
Primary Endonasal Endoscopic Powered Dacryocystorhinostomy for Low Obstruction of the Lacrimal Excretory System in Adult Patients: A 5-Years Experience
*Corresponding Author(s):
Eloy PDepartment Of Ent, CHU UCL Namur, Site De Godinne, Avenue Thérasse, 1, 5530, Yvoir, Belgium
Tel:+32 81423711,
Email:philippe.eloy@uclouvain.be
Abstract
Purpose: The aim of this retrospective study was to describe the outcomes of powered endonasal endoscopic dacryocystorhinostomy (DCR) performed for symptomatic low lachrymal obstruction in adult patients over a 5-year period by the same surgeon.
Methods: Retrospective study.
Patients: Patients suffering from a low obstruction of the lachrymal system documented by paraclinical investigations treated in the ENT department of CHU UCL Namur between September 2011 and September 2016.
Results: In total, 128 procedures were performed for 111 patients. The mean age at the time of surgery was 62.3 years (range: 18.1-93.0 years). There were 93 women and 18 men, and the mean follow-up duration was 40.5 months (range: 12.1-54.47 months). Aetiologies for the lacrimal obstruction included primary acquired nasolacrimal duct obstruction (PANDO; n=123), LeFort fractures (n=3), and radiotherapy/chemotherapy (n=2). Epiphora only (n=98), chronic purulent discharge (n=30), and acute dacryocystitis (n=4) were major symptoms. The stoma remained patent in115 of the 128 (89.8%) procedures, while111 (86.7%) procedures resulted in complete symptom resolution. Persistence of tearing without infection was observed after 17 (13.3%) procedures, with revision surgery performed in 13(10.1%) cases.
Conclusion: Endoscopic endonasal powered DCR is an effective surgery for primary saccal and post-saccal lachrymal obstruction in adults. The success rate is high and very competitive to that after an external approach. Powered instrumentation is a nice tool to make the resection of the bony part of the frontal process of the maxillary easier and quicker particularly in case of very thick bone. The postoperative morbidity is very low; The rehabilitation is fast. Septoplasty or sinus surgeries can be performed in the same operating setting when required with no deleterious impact on the success rate of DCR.
Keywords
Adult patients; Case series; Lower lachrymal pathway obstruction; Powered dacryocystorhinostomy
INTRODUCTION
Dacryocystorhinostomy (DCR) is a well-known procedure performed for the treatment of saccal and post-saccal obstruction refractory to medical treatment [1,2]. This type of obstruction occurs mainly after menopause in middle-aged women. In most cases, it is idiopathic, and the condition is commonly called as PANDO (Primary Acquired Nasolacrimal Duct Obstruction) by the ophthalmologists. On the other hand, SANDO (Secondary Acquired Nasolacrimal Duct Obstruction) has several etiologies divided into five categories: infectious, inflammatory, neoplastic, traumatic, and mechanical [3-5].
The aim of DCR is to create a communication between the lachrymal sac and nasal fossa through a rhinostomy performed at the level of the lacrimal eminence. The endonasal procedure was first described by Caldwell in 1893 [6]. However, it was rapidly abandoned because of the lack of appropriate visualization. In 1904, Toti proposed the external approach with a high rate of success, and it was considered the gold standard for many years [7]. After the 80s, however, clinicians observed newinterest in the endonasal approach because of the development of new instruments specific to endonasal surgery [8-10]. Here we describe our 5-year experience with primary powered endoscopic endonasal DCR performed for low lacrimal obstruction in adult patients by the same surgeon. All the patients have been informed about the procedure and the risks.
STUDY AND METHODS
We retrospectively reviewed the medical records of patients who underwent powered DCR for low lacrimal obstruction in the ENT department of CHU UCL Namur between September 2011 and September 2016. All included procedures were performed by the same surgeon, and all patients exhibited symptomatic low lacrimal pathway obstruction that was confirmed by complete paraclinical assessments. The diagnostic workup included complete ophthalmological examination, lachrymal syringing, nasal endoscopy, and assessment using a dacryoscanner. Magnetic Resonance Imaging (MRI) was additionally performed in some cases. All the procedure performed in this study involving human participants were in accordance with the ethical standards of the institutional research committee (NUB: B039201941453) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. No informed consent was waived because of the retrospective nature of the study.
The procedure was performed under general anesthesia with orotracheal intubation. Neurosurgical pledgets soaked with xylocaine 5% plus adrenaline were placed for 15 min before initiation of the surgery. In addition, 5 mL of xylocaine 1% plus adrenaline was infiltrated at the heads of the inferior and middle turbinates and the lacrimal eminence (Figure 1A). The inferior turbinate was lateralized and the middle turbinate was medialized using a Cottle elevator (Figure 1B). The mucosa just in front of the lacrimal eminence was vertically incised using a monocautery device (Figure 1C). The incision began at the level of the axilla of the middle turbinate and extended downward to the superior edge of the inferior turbinate. A nasal mucosal flap was elevated using the Freer elevator in the subperiosteal plane and removed. Vertical uncinectomy was performed when the uncinate process was attached to the posterior edge of the frontal process of the maxilla. The Agger nasi cell was identified in order to determine the superior limit of the surgical field. A Kerrisson forceps was used to begin resection of the bony inferior portion of the frontal process of the maxilla (Figure 1D). When the bone became too thick, a curved diamond drill (Medtronic Xomed 2.5 mm, 15°) was used to drill the bone up to the axilla (Figure 1E). Once drilling was complete, the mucosa of the nasolacrimal duct and lacrimal sac was exposed. A Bowman stent was used to probe the canalicular system. It was pushed toward the lacrimal sac for tenting of the medial wall. We infiltrated the lumen of the nasolacrimal duct with xylocaine and incised the mucosa of the lacrimal system from inferior to superior using a sickle knife (Figure 1F).At this time, extrusion of pus or xylocaine confirmed identification of the lacrimal pathway lumen. Then, the lacrimal mucosa was pushed backwards. No suture or metallic clips were necessary for securing the lachrymal flap posteriorly.
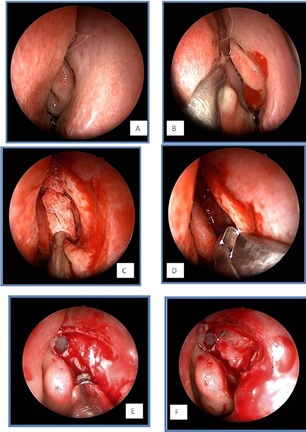
Figures 1(A-F): Surgical technique for primary powered endoscopic dacryocystorhinostomy, A. Endoscopic visualization of the left nasal fossa, B. Visualization of the middle meatus after displacement of the nasal septum, C. Exposure of the frontal process of the maxilla, D. Resection of the inferior portion of the frontal process using Kerrisson forceps, E. Drilling of the frontal process and visualization of the Agger nasi cell, F. Opening of the lacrimal pathway. Lachrymal flap with posterior attachment. Visualization of the tip of the Bowman probe.
At the end of the procedure, the lachrymal system was flushed with saline for confirmation of a patent stoma and exclusion of any canalicular obstruction. Punctoplasty was performed when the lower lachrymal punctum appeared stenotic. In all but one case a silicone tube was placed. Nasal packing was not required in most cases.
POSTOPERATIVE CARE
After surgery, the patients were instructed to clean their nasal cavities with nasal douching at least four times a day.In addition, all patients except those with glaucoma were prescribed eyedrops containing antibiotics and corticosteroidsthree times a day for 10 days. Patients with glaucoma used only antibiotic eye drops.
The first postoperative consultation was scheduled on day 10. All crusts were removed under endoscopic guidance where necessary. A topical nasal spray (momethasone) was prescribed until the second postoperative consultation (approximately 2 weeks later). The silicone tube was removed once crusting was no longer observed (approximately 4 to 6 weeks after surgery). Thereafter, all patients were followed up every 3 months during the first year after surgery.
RESULTS
In total, 128 procedures performed in 111 patients were evaluated. There were 17 bilateral procedures, and 74 (57.8%) and 54 (42.2%) were performed on the left and right sides, respectively. There were 93 women and 18 men with a mean age of 62.3 years (range: 18.1-93.0 years) at the time of surgery. The mean follow-up duration was 40.5 months (range: 12.1-54.47 months; Table 1).
Aetiologies of the lachryimal obstruction were listed in the table 1. Major symptoms included epiphora (n=98), chronic purulent discharge (n = 30), and acute dacryocystitis (n=4; Table 1).
|
Characteristics |
|
|
Number of patients |
111 |
|
Number of procedures |
128 |
|
Left side |
74 (57.8%) |
|
Right side |
54 (42.2%) |
|
Mean age, years (range) |
62.3 (18.1-93.0) |
|
Sex ratio (female:male) |
5.2 (93:18) |
|
Mean follow-up, months (range) |
40.5 (12.0-83.28) |
|
Etiology Idiopathic |
128 123 (96.1%) |
|
Facial fractures |
3 (2.3%) |
|
Chemotherapy and radiotherapy |
2 (1.6%) |
|
Concomitant punctual stenosis |
15 (11.7%) |
|
Symptoms |
128 |
|
Epiphora only |
94 (73.4%) |
|
Chronic purulent discharge |
30 (23.4%) |
|
Acute dacryocystitis only |
4 (3.1%) |
|
Radiographic investigations |
128 |
|
Paranasal sinus CT |
103 (80.5%) |
|
Dacryoscanner |
21 (16.4%) |
|
Paranasal sinus Magnetic Resonance Imaging |
7 (5.5%) |
|
Dacryography |
20 (15.6%) |
Table 1: Demographic characteristics.
Forty-one additional sinonasal procedures were performed in the same surgical setting in 34 patients (30.6%); these included 29 septoplasties, nine paranasal sinus procedures, and three turbinoplasties. Simultaneous punctoplasty for suspected concomitant lower punctal stenosis was performed in 13 procedures (10.2%; Table 2). From the 128 procedures, 111 (86.7%) resulted in complete symptom resolution while 17 (13.3%) were associated with persistent tearing without infection. Revision surgery was performed in 13 (10.2%) cases. Rhinostomy closure, a pyogenic granuloma, and an occlusive synechia between the middle turbinate and the lateral nasal wallwere observed during revision surgery in nine, two, and two cases, respectively (Table 3). All revision surgeries resulted in symptom resolution over a mean follow-up period of 38.8 months.
|
Total patients |
111 |
|
General anesthesia |
111 (100%) |
|
Ambulatory surgery |
41 (36.9%) |
|
Additional sinonasal surgery |
128 |
|
Septoplasty |
29 (22.7) |
|
Paranasal sinus surgery |
9 (7.0%) |
|
Turbinoplasty |
3 (2.3%) |
|
Punctoplasty |
13/128 (10.2%) |
|
Intraoperative findings |
128 |
|
Dacryolith |
9 (7.0%) |
|
Purulent discharge |
40 (31.3%) |
|
Stenting |
127/128 (99.2%) |
|
Mean stenting duration, days |
31.3 |
|
Complications |
0 |
|
Acute rhinosinusitis |
0 (0%) |
|
Skull base |
0 (0%) |
|
Injury/cerebrospinal fluid leakage |
0 (0%) |
|
Orbital complications |
0 (0%) |
Table 2: Intraoperative features.
|
Anatomically patent rhinostomy |
115/128 (89.8%) |
|
Complete symptom resolution |
111/128 (86.7%) |
|
Persistence of symptoms |
17/128 (13.3%) |
|
Re-stenosis |
9 (53.0%) |
|
Granuloma |
2 (11.8%) |
|
Turbinalsynechia |
2 (11.8%) |
|
Punctalstenosis |
2 (11.8%) |
|
Canalicular obstruction |
2 (11.8%) |
|
Mean time to revision surgery, months |
34.4 (5.8-119.5) |
Table 3: Postoperative data.
DISCUSSION
In the present study, we described the outcomes of primary powered DCR for low lachrymal pathway obstruction in adult patients. DCR is a well-known surgical procedure that can be performedby either ophthalmologists or Ear-Nose-Throat (ENT) surgeons’ through an external or endonasal approach. For the best surgical outcomes, preoperative identification of the obstruction level is mandatory. Indeed saccal and post-saccal obstructions are the best operative indications [2]. This requires thorough clinical examination with or without imaging of the lachrymal pathway.
Suggestive signs of lower lachrymal pathway obstruction during clinical examination include purulent tearing, a lump at the medial canthus, pus extrusion on finger pressure at the medial cantus, or acute dacryocystitis. Complementary testsmade in the physician’s office include lachrymal syringing with saline solution and probing. During lachrymal syringing, when saline is injected into the lower canaliculus and reflux is observed through the superior canaliculus, the presence of lower lachrymal obstruction is likely. On the other hand, if reflux is observed through the ipsilateral canaliculus, higher lachrymal obstruction (canalicular obstruction) is the first suspicion. In such cases, endonasal DCR is certainly not an indication. During probing, a Bowman stent is inserted into the lower canaliculus until it contacts the medial wall of the lachrymal fossa. If there ishard contact (bony contact), the obstruction is considered to be low, beyond the common canaliculus. If the contact is soft,the obstruction is high; this is also known as pre-saccal or canalicular obstruction. The advantage of probing is the eventual possibility to discover the level of the lacrimal canalicular obstruction.
For most ophthalmologists, these two tests are sufficient for the diagnosis of lacrimal obstruction and confirm the need for DCR. Therefore, imaging of the lachrymal system is not always necessary in all cases. However, it can help in refining the diagnosis and provides objective information about the anatomy of the lacrimal obstruction [11]. Imaging can include sinus Computed Tomography (CT), dacryoscanner, MRI, or digital subtraction dacryocystography.
In the past, clinicians sendpatients for conventional CT of the paranasal sinuses, which provided information about the anatomy of the nose and paranasal sinus cavities. Furthermore, it can demonstrate sinonasal pathologies or anatomical variations such as a septal deformity, concha bullosa, or chronic rhinosinusitis. The presence of these pathologiesjustifies the performance of nasal or sinonasal procedures along with DCR. In the present study, we performed 41 additional surgeries with DCR, including septoplasty, sinus surgery, and turbinoplasty (Table 2). Conventional CT provides information about the attachment of the uncinate process to the frontal process of the maxilla, pneumatization of the Agger nasi cell, and thickness of the bony structures forming the lacrimal eminence, particularly the frontal process of the maxilla. The varying positions of these anatomical structures have been reported by Fayet et al. in 2000 [12]. However, conventional CT cannot preciselylocalize the lachrymalobstruction in most cases.
Consequently, we tried to use a dacryoscanner rather than conventional CT before all DCR procedures in this study. A contrast agent (OmnipaqueR) was injected in the lachrymal system by our ENTspecialist during lachrymal syringing. This facilitated accurate localization of the lachrymal obstruction (pre-saccal, saccal, or post-saccal) and also providedinformation about the nasal and sinonasal anatomies [13,14].
Some patients (n=7) required an MRI for the differentiation of a dacryocele from a tumor, particularly because they presented with a hard lump at the medial canthus. T2-weightedsequenceswithout contrast medium injection are generally adequate for exploring the contents ofthe lacrimal pathway.However, MRI is not easily available; therefore, its use is limited [15].
Digital subtraction dacryocystography is reported the gold standard tool [16] for the evaluation of lachrymal obstruction. The images are digitalizedand the bony structures areeliminated, which allows proper visualization of the lacrimal pathway. Unfortunately, this modality requires a high radiation dose. Moreover the radio-opaque dye is pushed under high pressure in the lachrymal system and can annihilate an eventual partial obstruction [16].
Powered DCR is a standardizedprocedure with different steps around which we have some considerations.The first step involves the creation of a nasal mucosal flap. We have found no advantages of preserving this flap completely. Although this topic remains debatable in the literature, no randomized controlled trialhas determined the optimal solution for this.Next, we paid attention to the attachment of the uncinate process to the lacrimal eminence. As reported by Fayet in2002, there are three anatomical variations [17], which are clearly visible on axial CT images. When the uncinate is directly attached to the posterior edge of the frontal process of the maxilla, a vertical uncinectomy is performed. In other cases, uncinectomy is not necessary.
The next step involves identification of the Agger nasi cell. This provides information about the location of the lachrymal sacand indicates the superior limit of the surgical field. Wormald et al. confirmed that this step is important for ensuring an appropriate communication between the lacrimal sac and the nasal cavity [18].
Subsequently, the procedure goes on with the resection of the bony part of the frontal process of the maxilla. This can be done with “cold “instruments (Kerrisson forceps), or even an osteotome; however when the bone is thick, in the vicinity of the axilla of the middle turbinate, a 2.5-mm curved diamond drill (Medtronic Xomed) connected to a powered hand piece curved diamond drill is a very nice tool. This instrument allows precise drilling of the bone without damage to the lachrymal mucosa [19]. The drilling must be performed adequately anteriorly and superiorlyin order to expose widely the mucosa of the nasolacrimal duct and inferior part of the lachrymal sac.
Then, the lachrymal mucosa is vertically incised using a sickle knife from inferior to superior.
Finally, lachrymal syringing is performed for confirming the patency of the surgical stoma and excluding any canalicular obstruction.
At the end of the surgery, bicanalicular nasal stenting can be performed depending on the local conditions. We placed a silicone tube in our patients, although this is a highly controversial topic. The procedure has gained popularity since its first description by Gibbs in 1967 [20]. The stent remains in place during the first few postoperative weeks and maintains the patency of the rhinostomy and canaliculi. However, the healing process could result in clogging of the canaliculi and rhinostomy. It has been reported that iatrogenic stenosis of the lachrymal pathway eventually occurs because of local irritants such as a bilacrimal stent [21]; thus, some surgeons prefer to avoid the use of this device. Chong et al. conducted a randomizedtrial in 120 patients with a minimum follow-up period of 12 months and found no statistically significant difference between stented and non-stented endoscopic DCR [22]. Mohamad et al. reported that the success rate was considerably lower with stenting than without stenting [23]. On the other hand, Yung et al. [2] showed very good results of stenting in 1083 patients with a mean follow-up duration of 32 weeks.
In this study, we found excellent success rates, with 89.8% patent rhinostomies and 86.7% asymptomatic cases observed over mean and minimal follow-up periods of 40.5 and 24 months, respectively. These results are acceptable and comparable with those published in studies worldwide (Table 4). The high success rates are directly correlated with the surgical indication, quality of the procedure, localizationof the surgical opening, and postoperative care.
|
Authors |
Year |
Procedures |
Patients |
Mean follow-up |
Results reported in the article (%) |
Comments |
|
Coumou et al. [24] |
2016 |
624 |
Children and adults |
5.7 months |
90.1% |
Anatomically patent rhinostomy |
|
Yung et al. [2] |
2015 |
1083 |
Adults only |
32 weeks |
92.7% |
Symptom-free |
|
Karim et al. [25] |
2011 |
105 |
Adults only |
6 months |
82.4% |
Symptom-free |
|
Ali et al. [26] |
2015 |
196 |
Adults only |
17.1 |
93.0% |
Symptom-free |
|
Fayet et al. [27] |
2014 |
300 |
Adults only |
28 |
94.0% |
Symptom-free |
|
Kingdom et al. [28] |
2019 |
87 |
Adults only |
28.7 |
93.1% |
Symptom-free |
Table 4: Published outcomes of primary powered endoscopic dacryocystorhinostomy.
It is important to rule out upper lachrymal pathway obstruction.Concomitant punctoplasty should be performed if punctal stenosis is confirmed. In the present study, punctoplasty was performed in 13 out of 128 DCRs (Table 3).
The causes of surgical failure are well described in the literature [21], with strong agreement that inadequate osteotomy and cicatricial closure of the rhinostomy are the most common causes.In the present study, closure of the stoma was the most frequent cause of failure (Table 3). This problem can be prevented by a large surgical opening, daily and regular postoperative nasal douching, and meticulous postoperative care. Granuloma formation is another knowncause of failure that is caused by crusting or a foreign body reaction to silicone intubation. Accordingly, all patients should be closely monitored in the early postoperative period and receive the local treatments mentioned above. Early detection of a granuloma allows timely removal of the silicone tube and the administration of topical nasal steroids. Cauterization of the granuloma is another treatment option. If the granuloma still persists, symptomatic revision surgery is indicated.
This study has limitations associated with its retrospective design. Nonetheless, the mean follow-up duration of 40.5 months (minimum 12 months) is one of the longest reported in the literature concerning endoscopic DCR.Thusthe findings make an important contributionto the literature concerning endoscopic DCR.
CONCLUSION
In conclusion, our findings suggest that endoscopic powered DCR is a reliable procedure for the treatment of saccal or post-saccal obstruction, with high success rates. Complete preoperative lachrymal and sino nasal evaluations are mandatory for accurate diagnosis and obtaining information about the sinonasalanatomy. Powered instrumentation enables the creation of a wide opening thatincludes the lachrymal sac and the nasolacrimal duct. Moreover, endonasal DCR avoids a skin incision, allows the preservation of the lacrimal pump, and is associated with low morbidity and quick rehabilitation.
FUNDING
The authors have no proprietary or commercial interest in any materials discussed in this article.
CONFLICT OF INTEREST
The authors declare that they have no conflict of interest.
ETHICAL APPROVAL
All procedures performed in this study involving human participants were in accordance with the ethical standards of the institutional research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent: No informed consent was waived because of the retrospective nature of the study.
REFERENCES
- Weller C, Leyngold I (2018) Dacryocystorhinostomy: Indications and surgical technique. Oper Tech Otolayngol Head Neck Surg 29: 203-207.
- Yung MW, Hardman-Lea S (2002) Analysis of the results of surgical endoscopic dacryocystorhinostomy: effect of the level of obstruction. Br J Ophthalmol 86: 792-794.
- Kamal S, Ali MJ (2018) Primary Acquired Nasolacrimal Duct Obstruction (PANDO) and Secondary Acquired Lacrimal Duct Obstructions (SALDO). In: Ali M (ed) Principles and Practice of Lacrimal Surgery. Springer, Singapore, Pg no. 163-171.
- Bartley GB (1992) Acquired lacrimal drainage obstruction: an etiologic classification system, case reports, and a review of the literature. Part 2. Ophthalmic Plast Reconstr Surg 8: 243-249.
- Bartley GB (1993) Acquired lacrimal drainage obstruction: an etiologic classification system, case report, and a review of the literature. Part 3. Ophthalmic Plast Reconstr Surg 9: 11-26.
- Caldwell GW (1893) Two new operations for obstruction of the nasal duct, with preservation of the canaliculi and an incidental description of a new lacrimal probe. NY Med J 57: 581-582.
- Toti A (1904) Nuovo metodo conservatore dicura radicale delle suppurazioni croniche del sacco lacrimale (dacriocistorinostomia). Clin Mod (Firenze) 10: 385-387.
- Metson R (1991) Endoscopic surgery for lacrimal obstruction. Otolaryngol Head Neck Surg 104: 473-479.
- Wormald PJ (2002) Powered endoscopic dacryocystorhinostomy. Laryngoscope 112: 69-72.
- Yakopson VS, Flanagan JC, Ahn D, Luo BP (2011) Dacryocystorhinostomy: History, evolution and future directions. Saudi J Ophthalmol 25: 37-49.
- Roos JCP, Ezra DG, Rose GE (2017) Preoperative imaging should be performed for all cases of acquired nasolacrimal duct obstruction’-No. Eye (Lond) 31: 349-350.
- Fayet B, Racy E (2000) Is the uncinate process resection the key to endonasal dacryocystorhinostomy?. J Fr Ophtalmol 23: 433-436.
- Abi-Khalil S, Schakal A (2016) The dacryoscanner, an optimal technique for nasolacrimal system exploration. J Med Liban 64: 223-227.
- Ducasse A, Bonnet F, BrugniartC, Scholtes F (2008) 050 Intérêt du dacryoscanner en chirurgie lacrymale. Journal Français d Ophtalmologie 31: 30-30.
- Rubin PA, Bilyk JR, Shore JW, Sutula FC, Cheng HM (1994) Magnetic resonance imaging of the lacrimal drainage system. Ophthalmology 101: 235-243.
- Singh S, Ali MJ, Paulsen F (2019) Dacryocystography: From theory to current practice. Ann Anat 224: 33-40.
- Fayet B, Racy E, Assouline M (2002) Systematic unciformectomy for a standardized endonasal dacryocystorhinostomy. Ophthalmology 109: 530-536.
- Wormald PJ, Tsirbas A (2009) Powered endoscopic dacryocystorhinostomy with mucosal flaps. Oper Tech Otolayngol Head Neck Surg 20: 92-95.
- Codere F, Denton P, Corona J (2010) Endo nasal dacryocystorhinosotomy: a modified technique with preservation of the nasal and lacrimal mucosa. Ophthalmic Plast Reconstr Surg 26: 161-164.
- Gibbs DC (1967) New probe for the intubation of lacrimal canaliculi with silicone rubber tubing. Br J Ophthalmol 51: 198.
- Dave TV, Mohammed FA, Ali MJ, Naik MN (2016) Etiologic analysis of 100 anatomically failed dacryocystorhinostomies. Clin Ophthalmol 28.
- Chong KK, Lai FH, Ho M, Luk A, Wong BW, et al. (2013) Randomized trial on Silicine Intubation in endoscopic Mechanical Dacryocystorhinostomy (SEND) for primary Nasolacrimal Duct Obstruction. Ophthalmology 120: 2139-2145.
- Mohamad SH, Khan I, Shakeel M, Nandapalan V (2013) Long-term results of endonasal dacryocystorhinostomy with and without stenting. Ann R Coll Surg Engl 95: 196-199.
- Coumou AD, Genders SW, Smid TM, Saeed P (2016) Endoscopic dacryocystorhinostomy: long term experience and outcomes. Acta Ophthalmol 95: 74-78.
- Karim R, Ghabrial R, Lynch T, Tang B (2011) A comparison of external and endoscopic endonasal dacryocystorhinostomy for acquired nasolacrimal duct obstruction. Clin Ophthalmol 5: 979-989.
- Ali MJ, Psaltis AJ, Murphy J, Wormald PJ (2015) Powered endoscopic dacryocystorhinostomy: a decade of experience. Ophthalmic Plast Reconstr Surg 31: 219-221.
- Fayet B, Katowitz WR, Racy E, Ruban JM, Katowitz JA (2014) Endoscopic dacryocystorhinostomy: the keys to surgical success. Ophthalmic Plast Reconstr Surg 30: 69-71.
- Kingdom TT, Barham HP, Durairaj VD (2019) Long-term outcomes after endoscopic dacryocystorhinostomy without mucosal flap preservation. Laryngoscope 130: 12-17.
Citation: Renwart W, De Dorlodot C, Eloy P (2020) Primary Endonasal Endoscopic Powered Dacryocystorhinostomy for Low Obstruction of the Lacrimal Excretory System in Adult Patients: A 5-Years Experience. J Otolaryng Head Neck Surg 6: 40
Copyright: © 2020 Renwart W, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

