
Perceptions of Breast Cancer Survivors Participating In a Chinese Herbal Medicine Study: An Exploratory Narrative Study
*Corresponding Author(s):
Xiaoshu ZhuSchool Of Science And Health, Western Sydney University, Sydney, Australia
Abstract
Objective
Survivors report Hot Flushes/Night Sweats (HF/NS) as the most debilitating side-effect of breast cancer endocrine therapy with limited treatment options. The aim of the investigation was to gather information, not available in quantitative outcome measures, including narrative data of women’s experiences of HF/NS and personal experience of participating in a Chinese Herbal Medicine (CHM) trial to alleviate this bothersome side-effect. This is an important pilot feasibility study to evaluate the potential benefit of trialling a novel Chinese medicinal product for a symptom of HF/NS identified as an unmet need for breast cancer survivors and may help inform future studies.
Participants and Methods
Semi-structured focus groups were conducted with 8 breast cancer survivors with hot flush symptoms. All women had been randomised to participate in a CHM clinical trial for alleviation of HF/NS. A discussion guide was employed to provide insights regarding symptom experience, hot flush impact on quality of life, and qualitative data related to the acceptability and feasibility of CHM as a management option for HF/NS. Digitally recorded narratives were analysed using thematic analysis.
Results
Our analysis confirms CHM acceptability is complex. The taste was deemed “not pleasant”, however, the driver of hot flush reduction helped maintain adherence. Additionally, after completing their primary care participants reported a feeling of insecurity and not knowing where to turn for help with side-effects of treatment. These findings reveal that survivorship has its challenges and can impact quality-of-life with psychosocial and physiological implications long-term. Categories identified included ‘give it a go’, ‘avoidance and acceptance’, ‘routine and reward’, ‘transitioning to survivorship’.
Conclusion
Focus group findings report CHM through the hospital setting provided an additional safety measure. Drivers for acceptability of CHM included commitment to the trial, the belief that it would work, taking a more active role in their well-being and feeling valued as a participant in the trial. Other themes include opportunities for better transition to survivorship and management of adverse effects.
Keywords
ABBREVIATIONS
CHM - Chinese Herbal Medicine
CIM - Complementary and Integrative Medicine
HF/NS - Hot Flushes/Night Sweats
OTC - Over-The-Counter
QoL - Quality of Life
INTRODUCTION
METHODS
Ethics and recruitment
Participants
Demographic and clinical information for participants was collected at the beginning of the clinical trial. The women were aged from 46 to 64 years of age and diagnosed with clinical Stage I to III breast cancer and had completed their primary care (surgery, chemotherapy and radiation). All women participating in the focus groups were prescribed adjuvant hormonal therapy for Hormone Receptor (HR) positive primary breast cancer. At the time of diagnosis 1 was pre-menopausal, 4 were peri-menopausal and 3 were post-menopausal. All participants were given written and verbal information about the study and written informed consent for the focus group was obtained at the same time as consent for the clinical trial.
Focus group interviews
The interview prompts are presented in Table 1.
|
S. No |
Discussion Guide |
|
1 |
What was your initial reaction when CHM was suggested by your oncologist > Did you have any concerns > if yes what were they? |
|
2 |
Would you have used herbal medicine from another source > i.e. not suggested by oncologist at hospital but over-the-counter, something on the internet, suggestion from family/friend or CAM practitioner? |
|
3 |
What were your main reasons for joining the study? |
|
4 |
How did you find the taste and smell of the intervention? Did they smell and taste similar? |
|
5 |
Did you feel a level of safety taking the intervention through a hospital setting with pathology tests and oncologist support > any other comments? |
|
6 |
Tell me about your hot flushes/night sweats, what do you experience when you are having a hot flush/night sweat? |
|
7 |
What strategies do you use when you are having a hot flush/night sweat? |
|
8 |
Do you feel that exercise, stress or food/drinks you ingest may influence hot flushes i.e. coffee, tea, alcohol, spicy foods and if so, in what way? |
|
9 |
Did the intervention help with the hot flushes/night sweats> if so in what way> have any changes lasted? |
|
10 |
Do you wish to continue with the intervention now that the study period is over> if yes what factors would influence your use of CHM for hot flushes?> if no why not? |
This type of semi-structured interview employs a guiding theme with probing open-ended sub-questions but allows participants to pursue or respond in their own manner allowing for the discovery of new information [22]. The moderator had extensive knowledge of the topic under discussion and developed an interview guide to foster dialogue relevant to the study aims (Table 1). The research aims were based on a review of the literature reporting on the needs and experiences of women previously diagnosed with breast cancer. These aims are presented below:
The focus groups aim to:
• To explore the trial participants’ perceptions of VMS and their impact on their life
• To explore how their oncologist’s involvement may have affected their participation in the study
• To explore how the trial participants perceived the intervention
• To identify why some trial participants withdrew from the study
• To explore the trial participants perceptions of benefits or not from the intervention
• To identify what women diagnosed with breast cancer perceive as barriers or facilitators to their healthcare needs
• To explore what information breast cancer survivors would like to have available about CIM and how they would like this information disseminated
Analytical methods
From this, over-arching categories and repetitive concepts were extracted, compared and connected [22,36,37]. Common themes were identified and constantly reviewed to check that developing philosophical assumptions were correct, simultaneously linking them to my general reflections. The emerging categories were integrated into themes and labelled. The process was an iterative process, transcripts were read and re-read to confirm and refine presenting themes, collating and comparing between presenting concepts [22,36]. The second author (XZ) read the transcripts and independently reviewed the findings to ascertain validity of identified concepts and themes. Both authors (DP) and (XZ) collaborated, discussed and developed the themes and subthemes to ensure rigour.
RESULTS
Perspectives of cancer survivors
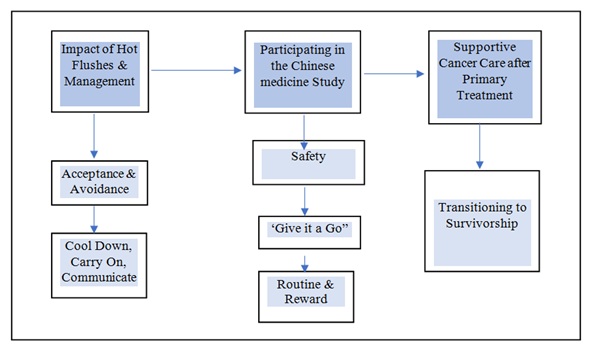 Figure 1: Diagram of themes and categories.
Figure 1: Diagram of themes and categories.Theme 1: Impact of hot flushes and their management
“Acceptance and Avoidance”
The over-arching theme of hot flush impact and management contained two subthemes: “cool-down, carry-on and communicate” and “acceptance and avoidance” - the women learnt to accept their unique situation, however, they employed strategies to help keep cooler by avoidance of triggers for hot flushes.
Mostly the women had learned to employ practical strategies to help with management and impact of hot flushes. These included wearing layers of clothing so that they could remove them as needed, carrying a fan or using electric fans or air-conditioning as much as possible. Some communicated with their work colleagues and friends about the hot flushes to help reduce embarrassment in these situations. At night they threw covers off and had cool showers or went outside to cool down. Most of the women avoided alcohol and coffee as they triggered hot flushes and exercise was also avoided due to the added impact of feeling hot and sweaty. In summary, the women adopted many approaches for coping with debilitating hot flushes including a change of attitude to acceptance and self-help strategies. Some of these strategies resulted in avoidance of exercise and dietary triggers. Hot flushes affected leisure activities due to their impact on appearance and mood. Disrupted sleep from hot flushes/night sweats resulted in fatigue which also impacted their social engagements.
“Jacket on, jacket off, people hand me paper towels and tissues. I don’t go red -
“I just get really hot and perspire” (n=1)
“It interferes with sleep, so throwing off covers and getting up continuously - interrupted sleep. You do feel tired the next day though” (n=8)
“I have air-conditioning going on and off all night” (n=5)
“I know that if I have coffee or wine that it is going to hit me. I tend to avoid going outwith friends as it goes hand in hand. Even with exercise - ten minutes later I know they (hot flushes) are going to hit me. I get sweaty and hot, so I avoid it. I don’t even want to go out, after 8 o’clock I am just too fatigued” (n=1)
“I have a little portable fan on my desk at work. I have told people about my problem and they turn it on for me. I don’t wear make-up as it just melts off -that sort of impact on how you present yourself” (n=1)
Theme 2: Participating in the Chinese herbal medicine study
“Safety”
“Routine and Reward”
The women decided to join the Chinese medicine study and “give it a go” in the hope that trying something new may reduce their hot flushes/night sweats. Personal motive was the main reason as they expressed desperation in their desire to reduce the unwanted side-effect. Many did not wish to take another pharmaceutical due to perceived or potential side-effects. The women expressed safety concerns about using over-the-counter remedies for menopausal symptoms and felt reassured of the safety of the CHM as it was referred by their oncologist, conducted at the hospital and they underwent regular pathology tests during the study.
The taste of the intervention was a barrier to acceptability. The CHM and placebo provided in the study was in powder format to be mixed with warm water and taken orally as a tea, twice daily. Most of the participants disliked the taste of the CHM/placebo (two withdrew from the study as they could not tolerate the taste; others diluted the powder with juice or gave themselves a reward afterwards). Participants found the appearance and smell of the intervention and placebo were similar, but one tasted more bitter than the other. Some women expressed the desire for the intervention to be available in capsule format. Other barriers that may influence long-term use, outside of the trial, were potential financial cost, quality and safety of the intervention and ease of availability.
The sub-theme of “routine and reward” also emerged from the analysis. Despite the unpalatable taste of the intervention some of the participants reported they started to look forward to the routine of taking the intervention twice daily - this was the first identified facilitator of acceptability. The second facilitator of acceptability included participants stateing a reduction in hot flush severity and wished to continue taking the intervention, with one participant also reporting a reduction in stress levels. This reduction in symptoms was perceived as a reward. Commitment to the trial, the belief that it would work, taking a more active role in their well-being and feeling valued as a participant in the trial where found to be mediators of acceptability.
Five of the women cited “desperate” as their primary motive (n=5)
“anything is better than nothing, lots of people say it (Chinese herbal medicine) works for a variety of things, so yes, willing to give it a go” (n=5)
“I was open to it, accepting of anything; give it a shot, why not?” (n=2)
“My doctor said it is either ‘Effexor’ (an anti-depressant) or this (Chinese herbal medicine trial). I didn’t want to take another prescription drug, so I thought - I’ll give it a go” (n=1)
“I felt a level of safety in general because of the hospital setting” (n=8)
“I felt safe having the pathology tests at different intervals” (n=6)
“The first one was terrible. I tried it with juice and then chocolate milk and that seemed to work. It was so bitter. The taste was different in the second lot, though they smelt and looked the same” (n=1)
“I felt I started to look for it as it started to become part of my daily routine. I don’t drink tea or coffee so it was herb time” (n=1) “I felt it was my morning and afternoon ritual. Though I feel if it is successful that it should be compounded into a capsule so that it would be easier to take or perhaps enhance the flavour” (n=1)
“I felt it was not unpleasant to the point the gagging, but I rewarded myself afterwards with a piece of chocolate” (n=1)
“Cost would definitely influence my use” (n=4)
“Yes, cost would be a factor, $20 per week would be ok, otherwise for long-term use it would be difficult to pay more” (n=3)
“I would like it to be easily available and to know that what you are getting is safe, tested and best quality” (n=4)
Theme 3: Supportive Cancer Care after Primary Treatment
“excellent support during and just after diagnosis and acute treatment. However, if you have any issues down the track it is hard to get help. I saw my GP and went back to see the oncologist. I needed answers - you do feel a bit cut off once treatment is finished. My GP didn’t know a lot about after-effects of radiation” (n=1)
“the doctors do a fantastic job, but once acute treatment is over you feel a bit abandoned. I understand that they are busy and other patients are needing their time, but side-effects are dismissed as part of the treatment.You don’t get much help or advice then” (n=4)
“there is no discussion about what you are doing e.g., if you are taking vitamins, no advice on diet even with the breast cancer nurse” (n=5).
“Yes, more information about alternative therapies would be useful. I wasn’t game to try any other remedies due to safety concerns” (n=4).
The categories and themes are presented in table 2.
|
|
Main Themes Identified |
|
Chinese herbal medicine decision-making experience |
“Give it a go - anything is better than nothing” |
|
Reasons for joining study |
“felt safe as it was through hospital” |
|
Hot flushes/night sweats - impact on QoL |
Sleep, anxiety regarding social relationships and work, appearance, exercise, levels of fatigue were all negatively influenced. Feelings ranged from an inability to cope with the distress to acceptance of the side-effect or avoidance of triggers |
|
Taking control - Self-care strategies Intervention - taste |
Cool down, carry on and communicate |
|
- outcome |
Some participants were not sure if the intervention helped in either arm of the study, others felt severity of hot flushes and night sweats was reduced. One participant said stress levels felt less. |
|
- continuation |
Ease of availability and cost may prevent long-term usage |
|
Burden of attending study visits |
No participant found it a burden to attend or fill out the forms |
|
Seeking treatment information |
Transitioning to survivorship –Participants felt well-supported during acute stage of BrCa treatment; but felt more support should be provided for side-effects including hot flush management. Transitioning into survivorship - identified a need for a survivorship plan |
|
- information on CIM |
Participants felt that it would be valuable to have evidence-based information regarding CIM treatments after BrCa, particularly relating to alleviation of side-effects. |
DISCUSSION
With increased survivorship after breast cancer the need for effective management of symptoms and side-effects is important as quality of life is a major objective in oncology care [38,39]. Using in-depth focus group techniques this study explored the lived experiences of breast cancer survivors after participating in a CHM medicine study for managing HF/NS and explored the impact of HF/NS on everyday life. HF/NS are the most reported and the most debilitating side-effect of treatment after breast cancer and there is a major gap in their management after a diagnosis [7,10,11,40-42]. Many survivors do not want to take another pharmaceutical due to potential side-effects and Hormone Replacement Therapy is contra-indicated due to risk of recurrence [43].
The first research aim was to explore how HF/NS impacted daily life. All participants reported the experience was stressful and impacted on sleep and how their work and social lives were changed. The women described how they learned to employ practical strategies to deal with management of HF/NS as outlined in the narratives above. Although they reluctantly had to accept HF/NS as part of their life, they avoided exercise and social engagements due to how HF/NS impacted on appearance and mood. Disrupted sleep also resulted in fatigue and impacted their ability to attend social engagements. Similar to other studies the findings have highlighted the negative impact that HF/NS have on daily QoL of breast cancer survivors [2,44,45].
Participants were asked if their oncologist’s involvement helped in their decision to participate in the study. Although the women were struggling with their HF/NS they did not wish to take another pharmaceutical but were also reluctant to take over-the-counter remedies due to safety concerns after their diagnosis. The referral from their oncologist, with the study being conducted in a hospital including regular pathology tests, helped alleviate any participant concerns about the safety of the Chinese herbal medicine.
Another aim of the study was to identify what breast cancer survivors perceive as barriers or facilitators to their healthcare needs. An important theme uncovered during the study included the ‘lived experience’ of cancer survivors as they transition from patient to survivor [46,47]. They were living with their diagnosis daily but desired to have a better quality of life [48,49]. The women expressed concerns regarding unmet needs for treatment side-effects, their impact on daily quality of life and who to turn to for advice.
The women valued their relationship with their oncologist and satisfaction with their acute primary care, however they expressed concern and frustration about the transition process post-treatment and the need for more support for physical and emotional side-effects [50]. Their perspectives included confidence in their medical treatment but a feeling of being overwhelmed and vulnerable as the intense primary treatment period ceased. It has been previously reported that patients can transfer the burden of medical decision-making to their physician during the treatment period as they learn to accept their diagnosis [51]. At conclusion of primary care, the women in this study reported feeling a bit lost, having developed a dependency on the medical staff and a routine of sometimes daily visits to the hospital. Other studies have reported similar findings, with patients feeling as if their safety net had been taken away as they finish primary treatment and try to come to terms with their new reality [52-54].
The findings may provide guidance for healthcare policy through added support for addressing treatment side-effects and provision of survivorship care planning with evidence-based information. It was clear from the stories shared during the focus groups that survivorship, including learning how to live with side-effects and the process of transitioning from patient to survivor, required additional support after the acute phase into recovery. Additional sources of support (medical and non-medical) may help with co-ordination of care and a smoother transition. Similar plans have been studied and implemented in other countries to reduce impact on physical and psycho-social well-being during this period [55-58].
The final and main aim of the focus groups was to inform regarding acceptability of the intervention and perceived benefits and/or burden of participating in the trial. Our findings revealed that the unpalatable taste of the powder herbal mixture was a problem. The clinical trial did have some withdrawals due to the taste of the intervention. In principle, the taste of the intervention was not acceptable, however, participants continued for a variety of reasons. These mediators of acceptability included commitment to the study, reduction in HF/NS, confidence in the safety of the intervention through the hospital setting, taking a more active role in their well-being and feeling valued as a participant in the trial.
Future studies may need to focus on feasibility and acceptability, perhaps offering the intervention in two forms - powder and encapsulated - to help maintain participation. Many of the participants desired to continue with the CHM after the study concluded, as they found a reduction in the severity of their HF/NS, but cited cost as the major obstacle with ease of availability and quality control as additional concerns. None of the participants found it a burden to maintain the study visits every 4 weeks and wished to honour their commitment to the completion of the study. These findings provided an insight into the acceptability of the intervention which may help inform the design of future Chinese herbal medicine studies regarding feasibility, acceptability and compliance.
Five main narrative themes emerged from the data - ‘give it a go’, ‘acceptance and avoidance’, ‘cool down, carry on and communicate’, ‘routine and reward’ and ‘transitioning to survivorship”.
STRENGTHS AND LIMITATIONS OF THE STUDY
CONCLUSION
CONFLICT OF INTEREST STATEMENT
FINANCIAL SUPPORT
ETHICAL APPROVAL
ACKNOWLEDGEMENT
AUTHOR CONTRIBUTIONS
REFERENCES
- National Cancer Control Indicators (2019) Mortality-to-incidence ratio. National Cancer Control Indicators, Australia.
- Tiezzi MF, de Andrade JM, Romão AP, Tiezzi DG, Lerri MR, et al. (2017) Quality of Life in Women With Breast Cancer Treated With or Without Chemotherapy. Cancer Nurs 40: 108-116.
- Rustøen T, Begnum S (2000) Quality of life in women with breast cancer: A review of the literature and implications for nursing practice. Cancer Nurs 23: 416-421.
- Stein KD, Jacobsen PB, Hann DM, Greenberg H, Lyman G (2000) Impact of Hot Flashes on Quality of Life Among Postmenopausal Women Being Treated for Breast Cancer. J Pain Symptom Manage 19: 436-445.
- Fisher WI, Johnson AK, Elkins GR, Otte JL, Burns DS, et al. (2013) Risk factors, pathophysiology, and treatment of hot flashes in cancer. CA Cancer J Clin 63: 167-192.
- Cella D, Fallowfield LJ (2008) Recognition and management of treatment-related side effects for breast cancer patients receiving adjuvant endocrine therapy. Breast Cancer Res Treat 107: 167-180.
- Adelson KB, Loprinzi CL, Hershman DL (2005) Treatment of hot flushes in breast and prostate cancer. Expert Opin Pharmacother 6: 1095-1106.
- Carpenter JS, Andrykowski MA, Cordova M, Cunningham L, Studts J, et al. (1998) Hot flashes in postmenopausal women treated for breast carcinoma: prevalence, severity, correlates, management, and relation to quality of life. Cancer 82: 1682-1691.
- Harris PF, Remington PL, Trentham-Dietz A, Allen CI, Newcomb PA (2002) Prevalence and Treatment of Menopausal Symptoms Among Breast Cancer Survivors. J Pain Symptom Manage 23: 501-509.
- Partridge AH, Wang PS, Winer EP, Avorn J (2003) Nonadherence to adjuvant tamoxifen therapy in women with primary breast cancer. J Clin Oncol 21: 602-606.
- Partridge AH (2006) Non-adherence to endocrine therapy for breast cancer. Ann Oncol 17: 183-184.
- Davies C, Pan H, Godwin J, Gray R, Arriagada R, et al. (2013) Long-term effects of continuing adjuvant tamoxifen to 10 years versus stopping at 5 years after diagnosis of oestrogen receptor-positive breast cancer: ATLAS, a randomised trial. Lancet 381: 805-816.
- Bordeleau L, Pritchard K, Goodwin P, Loprinzi C (2007) Therapeutic options for the management of hot flashes in breast cancer survivors: an evidence-based review. Clin Ther 29: 230-241.
- Amir E, Seruga B, Niraula S, Carlsson L, Ocaña A (2011) Toxicity of Adjuvant Endocrine Therapy in Postmenopausal Breast Cancer Patients: A Systematic Review and Meta-analysis. J Natl Cancer Inst 103: 1299-1309.
- Gupta P, Sturdee DW, Palin SL, Majumder K, Fear R, et al. (2006) Menopausal symptoms in women treated for breast cancer: the prevalence and severity of symptoms and their perceived effects on quality of life. Climacteric 9: 49-58.
- Kremser T, Evans A, Moore A, Luxford K, Begbie S, et al. (2008) Use of complementary therapies by Australian women with breast cancer. Breast 17: 387-394.
- Tautz E, Momm F, Hasenburg A, Guethlin C (2012) Use of Complementary and Alternative Medicine in breast cancer patients and their experiences: A cross-sectional study. Eur J Cancer 48: 3133-3139.
- Antoine C, Liebens F, Carly B, Pastijn A, Rozenberg S (2007) Safety of alternative treatments for menopausal symptoms after breast cancer: a qualitative systematic review. Climacteric 10: 23-26.
- Porter DP, Zhu X, Bensoussan A, De Souza P (2019) The Safety and Efficacy of a Chinese Herbal Medicine to Help Alleviate Vasomotor Symptoms in Breast Cancer Survvivors. Health Research Institute, Western Sydney University, Sydney, Australia.
- Morse JM (2000) Determining Sample Size. Qualitative Health Research 10: 3-5.
- Sandelowski M (1995) Sample size in qualitative research. Research in Nursing & Health 18: 179-183.
- Broom A (2005) Using qualitative interviews in CAM research: A guide to study design, data collection and data analysis. Complement Ther Med 13: 65-73.
- Kitzinger J (1994) The methodology of Focus Groups: the importance of interaction between research participants. Sociology of Health & Illness 16: 103-121.
- Glaser BG, Strauss AL (1967) The Discovery of Grounded Theory: Strategies for Qualitative Research. Aldine Publishing Company Pg no: 271.
- Heath J, Williamson H, Williams L, Harcourt D (2018) “It's just more personal”: Using multiple methods of qualitative data collection to facilitate participation in research focusing on sensitive subjects. Appl Nurs Res 43: 30-35.
- Bazeley P (2013) Qualitative data analysis : practical strategies. SAGE Publications Ltd Pg no: 472.
- Charmaz K (2016) Constructivist grounded theory. The Journal of Positive Psychology 12: 299-300.
- Charmaz K (2017) The Power of Constructivist Grounded Theory for Critical Inquiry. Qualitative Inquiry 23: 34-45.
- Primeau LA (2003) Reflections on self in qualitative research: stories of family. Am J Occup Ther 57: 9-16.
- Charmaz K, Kathy Charmaz (2017) Discusses Constructivist Grounded Theory. SAGE, London, UK.
- Chesney M (2000)Interaction and understanding: 'me' in the research Nurse Researcher 7: 58-69.
- Guillemin M, Gillam L (2004)Ethics, Reflexivity, and “Ethically Important Moments” in Research 10: 261-280.
- Eppley K (2006)Review Essay: Defying Insider-Outsider Categorization: One Researcher's Fluid and Complicated Positioning on the Insider-Outsider Continuum. Forum Qualitative Social Research7: 1-9.
- Harding S (1991)Whose Science? Whose Knowledge?: Thinking from Women's Lives.Cornell University Press, New York, United States.
- Berger R (2015) Now I see it, now I don’t: Researcher’s position and reflexivity in qualitative research. Qualitative research15: 219-234.
- Charmaz K (2006) Constructing grounded theory : a practical guide through qualitative analysis. SAGE Publications, California, USA.
- Freeman T (2006) 'Best practice' in focus group research: making sense of different views.J Adv Nurs 56: 491-497.
- Ganz P (2008) Psychological and social aspects of breast cancer. Oncology (Williston Park) 22: 642-650.
- DuMontier C, Clough-Gorr KM, Silliman RA, Stuck AE, Moser A (2018) Health-Related Quality of Life in a Predictive Model for Mortality in Older Breast Cancer Survivors.J Am Geriatr Soc 66: 1115-1122.
- Love RR, Cameron L, Connell BL, Leventhal H (1991) Symptoms associated with tamoxifen treatment in postmenopausal women.Arch Intern Med 151: 1842-1847.
- Loprinzi CL, Zahasky KM, Sloan JA, Novotny PJ, Quella SK (2000) Tamoxifen-induced hot flashes.Clin Breast Cancer 1: 52-56.
- Loprinzi CL, Barton DL, Rhodes D (2001)Management of hot flashes in breast-cancer survivors. Lancet Oncology2: 199-204.
- Beral V (2003) Breast cancer and hormone-replacement therapy in the Million Women Study. The Lancet362: 419-427.
- White A (2013) Hot Flushes in Patients with Breast Cancer. Acupuncture in Medicine31: 341-341.
- Stearns V, Chapman JA, Ma CX, Ellis MJ, Ingle JN, et al. (2015) Treatment-associated musculoskeletal and vasomotor symptoms and relapse-free survival in the NCIC CTG MA.27 adjuvant breast cancer aromatase inhibitor trial.J Clin Oncol 33: 265-271.
- van Londen G, Beckjord E, Dew M, Cuijpers P, Tadic S, et al. (2013) Breast cancer survivorship symptom management: current perspective and future development. Breast Cancer Manag 2:71-81.
- Burke NJ, Napoles TM, Banks PJ, Orenstein FS, Luce JA, et al. (2016) Survivorship Care Plan Information Needs: Perspectives of Safety-Net Breast Cancer Patients. PLoS One11: 0168383.
- Lee Mortensen G, Madsen IB, Krogsgaard R, Ejlertsen B (2018) Quality of life and care needs in women with estrogen positive metastatic breast cancer: a qualitative study. Acta Oncol57: 146-151.
- Aranda S, Schofield P, Weih L, Yates P, Milne D, et al. (2005) Mapping the quality of life and unmet needs of urban women with metastatic breast cancer.Eur J Cancer Care (Engl) 14: 211-222.
- Moore HCF, Ali MKM, Moeller M, Rybicki LA (2016) Timing of the survivorship visit and symptom prevalence in breast cancer survivors. JCO 34: 125-125.
- Blumenthal-Barby JS (2017) ‘That’s the doctor’s job’: Overcoming patient reluctance to be involved in medical decision making. Patient Education and Counseling 100: 14-17.
- Ganz PA, Kwan L, Stanton AL, Krupnick JL, Rowland JH, et al. (2004) Quality of Life at the End of Primary Treatment of Breast Cancer: First Results From the Moving Beyond Cancer Randomized Trial. J Natl Cancer Inst 96: 376-387.
- Knobf MT (2015) The Transition Experience to Breast Cancer Survivorship. Semin Oncol Nurs 31: 178-182.
- Pauwels EE, Charlier C, De Bourdeaudhuij I, Lechner L, Van Hoof E (2013) Care needs after primary breast cancer treatment. Survivors' associated sociodemographic and medical characteristics. Psychooncology 22: 125-132.
- Sutherby CM (2017) Growing a cancer survivorship care plan program. JCO 35: 58-58.
- Grunfeld E, Levine MN, Julian JA, Pond GR, Maunsell E, et al. (2011) Results of a multicenter randomized trial to evaluate a survivorship care plan for breast cancer survivors. JCO 29: 9005-9005.
- Coyle D, Grunfeld E, Coyle K, Pond G, Julian JA, et al. (2011) Cost effectiveness of a survivorship care plan for breast cancer survivors. JCO 29: 6082-6082.
- Alfano CM, Smith T, de Moor JS, Glasgow RE, Khoury MJ, et al. (2014) An Action Plan for Translating Cancer Survivorship Research Into Care. J Natl Cancer Inst 106.
Citation: Porter D, Zhu X, Bensoussan A, Souza PD (2019) Perceptions of Breast Cancer Survivors Participating In a Chinese Herbal Medicine Study: An Exploratory Narrative Study. J Altern Complement Integr Med 5: 069.
Copyright: © 2019 Dianna Porter, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

