MVD. It is important for otolaryngologists as well as neurosurgeons to be aware of neurotologic complications in order to adequately counsel patients preoperatively and identify them postoperatively.
Keywords
INTRODUCTION
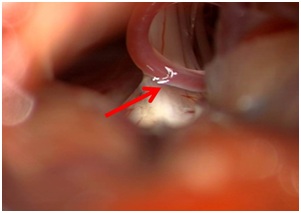
Figure 1: Intraoperative image of neurovascular conflict.
The first line of treatment for TN, HFS, and GPN is pharmacotherapy. Medical management may initially yield a satisfactory response, but persistence or recurrence of symptoms and side effects of the medications can be too severe for some patients to tolerate. For these patients, the next step in treatment may be surgical.
Peter Jannetta demonstrated the efficacy of Microvascular Decompression (MVD) in 1967 [4]. The procedure involves correcting the neurovascular conflict by repositioning the offending vessel and placing spacer material between it and the affected cranial nerve [2]. This decompresses the nerve root exit zone and has been shown to ameliorate symptoms. A retrosigmoid approach has historically been used to approach the Cerebellopontine Angle (CPA). Other options such as balloon compression, rhizotomy and radiosurgery are available, but MVD has been shown to be the procedure of choice for TN [5], HFS [2,6] and GPN [1]. MVD has also been shown to be effective for less common indications such as tinnitus and vagal palsy [7].
MVD has consistently been validated with reports of high satisfaction rates and long term results [8-12]. However, some patients suffer postoperative neurotologic complications such as hearing loss, tinnitus, vertigo, and hemifacial paresis [13]. Neurotologic complications are thought to be secondary to: stretching of CN VII and VIII during cerebellar retraction, direct trauma to CN VII or VIII, excessive manipulation of vasculature (especially the labyrinthine artery and anteroinferior cerebellar artery) or neocompression of nerves with the spacer placement [14,15].
There have been several studies that examined hearing loss, but very few have evaluated tinnitus, vertigo and hemifacial paresis [14,16]. There is also a relative lack of studies distinguishing between transient, Conductive Hearing Loss (CHL) and permanent Sensorineural Hearing Loss (SNHL). Our study aims to determine the frequency of neurotologic complications after posterior fossa MVD in our past 20 year experience.
PATIENTS AND METHODS
Patients were excluded if they had undergone previous radio surgery, had a history of CPA tumor, had a previous surgery in the CPA other than MVD, or had follow-up of less than two weeks. A total of 32 patients were excluded based on these criteria. Neurotologic complications examined were hearing loss, tinnitus, dizziness, and hemifacial paresis. During postoperative visits patients were consistently asked about these conditions and cranial nerve exams were performed. Intraoperative brainstem auditory evoked potential monitoring and cranial nerve monitoring were not routinely used. P-values for the comparison of complication rates were calculated using a two-tailed fisher exact probability test.
RESULTS
| Age (n=183) | |
| Range | 28-92 y |
| Mean ± SD | 58.52 ± 14.36 y |
| Sex (n=183) | |
| Male | 52 |
| Female | 131 |
| MVD indication (n=200)a | |
| TN | 162 |
| HFS | 23 |
| GPN | 13 |
| Vagal palsy | 1 |
| Tinnitus | 1 |
| Follow-up duration (n=183) | |
| Range | 0.5-208 mos |
| Mean ± SD | 35.42 ± 47.21 mos |
| Side of operation (n=192) | |
| Left | 95 |
| Right | 97 |
| MVD revision operations | 22 |
Hearing loss
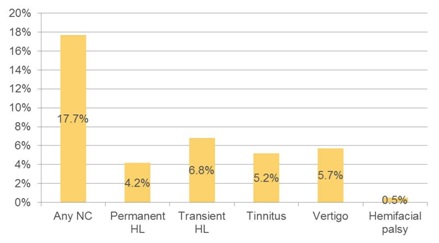
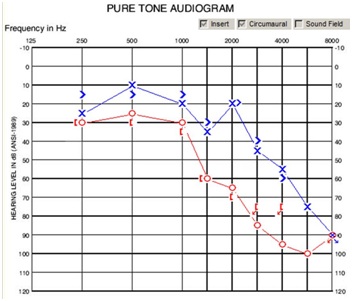
Figure 4: Audiogram revealing right sided sensorineural hearing loss in a patient who underwent microvascular decompression for trigeminal neuralgia three months previously.
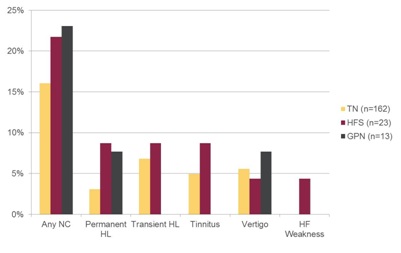
Figure 5: Frequency of neurotologic complications by indication.
NC=Neurotologic Complication; HL=Hearing Loss; HF=Hemifacial
Thirteen patients had transient hearing loss postoperatively (6.77%). The mean age for these patients was 55.31 years, with standard deviation 11.85 years and range 37-74 years. Nine were females and four were males. Indications for surgery were TN for 11 patients, HFS for one patient, and both TN and HFS for one patient. These all represented primary MVD. The consistent complaints for these patients were aural fullness or muffled hearing. All complaints resolved within four weeks and none of the patients underwent an audiogram.
There were 12 patients who had a documented hearing loss prior to surgery that remained stable postoperatively. Only five of these patients had preoperative audiograms in their medical record. The others denied worsening of their hearing loss postoperatively.
There was no significant difference in risk for permanent (Relative Risk (RR)=0.3395, p=0.1797) or transient (RR=1.4938, p=0.6938) hearing loss between TN and HFS. There was also no significant difference in risk of hearing loss between primary and revision surgery (permanent; RR=0.4373, p=0.5652, transient; RR=0.2754, p=0.3648).
Tinnitus
Vertigo
Hemifacial paresis
DISCUSSION
Following post-auricular operations, effusions can frequently evolve in the middle ear leading to CHL. Patients describe this as muffled hearing or aural fullness [16]. It is appropriate to wait at least two months postoperatively before pursuing an audiogram in order to allow a potential effusion to resolve. At that point, an audiogram should be performed with air and bone conduction thresholds. SNHL is far more likely to be permanent than CHL.
Injuries to the vestibulocochlear nerve or inner ear vasculature can also result in potentially disabling postoperative tinnitus. Tinnitus tends to correlate with hearing loss, and patients will often complain of the ringing more than their hearing loss [21,22]. Since tinnitus often resolves in conjunction with hearing loss, it is also appropriate to wait at least two months postoperatively before any intervention is pursued.
Patients may also experience vestibular dysfunction after CPA surgery, which may be temporary or permanent. In the short-term, the patient may suffer transient vertigo that prolongs the hospital stay [18]. In the long-term, the patient may note persistent imbalance, made worse with fatigue, alcohol consumption, and visual loss.
Hemifacial paresis is another complication that can occur after MVD. While considerably less common than the other neurotologic complications, it can be the most distressing to patients. Corticosteroid treatment is recommended in these patients, however their efficacy after MVD is unclear at this point [23,24].
There was no significant difference in complication rates between TN and HFS. It is logical to expect that HFS would have a higher complication rate given its closer proximity to both the CN VII/CN VIII bundle and the inner ear vasculature. It is possible that in a study with a larger sample size a significant difference would be revealed.
While some neurotologic complications are inevitable, all possible measures to minimize their occurrence and severity should be undertaken. This includes limiting cerebellar retraction, minimizing trauma to CN VII and VIII, and protecting vessels that are important for inner ear function [25]. Utilizing intraoperative monitoring of brainstem auditory evoked potentials has generally been shown to decrease the risk of hearing impairment after MVD [26,27].
To underscore the importance of appropriate preoperative audiometric documentation, twelve of our patients had written documentation of preoperative hearing loss, but only five had preoperative audiograms to quantify this loss. The lack of a preoperative audiogram for the other seven patients made it impossible to objectively assess whether their postoperative hearing loss was stable, worse, or improved. Insufficient perioperative audiometric evaluation in such cases leaves neurosurgeons vulnerable to medicolegal ramifications in situations where they may not have even caused a supposed complication. To prevent this, it is critical that perioperative audiograms are performed on a routine basis.
Counselling patients before surgery to ensure understanding of the neurotologic risks of undergoing MVD is critical. It is also important to monitor patients postoperatively for these complications with thorough histories and physical exams. We recommend an audiogram within six months before surgery and then another two to three months postoperatively in all patients.
CONCLUSION
REFERENCES
- Gaul C, Hastreiter P, Duncker A, Naraghi R (2011) Diagnosis and neurosurgical treatment of glossopharyngeal neuralgia: clinical findings and 3-D visualization of neurovascular compression in 19 consecutive patients. J Headache Pain 12: 527-534.
- Qi H, Zhang W, Zhang X, Zhao C (2016) Microvascular decompression surgery for hemifacial spasm. J Craniofac Surg 27: 124-127.
- Devor M, Amir R, Rappaport ZH (2002) Pathophysiology of trigeminal neuralgia: the ignition hypothesis. Clin J Pain 18: 4-13.
- Jannetta PJ (1967) Arterial compression of the trigeminal nerve at the pons in patients with trigeminal neuralgia. J Neurosurg 26: 159-162.
- Sarsam Z, Garcia-Fiñana M, Nurmikko TJ, Varma TRK, Eldridge P (2010) The long-term outcome of microvascular decompression for trigeminal neuralgia. Br J Neurosurg 24: 18-25.
- Hitchon PW, Zanaty M, Moritani T, Uc E, Pieper CL, et al. (2015) Microvascular decompression and MRI findings in trigeminal neuralgia and hemifacial spasm. A single center experience. Clin Neurol Neurosurg 139: 216-220.
- Zhang L, Yu Y, Yuan Y, Xu J, Xu X, et al. (2012) Microvascular decompression of cochleovestibular nerve in patients with tinnitus and vertigo. Neurol India 60: 495-497.
- McLaughlin MR, Jannetta PJ, Clyde BL, Subach BR, Comey CH, et al. (1990) Microvascular decompression of cranial nerves: lessons learned after 4400 operations. J Neurosurg 90: 1-8.
- Barker FG, Jannetta PJ, Bissonette DJ, Larkins MV, Jho HD (1999) The long-term outcome of microvascular decompression for trigeminal neuralgia. N Engl J Med 334: 1077-1083.
- Oesman C, Mooij JJ (2011) Long-Term Follow-Up of Microvascular Decompression for Trigeminal Neuralgia. Skull Base 21: 313-322.
- Jagannath PM, Venkataramana NK, Bansal A, Ravichandra M (2012) Outcome of microvascular decompression for trigeminal neuralgia using autologous muscle graft: A five-year prospective study. Asian J Neurosurg 7: 125-130.
- Burchiel KJ, Clarke H, Haglund M, Loeser JD (1988) Long-term efficacy of microvascular decompression in trigeminal neuralgia. J Neurosurg 69: 35-38.
- Huh R, Han IB, Moon JY, Chang JW, Chung SS (2008) Microvascular decompression for hemifacial spasm: analyses of operative complications in 1582 consecutive patients. Surg Neurol 69: 153-157.
- Park K, Hong SH, Hong SD, Cho YS, Chung WH, et al. (2009) Patterns of hearing loss after microvascular decompression for hemifacial spasm. J Neurol Neurosurg Psychiatry 80: 1165-1167.
- Lee MH, Lee HS, Jee TK, Jo KI, Kong DS, et al. (2015) Cerebellar retraction and hearing loss after microvascular decompression for hemifacial spasm. Acta Neurochir (Wien) 157: 337-343.
- Shah A, Nikonow T, Thirumala P, Hirsch B, Chang Y, et al. (2012) Hearing outcomes following microvascular decompression for hemifacial spasm. Clin Neurol Neurosurg 114: 673-677.
- [No authors listed] (1995) Committee on Hearing and Equilibrium guidelines for the evaluation of hearing preservation in acoustic neuroma (vestibular schwannoma). American Academy of Otolaryngology-Head and Neck Surgery Foundation, INC. Otolaryngol Head Neck Surg 113: 179-180.
- Heuser K, Kerty E, Eide PK, Cvancarova M, Dietrichs E (2007) Microvascular decompression for hemifacial spasm: postoperative neurologic follow-up and evaluation of life quality. Eur J Neurol 14: 335-340.
- Møller MB, Møller AR (1985) Loss of auditory function in microvascular decompression for hemifacial spasm. Results in 143 consecutive cases. J Neurosurg 63: 17-20.
- Chung SS, Chang JH, Choi JY, Chang JW, Park YG (2001) Microvascular decompression for hemifacial spasm: a long-term follow-up of 1,169 consecutive cases. Stereotact Funct Neurosurg 77: 190-193.
- Li D, Wang H, Fan Z, Fan Z (2010) Complications in retrosigmoid cranial nerve surgery. Acta Otolaryngol 130: 247-252.
- Youn J, Kwon S, Kim JS, Jeong H, Park K, et al. (2013) Safety and Effectiveness of Microvascular Decompression for the Treatment of Hemifacial Spasm in the Elderly. Eur Neurol 70: 165-171.
- Rhee DJ, Kong DS, Park K, Lee JA (2006) Frequency and prognosis of delayed facial palsy after microvascular decompression for hemifacial spasm. Acta Neurochir (Wien) 148: 839-843.
- Lee J, Fung K, Lownie SP, Parnes LS (2007) Assessing impairment and disability of facial paralysis in patients with vestibular schwannoma. Arch Otolaryngol Head Neck Surg 133: 56-60.
- Ying T, Thirumala P, Chang Y, Habeych M, Crammond D, et al. (2014) Emprical factors associated with Brainstem auditory evoked potential monitoring during microvascular decompression for hemifacial spasm and its correlation to hearing loss. Acta Neurochir 156: 571-575.
- Polo G, Fischer C, Sindou MP, Marneffe V (2004) Brainstem auditory evoked potential monitoring during microvascular decompression for hemifacial spasm: intraoperative brainstem auditory evoked potential changes and warning values to prevent hearing loss--prospective study in a consecutive series of 84 patients. Neurosurgery 54: 97-104.
- Joo BE, Park SK, Cho KR, Kong DS, Seo DW, et al. (2016) Real-time intraoperative monitoring of brainstem auditory evoked potentials during microvascular decompression for hemifacial spasm. J Neurosurg 125: 1061-1067.
Citation: Genno Karam N, Rima BT, Sawsan N, Nasseh I, Zeinoun T (2014) Focal Cemento-Osseous Dysplasia: A Case Report. J Clin Stud Med Case Rep 1: 16.
Copyright: © 2017 Matthew Bartindale, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

