
Pure Cannabidiol versus Cannabidiol-Containing Extracts: Distinctly Different Multi-Target Modulators
*Corresponding Author(s):
Gerhard NahlerClinical Investigation Support Gmbh, Kaiserstrasse 43, 1070 Wien, Austria
Tel:+43 15234015,
Email:cis-qa@aon.at
Abstract
Cannabis saliva L. strains can be divided into a number of groups according to their content of the psychotropic phytoCannabinoid delta-9-TetrahydroCannabinol (THC) and of the non-psychotropic Cannabinoid Cannabidiol (CBD). Although the main focus has been on THC in the past, there is growing interest on strains rich in CBD. Strains with a ratio of CBD to THC above one and a content of THC of less than 1%, often legally limited to 0.3%, are commonly designed as hemp (industrial hemp or fiber-type Cannabis) in contrast to THC-rich strains (drug-type Cannabis and marijuana), and are grown as outdoor cultures in many countries. Such strains contain CBD as the main Cannabinoid in addition to numerous other phytosubstances that are in general not further characterized but known to have beneficial effects on health. They are used for the preparation of extracts and other products e.g., essential oils, teas or edibles and promoted as nutraceuticals in hemp shops and on the internet. These products are increasingly popular, and a number of countries allow the cultivation of strains poor in THC. THC and CBD but also many other phytosubstances in hemp in particular terpenes and flavonoids target the so called endoCannabinoid system that regulates the homeostasis of vital processes. However, the chemical profile of hemp and derivatives is subject to a wide variability due to a number of factors such as the nature of cultivars, agroclimatic conditions and methods of preparation. Hemp strains and concentrates differ not only in their chemical composition but also in their physiological effects. This heterogeneity has led to conflicting results in clinical studies with Cannabis formulations in the past. The physiological effects of purified Cannabinoids differ from those observed with extracts. Most products from outdoor cultures cannot be sufficiently standardized, and so are currently unsuitable as medications. They may however play an important role in complementary and integrative medicine. For future clinical studies it is important that only well characterized products are used.
Keywords
Cannabidiol; Cannabis; Entourage effect; Flavonoids; Hemp; Terpens
INTRODUCTION
CBD, THE MAIN PHYTOCOMPOUND IN HEMP, IS A MODULATOR OF A NUMBER OF ENDOGENOUS PHYSIOLOGIC MECHANISMS
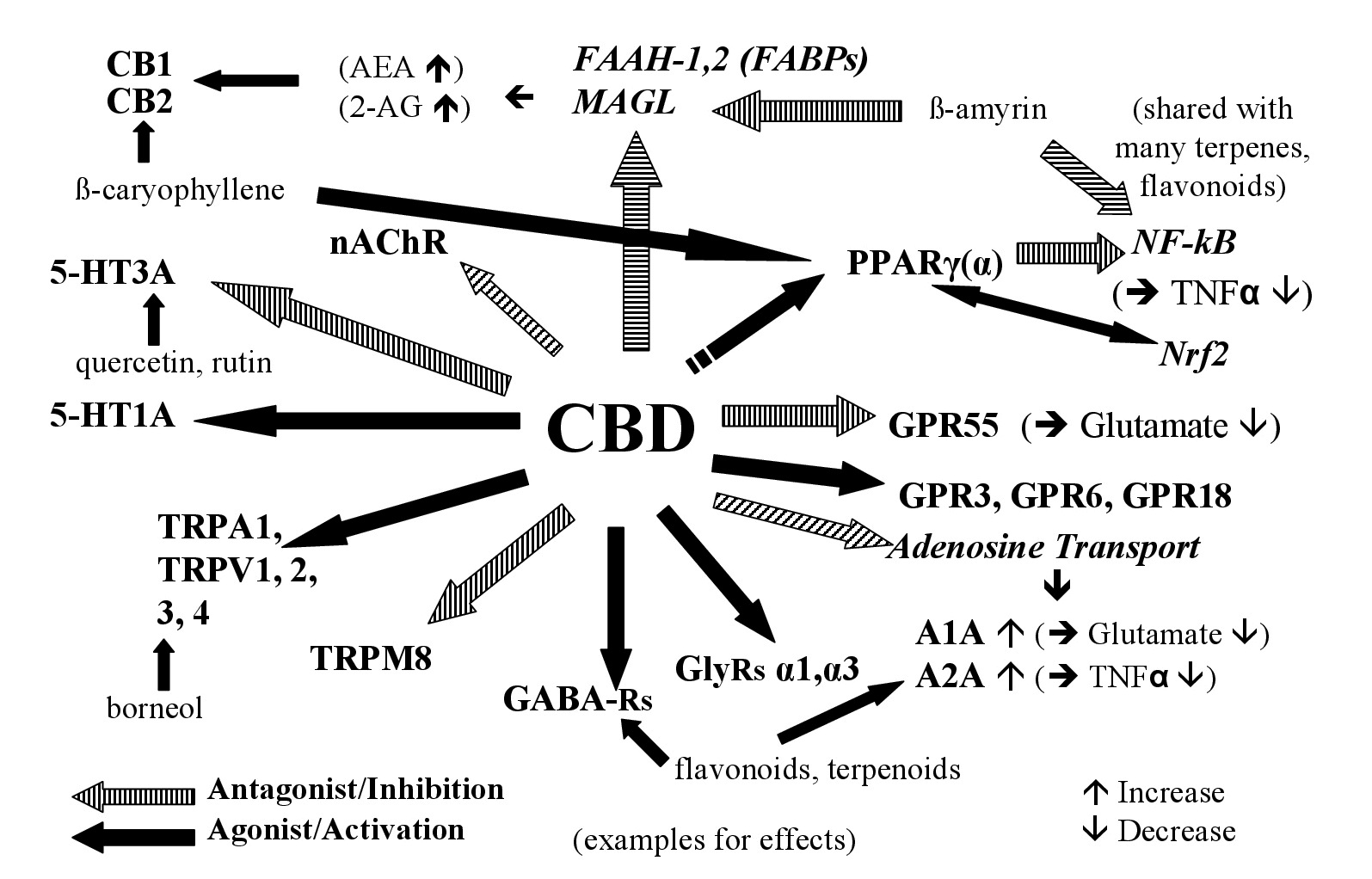 Figure 1: Examples of the interaction of CBD and non-Cannabinoids in hemp with the endoCannabinoid system (see tables for more details).
Figure 1: Examples of the interaction of CBD and non-Cannabinoids in hemp with the endoCannabinoid system (see tables for more details).| Cannabinoid | Targets | Effects (Examples) | Ref. |
| CBD | Agonist of 5-HT1A, TRPA1, TRPV1,2,3,4; PPARg, GPR3,6,18; antagonist of TRPM8; 5-HT3A, GPR55, adenosine transport protein; positive allosteric modulator of GABAA, GlyRs; inhibits n-AChR, NaV channels, LOX-5,-15; moderate inhibitor of FAAH | Anti-inflammatory, analgesic, anxiolytic, antidepressant; attenuates nausea, vomiting, motor and cognitive impairment; inhibits cancer cell growth | [14-24] |
| CBDA |
Agonist of 5HT1A, TRPA1, TRPV1, TRPV4; antagonist of TRPM8; inhibitor of COX-2, NAAA |
Anti-inflammatory, anxiolytic, antidepressant; attenuates nausea, vomiting, motor and cognitive impairment; antineoplastic |
[16,25,26] |
| THC | Agonist of CB1, CB2, TRPA1, TRPV2, TRPV3, TRPV4; GPR18, PPARg; potentiates Glycine receptors (GlyRs); antagonist of TRPM8, 5-HT3A | Anti-inflammatory, anxiolytic, pro-apoptotic effects; analgesic (additive with kappa-Opioid-receptor agonists) |
[16,27,28] |
| THCA |
Weak binding to CB1, CB2; agonist of PPARg, TRPA1, TRPV2; antagonist of TRPM8; weak inhibitor of FAAH, MAGL, COX-1,-2 |
Anti-inflammatory, neuroprotective, pro-apoptotic effects |
[29-31] |
| CBG |
Agonist of TRPA1, TRPV1, TRPV2, TRPV4, PPARg; alpha2-adrenoceptor, Antagonist of 5-HT1A, TRPM8, CB1; inhibits NaV channels, COX-2 |
Antiemetic (may oppose effects of CBD), anti-inflammatory, antineoplastic, antidepressant; stimulates appetite, neuroprotective | [19,32-34] |
HEMP (CANNABIS) CONTAINS ALSO FLAVONOIDS
| Terpenoid | Targets | Effects (Examples) | Ref. |
| α,β-Amyrin | Activates CB1 (more potent than d9-THC); inhibits hydrolysis of MAGL, ABHD6, -12 and 2-AG; prevents NF-kB activation |
Antinociceptive, anti-hyperglycemic, hypolipidemic; anti-inflammatory |
[56-59] |
| Borneol | Activates TRPV3; inhibits NF-kB; positive allosteric modulator of GABAA receptors | Neuroprotective; antibacterial; occurs in hemp in low concentrations | [60,61] |
| ß-Caryo-phyllene | Selective CB2-agonist; PPARg,-a-agonist; nAChR antagonist | Anti-inflammatory (comparable to dexamethasone), analgesic; antibiotic, antineoplastic; reduces intracellular triglyceride accumulation | [62-67] |
| α-Humulene (α-caryo-phyllene) | Prevents NF-kB and activator protein 1 (AP-1) activation | Anti-inflammatory (comparable to dexamethasone), anti-nociceptive; antineoplastic; antibacterial, appetite suppressant, insecticidal | [67-69] |
| D-Limonene + | Prevents activation of NF-kB | Anti-inflammatory; antineoplastic; anxiolytic, insect repellent |
[7,66, 70-72] |
|
D-Linalool Linalool oxide |
Agonist to PPARa | Anticonvulsive, antinociceptive, sedating, local anesthetic effects; reduces plasma triglycerides |
[37,65, 73,74] |
| ß-Myrcene + | Prevents activation of NF-kB | Anti-inflammatory, analgesic, sedative, muscle relaxant, blocks hepatic carcinogenesis by aflatoxin |
[37,40, |
| Nerolidol | Prevents activation of NF-kB; modulates GABAA receptors | Antinociceptive; anti-inflammatory, anxiolytic; enhances skin penetration, antimalarial | [76] |
| α-Pinene + | (+)-α-pinene prevents activation of NF-kB; more potent than (−)-α-Pinene | Anti-inflammatory; chondro-protective; acetylcholinesterase-inhibitor, bronchodilator, antifungal, insect repellent; antibacterial (against MRSA) |
[7,37, |
| α-Terpineol | Inhibition of COX-2 (superior to aspirin) | Anti-inflammatory, promotes wound healing | [70,78] |
|
Terpinolene (delta-terpinene) ° |
Inhibits AKT-formation in leukemia cells | Antiproliferative, sedative, promotes sleep; antibacterial, antifungal, insect repellent | [79,80] |
+ Present in hemp flower tee (Futura strain); ABHD - Alpha, Beta-Hydrolase; GABA - Gamma Aminobutyric Acid; MRSA - Methicillin-Resistant Staphylococcus Aureus; AKT - protein Kinase
| Flavonoid | Targets | Effects (Examples) | Ref. |
| Apigenin + |
Agonist of PPARg, Nrf-2; |
Anxiolytic, anti-inflammatory, lowers formation of amyloid ß (Ab1-40, Ab1-42); nephroprotective; inhibits xanthin oxidase/anti-uricaemic effect, antibacterial, antiviral; genoprotective |
[51] [81-87] |
| Cannflavin A,B | Inhibitor of prostaglandin PGE2 | Anti-inflammatory (more effective than aspirin but less than dexamethasone); anti-protozoal-, anti-leishmanial activity | [88-90] |
| Genistein |
Upregulation/agonist of PPARg, Nrf2; downregulates NF-kB; modest inhibitor of FAAH |
Reduces hepatic fibrosis, downregulates lipogenesis; nephroprotective, anti-uricaemic effect; lowers amyloid-ß; reactivates methylation-silenced genes in cancer cells; phytoestrogen |
[83,84, 86,91-93] |
| Kaempferol + | Inhibits COX-1, COX-2, LOX; agonist of PPARg, Nrf2; downregulates NF-kB; modest inhibitor of FAAH; | Antineoplastic; anti-cholinesterase activity, lowers amyloid-ß formation, plasmatic triglycerides; weight reducing; antidepressant; antibacterial, antiviral, antifungal, antiprotozoal | [83-85,93-96] |
| Luteolin + | Upregulates PPARg, Nrf-2; downregulates NF-kB; | Anti-inflammatory; antineoplastic, increases DNA-repair/rejoining of strand breaks; anti-uricaemic; stimulates mineralization of osteoblasts | [51,81,83-87,91,97] |
| Myricetin | Downregulates NF-kB; | Antineoplastic; potentiates sperm function; antidiabetic |
[98-100] |
|
Naringenin (a glycone of naringin) |
Agonist of PPARg, PPARa, Nrf2; inhibits NF-kB, COX-2 |
Inhibits osteoclast formation, decreases fibrosis, hepato- and neuro-protective; crosses the BB barrier; antigenotoxic, decreases cholesterol and metabolic syndrome; inhibits S. aureus | [51,65,81,83,84,86,91,100-103] |
| Orientin + | NF-kB inhibition; | Anti-inflammatory, antineoplastic; antibiotic, enhances repair of radiation damages | [104-106] |
| Quercetin + | Induces PPARg, Nrf-2, downregulates NF-kB; inhibits 5-LOX and COX-1, COX-2 | Pro-apoptotic, antihistaminic; hepato-protective; anti-inflammatory; inhibits amyloid ß; anti-cholinesterase activity; antiviral, antibacterial; reduces blood pressure in hypertensive patients | [51,61,81,84-87,91,96,97,100,107-111] |
| Rutin | (peripheral) CB1 agonist; downregulates NF-kB; inhibits 5-HT3A, GABAc receptors, COX-2 | Antifibrotic; decreases oxidative DNA damages; may reduce seizures and epilepsy-associated cognitive/behavioural symptoms; |
[59,61,86,91,100] |
|
Vitexin + (apigenin-8-C-glucoside) |
Downregulates NF-kB | Anti-inflammatory, antihyperalgesic, antihypertensive, anticonvulsant; antineoplastic, protects pancreatic ß-cells, cardio- and neuro-protective, enhances memory | [112-115] |
THE COMPOSITION OF PHYTOCOMPONENTS IN OUTDOOR CULTURES IS HIGHLY VARIABLE
| Agro-climatic factor | CBD | THC | Influence on the content of CBD and THC |
| Soil temperature | ↑ |
~ |
Soil temperature at 5 cm has a positive influence on the content of CBD |
| Air humidity | ~ - |
↑↑ | Air humidity has a positive influence which is more pronounced for the content of THC than CBD |
| Average temperature in the entire growing period | ↑↑ | ↑ | The positive influence on CBD is about twice as high as for THC |
| Growing Season precipitation | ↓↓ | ↓ | The negative influence of precipitations is more pronounced for the CBD content |
| Fertilization (K, N, P) | ↑ (NK) |
↑ (PK) |
Max amount of CBD observed at NK-, lowest at NPK fertilization Max amount of THC observed at PK-, the lowest at NP fertilization |
| Nitrogen fertilizer | ↓ (NPK) |
↓ (NP) |
Lowest amount of CBD observed at NPK fertilization; Lowest amount of THC observed at NP fertilization |
| Age of leaves | ↓ | ↓ | Older leaves contained less Cannabinoids than younger ones |
| Stage of plant development | ↑ | ↑ | The content of Cannabinoids and terpenoids increases during growth in fiber-type but tend to decrease in the last stages of vegetation in drug-type Cannabis |
(i) Different strains may have different therapeutic effects on the body and/or mind; CBD-rich (hemp) preparations (extracts) are likely to differ from THC-rich (drug-type) preparations (extracts)
(ii) Due to the postulated entourage effect the therapeutic effects of hemp preparations (extracts) are likely to differ from pure CBD, and
(iii) Drug-type preparations (extracts) are likely to differ from pure THC
| Extract | Comparator | Effects | Reference |
|
65.6% CBD, (THC not given) |
CBD |
In vitro; effect on human bladder contractility; extract more effective than CBD | [135] |
|
64.5% CBD, |
CBD (10 mg p.o./kg); (THC had no effect) |
Rat model; extract completely relieved thermal hyperalgesia and partially attenuated mechanical allodynia; chronic CBD had only a partial effect |
[136] |
|
17.9% CBD, 1.1% THC, |
CBD, dose range paw swelling: 1-50mg/kg; dose range pain: 10-150mg/kg |
Mouse model; max. effect on paw swelling and pain after 5mg CBD i.p./kg compared to 50mg E i.p./kg; Orally, max. effect on paw swelling and pain after 25mg CBD/kg compared to 50mg E/kg on swelling and 150mg E/kg on pain; E was more effective on swelling after oral, CBD was more effective after i.p. administration (based on the CBD content); CBD showed a bell shaped dose-response curve |
[137] |
|
Extract with ≈. 70% CBD (Nabidiolex) |
CBD | In vitro, eight different cancer cell liness; E mostly equipotent to CBD; CBD was the most potent (CBDA the least) out of 6 pure Cannabinoids |
[138] |
| Extract (≈. 70% CBD) 6.5mg E i.p./kg/dose | 5mg CBD i.p./kg /dose | In vivo (mice), human breast cancer xenograft; CBD was slightly more potent than the extract |
[138] |
|
64.6% CBD, 2.5% THC (Nabidiolex) |
CBD |
In vitro, Ca++ response in neurons and glia cells; pure CBD induced a larger response than E in neurons; in glia no such difference was observed |
[139] |
|
72.6% THC, 2.5% CBD, (Tetrabinex) |
THC |
In vitro, Ca++ response in neurons and glia cells; pure THC induced a larger response than E in neurons; in glia no such difference was observed |
[139] |
| 65.9% CBD, 2.4% THC, |
CBD |
In vitro, proliferation in colorectal cancer cells; both, CBD and E reduced cells (no significant differences) |
[36] |
| 63.5% CBD, 3.6% THC, | CBD | In vitro, glioma cell lines; CBD was 1.1 to 2.5x more active than E |
[140] |
| 65.4% THC, 0.4% CBD | THC | In vitro, glioma cell lines; extract is 1.2 to 1.3x more active than pure THC |
[140] |
| 20% THC, (minor content of CBD, CBN) |
THC (CBD had no effect) |
In vivo, mouse MS-model; more rapid relief from spasticity with the extract than after pure THC but size of antispastic effect is similar; |
[141] |
| 20% THC, (minor content of CBD, CBN) |
THC (CBD had no effect) |
In vitro, rat brain slice model of epilepsy; more rapid onset of anti-convulsant activity with the extract than with pure THC |
[141] |
• Choice of the strain, its composition of phytocompounds, in addition to Cannabinoids
• Agroclimatic/growth conditions: precipitation, sunshine, soil, use of fertilizers, pesticides
• Harvest: time, parts of the plant harvested, transport, drying and storage conditions
• Extraction: methods, solvents, temperature
CONFLICT OF INTEREST
REFERENCES
- Lynch RC, Vergara D, Tittes S, White K, Schwartz CJ, et al. (2016) Genomic and chemical diversity in Cannabis. J Critical Reviews in Plant Sci 35: 349-363.
- Arno H, Katerina T, Stelios P (2016) Cannabis: From cultivar to chemovar II-A metabolomics approach to Cannabis classification. Cannabis Cannabinoid Res 1: 202-215.
- Brenneisen R (2007) Chemistry and analysis of phytoCannabinoids and other Cannabis constituents. In: ElSohly MA (ed.). Marijuana and the Cannabinoids. Humana Press, Totowa, New Jersey, USA.
- Aizpurua-Olaizola O, Soydaner U, Öztürk E, Schibano D, Simsir Y, et al. (2016) Evolution of the Cannabinoid and Terpene Content during the Growth of Cannabis sativa Plants from Different Chemotypes. J Nat Prod 79: 324-331.
- De Meijer EPM, Hammond KM (2005) The inheritance of chemical phenotype in Cannabis sativa L. (II): Cannabigerol predominant plants. Euphytica 145: 189-198.
- Fischedick JT, Hazekamp A, Erkelens T, Choi YH, Verpoorte R (2010) Metabolic fingerprinting of Cannabis sativa L., Cannabinoids and terpenoids for chemotaxonomic and drug standardization purposes. Phytochemistry 71: 2058-2073
- Mediavilla V, Steinemann S (1997) Essential oil of Cannabis sativa L. strains. J Int Hemp Association 4: 80-82.
- Carlini EA, Cunha JM (1981) Hypnotic and antiepileptic effects of Cannabidiol. J Clin Pharmacol 21: 417-427.
- Ibeas Bih C, Chen T, Nunn AV, Bazelot M, Dallas M, et al. (2015) Molecular targets of Cannabidiol in neurological disorders. Neurotherapeutics 12: 699-730.
- Burstein S (2015) Cannabidiol (CBD) and its analogs: a review of their effects on inflammation. Bioorg Med Chem 23: 1377-1385.
- Pisanti S, Malfitano AM, Ciaglia E, Lamberti A, Ranieri R, et al. (2017) Cannabidiol: State of the art and new challenges for therapeutic applications. Pharmacol Ther 175: 133-150.
- Turner SE, Williams CM, Iversen L, Whalley BJ (2017) Molecular pharmacology of phytoCannabinoids. Prog Chem Org Nat Prod 103: 61-101.
- Laprairie RB, Bagher AM, Kelly ME, Denovan-Wright EM (2015) Cannabidiol is a negative allosteric modulator of the Cannabinoid CB1 receptor. Br J Pharmacol 172: 4790-4805.
- Bakas T, van Nieuwenhuijzen PS, Devenish SO, McGregor IS, Arnold JC, et al. (2017) The direct actions of Cannabidiol and 2-arachidonoyl glycerol at GABAA receptors. Pharmacol Res 119: 358-370.
- Davies PA (2011) Allosteric modulation of the 5-HT3 receptor. Curr Opin Pharmacol 11: 75-80.
- De Petrocellis L, Ligresti A, Moriello AS, Allarà M, Bisogno T, et al. (2011) Effects of Cannabinoids and Cannabinoid-enriched Cannabis extracts on TRP channels and endoCannabinoid metabolic enzymes. Brit J Pharmacol 163: 1479-1494.
- Di Marzo V, Stella N, Zimmer A (2015) EndoCannabinoid signalling and the deteriorating brain. Nat Rev Neurosci 16: 30-42.
- Fernández-Ruiz J, Sagredo O, Pazos MR, García C, Pertwee R, et al. (2013) Cannabidiol for neurodegenerative disorders: important new clinical applications for this phytoCannabinoid? Br J Clin Pharmacol 75: 323-333.
- Hill AJ, Jones NA, Smith I, Hill CL, Williams CM, et al. (2014) Voltage-gated sodium (NaV) channel blockade by plant Cannabinoids does not confer anticonvulsant effects per se. Neurosci Lett 566: 269-274.
- Kozela E, Juknat A, Gao F, Kaushansky N, Coppola G, et al. (2016) Pathways and gene networks mediating the regulatory effects of Cannabidiol, a nonpsychoactive Cannabinoid, in autoimmune T cells. J Neuroinflamm 13: 136.
- Linge R, Jiménez-Sánchez L, Campa L, Pilar-Cuéllar F, Vidal R, et al. (2016) Cannabidiol induces rapid-acting antidepressant-like effects and enhances cortical 5-HT/glutamate neurotransmission: role of 5-HT1A receptors. Neuropharmacol 103: 16-26.
- Lu Y, Anderson HD (2017) Cannabinoid signaling in health and disease. Can J Physiol Pharmacol 95: 311-327.
- Mahgoub M, Keun-Hang SY, Sydorenko V, Ashoor A, Kabbani N, et al. (2013) Effects of Cannabidiol on the function of α7-nicotinic acetylcholine receptors. Eur J Pharmacol 720: 310-319.
- Morales P, Hurst DP, Reggio PH (2017) Molecular Targets of the PhytoCannabinoids-A Complex Picture. Prog Chem Org Nat Prod 103: 103-131.
- Bolognini D, Rock EM, Cluny NL, Cascio MG, Limebeer CL, et al. (2013) Cannabidiolic acid prevents vomiting in Suncus murinus and nausea-induced behaviour in rats by enhancing 5-HT1A receptor activation. Br J Pharmacol 168: 1456-1470.
- Takeda S, Misawa K, Yamamoto I, Watanabe K (2008) Cannabidiolic acid as a selective cyclooxygenase-2 inhibitory component in Cannabis. Drug Metab Dispos 36: 1917-1921.
- Appendino G, Chianese G, Taglialatela-Scafati O (2011) Cannabinoids: occurrence and medicinal chemistry. Curr Med Chem 18: 1085-1099.
- Lowin T, Straub RH (2015) Cannabinoid-based drugs targeting CB1 and TRPV1, the sympathetic nervous system, and arthritis. Arthritis Res Ther 17: 226.
- McPartland JM, MacDonald C, Young M, Grant PS, Furkert DP, et al. (2017) Affinity and efficacy studies of tetrahydroCannabinolic acid A at Cannabinoid receptor types one and two. Cannabis Cannabinoid Res 2: 87-95.
- Moreno-Sanz G (2016) Can You Pass the Acid Test? Critical Review and Novel Therapeutic Perspectives of Δ9-TetrahydroCannabinolic Acid A. Cannabis Cannabinoid Res 1: 124-130.
- Nadal X, Del Río C, Casano S, Palomares B, Ferreiro-Vera C, et al. (2017) TetrahydroCannabinolic acid is a potent PPARγ agonist with neuroprotective activity. Br J Pharmacol 174: 4263-4276.
- Borrelli F, Pagano E, Romano B, Panzera S, Maiello F, et al. (2014) Colon carcinogenesis is inhibited by the TRPM8 antagonist Cannabigerol, a Cannabis-derived non-psychotropic Cannabinoid. Carcinogenesis 35: 278-297.
- Borrelli F, Fasolino I, Romano B, Capasso R, Maiello F, et al. (2013) Beneficial effect of the non-psychotropic plant Cannabinoid Cannabigerol on experimental inflammatory bowel disease. Biochem Pharmacol 85: 1306-1316.
- Cascio MG, Gauson LA, Stevenson LA, Ross RA, Pertwee RG (2010) Evidence that the plant Cannabinoid Cannabigerol is a highly potent α2-adrenoceptor agonist and moderately potent 5HT1A receptor antagonist. Br J Pharmacol 159: 129-141
- McPartland JM, Russo EB (2002) Cannabis and Cannabis extracts: Greater than the sum of their parts? J Cannabis Therapeutics 1: 103-132.
- Romano B (2014) Non-psychotropic phytoCannabinoids in intestinal inflammation and colon cancer. University of Naples Federico II, Italy.
- Russo EB (2011) Taming THC: potential Cannabis synergy and phytoCannabinoid-terpenoid entourage effects. Brit J Pharmacol 163: 1344-1364.
- Giese MW, Lewis MA, Giese L, Smith KM (2015) Development and validation of a reliable and robust method for the analysis of Cannabinoids and terpenes in Cannabis. J AOAC Int 98: 1503-1522.
- Casano S, Grassi G, Martini V, Michelozzi M (2011) Variations in terpene profiles of different strains of Cannabis sativa L. Acta Hortic 925: 115-121.
- Rao VS, Menezes AM, Viana GS (1990) Effect of myrcene on nociception in mice. J Pharm Pharmacol 42: 877-878.
- Hillig KW (2004) A chemotaxonomic analysis of terpenoid variation in Cannabis. Biochem Systemat Ecol 32: 875-891.
- Elzinga S, Fischedick J, Podkolinski R, Raber JC (2015) Cannabinoids and terpenes as chemotaxonomic markers in Cannabis. Nat Prod Chem Res 3: 181.
- Miyazawa M, Yamafuji C (2005) Inhibition of acetylcholinesterase activity by bicyclic monoterpenoids. J Agric Food Chem 53: 1765-1768.
- Pinheiro Mde A, Magalhães RM, Torres DM, Cavalcante RC, Mota FS, et al. (2015) Gastroprotective effect of alpha-pinene and its correlation with antiulcerogenic activity of essential oils obtained from Hyptis species. Pharmacogn Mag 11: 123-130.
- Srivastava N, Chauhan AS, Sharma B (2012) Isolation and characterization of some phytochemicals from Indian traditional plants. Biotechnol Res Int 2012: 549850.
- Cabrera C, Artacho R, Giménez R (2006) Beneficial effects of green tea--a review. J Am Coll Nutr 25: 79-99.
- Hertog MG, Feskens EJ, Hollman PC, Katan MB, Kromhout D (1994) Dietary flavonoids and cancer risk in the Zutphen Elderly Study. Nutr Cancer 22: 175-184.
- Shehzad A, Anwar MN, Zahid H, Ravinayagam V, Al-Rumaih HS, et al. (2016) Multifactorial role of flavonoids in prevention and treatment of various cancers. An Real Acad Farm 82: 297-302.
- Shukla S, Gupta S (2010) Apigenin: a promising molecule for cancer prevention. Pharm Res 2010 27: 962-978.
- Dzialo M, Mierziak J, Korzun U, Preisner M, Szopa J, et al. (2016) The potential of plant phenolics in prevention and therapy of skin disorders. Int J Mol Sci 17:160.
- Namratha V, Merugu R, Devanuri N (2015) Natural products with special reference to pharmacological effects of flavonoids: a mini review. Int J of Pharm Tech Research 8: 26-31.
- Tiwari SC, Husain N (2017) Biological activities and role of flavonoids in human health-a review. Indian J Sci Res 12: 193-196.
- Asif M, Khodadadi E (2013) Medicinal uses and chemistry of flavonoid contents of some common edible tropical plants. J Paramedical Sciences 4: 119-138.
- Clark MN, Bohm BA (1979) Flavonoid variation in Cannabis L. Botanical J Linnean Society 79: 249-257.
- Clark MN (1978) A study of infraspecific flavonoid variation of Cannabis sativa L. (Cannabaceae). University of British Columbia, Vancouver, Canada.
- Badal S, Smith KN, Rajnarayanan R (2017) Analysis of natural product regulation of Cannabinoid receptors in the treatment of human disease. Pharmacol Ther 180: 24-48.
- Chicca A, Marazzi J, Gertsch J (2012) The antinociceptive triterpene β-amyrin inhibits 2-arachidonoylglycerol (2-AG) hydrolysis without directly targeting Cannabinoid receptors. Br J Pharmacol 167: 1596-1608.
- Scalvini L, Piomelli D, Mor M (2016) Monoglyceride lipase: structure and inhibitors. Chem Phys Lipids 197: 13-24.
- Sharma C, Sadek B, Goyal SN, Sinha S, Kamal MA, et al. (2015) Small molecules from nature targeting G-protein coupled Cannabinoid receptors: Potential leads for drug discovery and development. Evid-Based Complement Alternat Med 2015: 238482.
- Johnston GA, Hanrahan JR, Chebib M, Duke RK, Mewett KN (2006) Modulation of ionotropic GABA receptors by natural products of plant origin. Adv Pharmacol 54: 285-316.
- Sucher NJ, Carles MC (2015) A pharmacological basis of herbal medicines for epilepsy. Epilepsy Behav 52: 308-318.
- Gertsch J (2008) Anti-inflammatory Cannabinoids in diet: towards a better understanding of CB(2) receptor action? Commun Integr Biol 1: 26-28.
- Sharma C, Al Kaabi JM, Nurulain SM, Goyal SN, Kamal MA, et al. (2016) Polypharmacological properties and therapeutic potential of β-caryophyllene: a dietary phytoCannabinoid of pharmaceutical promise. Curr Pharm Des 22: 3237-3264.
- Paula-Freire LI, Andersen ML, Gama VS, Molska GR, Carlini EL (2014) The oral administration of trans-caryophyllene attenuates acute and chronic pain in mice. Phytomedicine 21: 356-362.
- Rigano D, Sirignano O, Taglialatela-Scafati O (2017) The potential of natural products for targeting PPARα. Acta Pharmaceutica Sinica B 7: 427-438.
- Rufino AT, Ribeiro M, Sousa C, Judas F, Salgueiro L, et al. (2015) Evaluation of the anti-inflammatory, anti-catabolic and pro-anabolic effects of E-caryophyllene, myrcene and limonene in a cell model of osteoarthritis. Eur J Pharmacol 750: 141-150.
- Fernandes ES, Passos GF, Medeiros R, da Cunha FM, Ferreira J, et al. (2007) Anti-inflammatory effects of compounds alpha-humulene and (-)-trans-caryophyllene isolated from the essential oil of Cordia verbenacea. Eur J Pharmacol 569: 228-236.
- Rogerio AP, Andrade EL, Leite DF, Figueiredo CP, Calixto JB (2009) Preventive and therapeutic anti-inflammatory properties of the sesquiterpene alpha-humulene in experimental airways allergic inflammation. Br J Pharmacol 158: 1074-1087.
- Satsu H, Matsuda T, Toshimitsu T, Mori A, Mae T, et al. (2004) Regulation of interleukin-8 secretion in human intestinal epithelial Caco-2 cells by alpha-humulene. Biofactors 21: 137-139.
- de Cássia da Silveira e Sá R, Andrade LN, de Sousa DP (2013) A Review on anti-inflammatory activity of monoterpenes. Molecules 18: 1227-1254.
- McPartland JM (1997) Cannabis as repellent and pesticide. J Int Hemp Assoc 4: 89-94.
- Yu L, Yan J, Sun Z (2017) D-limonene exhibits anti-inflammatory and antioxidant properties in an ulcerative colitis rat model via regulation of iNOS, COX-2, PGE2 and ERK signaling pathways. Mol Med Rep 15: 2339-2346.
- Jun HJ, Lee HJ, Kim J, Jia Y, Kim KH, et al. (2014) Linalool is a PPARα ligand that reduces plasma TG levels and rewires the hepatic transcriptome and plasma metabolome. J Lipid Res 55: 1098-1110.
- Souto-Maior FN, Fonseca DV, Salgado PR, Monte LO, de Sousa DP, et al. (2017) Antinociceptive and anticonvulsant effects of the monoterpene linalool oxide. Pharm Biol 55: 63-67.
- Guimarães AG, Quintans JS, Quintans LJ Jr (2013) Monoterpenes with analgesic activity--a systematic review. Phytother Res 27: 1-15.
- Chan WK, Tan LTH, Chan KG, Lee LH, Goh BH (2016) Nerolidol: a sesquiterpene alcohol with multi-faceted pharmacological and biological activities. Molecules 21: 529.
- Kim DS, Lee HJ, Jeon YD, Han YH, Kee JY, et al. (2015) Alpha-Pinene Exhibits Anti-Inflammatory Activity Through the Suppression of MAPKs and the NF-κB Pathway in Mouse Peritoneal Macrophages. Am J Chin Med 43: 731-742.
- Barreto RS, Albuquerque-Júnior RL, Araújo AA, Almeida JR, Santos MR, et al. (2014) A systematic review of the wound-healing effects of monoterpenes and iridoid derivatives. Molecules 19: 846-862.
- Aydin E, Türkez H, Tasdemir S (2013) Anticancer and antioxidant properties of terpinolene in rat brain cells. Arh Hig Rada Toksikol 64: 415-424.
- Ito K, Ito M (2013) The sedative effect of inhaled terpinolene in mice and its structure-activity relationships. J Nat Med 67: 833-837.
- Almeida V, Levin R, Peres FF, Niigaki ST, Calzavara MB, et al: Cannabidiol exhibits anxiolytic but not antipsychotic property evaluated in the social interaction test. Prog Neuropsychopharmacol Biol Psychiatry 41: 30-35.
- Hassan SM, Khalaf MM, Sadek SA, Abo-Youssef AM (2017) Protective effects of apigenin and myricetin against cisplatin-induced nephrotoxicity in mice. Pharm Biol 55: 766-774.
- Pallauf K, Duckstein N, Hasler M, Klotz L-O, Rimbach G (2017) Flavonoids as Putative Inducers of the Transcription Factors Nrf2, FoxO, and PPARγ. Oxid Med Cell Longev 2017:4397340.
- Panche AN, Diwan AD, Chandra SR (2016) Flavonoids: an overview. J Nutr Sci 5: 47.
- Paris D, Mathura V, Ait-Ghezala G, Beaulieu-Abdelahad D, Patel N, et al. (2011) Flavonoids lower Alzheimer’s Aß production via an NFkB dependent mechanism. Biomedical Informatics 6: 229-236.
- 86. Ramos AA, Lima CF, Pereira-Wilson C (2011) DNA Damage Protection and Induction of Repair by Dietary Phytochemicals and Cancer Prevention: What Do We Know? In: Chen C (ed.). Selected topics in DNA repair. IntechOpen Limited, London, UK.
- Weiskirchen R (2015) Hepatoprotective and anti-fibrotic agents: It’s time to take the next step. Front Pharmacol 6: 303.
- Ramirez MR (2016) Potential health benefits of Cannabis extracts: a review. Int J Chemical Biomed Sci 2: 1-8.
- Ibrahim AK, Radwan MM, Ahmed SA, Slade D, Ross SA, et al. (2010) Microbial metabolism of cannflavin A and B isolated from Cannabis sativa. Phytochemistry 71: 1014-1019.
- Barrett ML, Gordon D, Evans FJ (1985) Isolation from Cannabis sativa L. of cannflavin--a novel inhibitor of prostaglandin production. Biochem Pharmacol 34: 2019-2024.
- Domitrovi? R, Poto?njak I (2016) A comprehensive overview of hepatoprotective natural compounds: mechanism of action and clinical perspectives. Arch Toxicol 90: 39-79.
- Gertsch J (2017) Cannabimimetic phytochemicals in the diet - an evolutionary link to food selection and metabolic stress adaptation? Br J Pharmacol 174: 1464-1483.
- McPartland JM, Guy GW, Di Marzo V (2014) Care and feeding of the endoCannabinoid system: A systematic review of potential clinical interventions that upregulate the endoCannabinoid system. PLoS One 9: 89566.
- Calderón-Montaño JM, Burgos-Morón E, Pérez-Guerrero C, López-Lázaro M (2011) A Review on the Dietary Flavonoid Kaempferol. Mini Rev Med Chem 11: 298-344.
- Devi KP, Malar DS, Nabavi SF, Sureda A, Xiao J, et al. (2015) Kaempferol and inflammation: From chemistry to medicine. Pharmacol Res 99: 1-10.
- Kumar S, Pandey AK (2013) Chemistry and biological activities of flavonoids: an overview. The ScientificWorld J 2013: 1-16.
- Weiskirchen R (2015) Hepatoprotective and anti-fibrotic agents: It’s time to take the next step. Front Pharmacol 6: 303.
- Knickle AF (2016) The dietary phytochemical myricetin induces ROS-dependent breast cancer cell death. Dalhousie University, Halifax, Nova Scotia, Canada.
- Aquila S, Santoro M, De Amicis F, Guido C, Bonofiglio D, et al. (2013) Red wine consumption may affect sperm biology: the effects of different concentrations of the phytoestrogen myricetin on human male gamete function. Mol Reprod Dev 80: 155-165.
- Bai HW, Zhu BT (2008) Strong activation of cyclooxygenase I and II catalytic activity by dietary bioflavonoids. J Lipid Res 49: 2557-2570.
- Ang ES, Yang X, Chen H, Liu Q, Zheng MH, et al. (2011) Naringin abrogates osteoclastogenesis and bone resorption via the inhibition of RANKL-induced NF-κB and ERK activation. FEBS Lett 585: 2755-2762.
- Alam MA, Subhan N, Rahman MM, Uddin SJ, Reza HM, et al. (2014) Effect of citrus flavonoids, naringin and naringenin, on metabolic syndrome and their mechanisms of action. Adv Nutr 5: 404-417.
- Park SK, Seo JB, Lee MY (2012) Proteomic profiling of hempseed proteins from Cheungsam. Biochim Biophys Acta 1824: 374-382.
- Al Mamun MA, Hosen MJ, Islam K, Khatun A, Alam MM, et al. (2015) Tridax procumbens flavonoids promote osteoblast differentiation and bone formation. Biol Res 48: 65.
- Lam KY, Ling APK, Koh RY, Wong YP, Say YH (2016) A Review on Medicinal Properties of Orientin. Adv Pharmacol Sci 2016: 4104595.
- Xiao Q, Qu Z, Zhao Y, Yang L, Gao P (2017) Orientin Ameliorates LPS-Induced Inflammatory Responses through the Inhibitory of the NF-κB Pathway and NLRP3 Inflammasome. Evidence-Based Complement Alternat Med 2017: 2495496.
- Anhê GF, Okamoto MM, Kinote A, Sollon C, Lellis-Santos C, et al. (2012) Quercetin decreases inflammatory response and increases insulin action in skeletal muscle of ob/ob mice and in L6 myotubes. Eur J Pharmacol 689: 285-293.
- Yamaguchi M, Weitzmann MN (2011) Quercetin, a potent suppressor of NF-κB and Smad activation in osteoblasts. Int J Mol Med 28: 521-525.
- Salvamani S, Gunasekaran B, Shaharuddin NA, Ahmad SA, Shukor MY (2014) Antiartherosclerotic Effects of Plant Flavonoids. BioMed Res Int 2014: 480258.
- Serban MC, Sahebkar A, Zanchetti A, Mikhailidis DP, Howard G, et al. (2016) Effects of Quercetin on Blood Pressure: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. J Am Heart Assoc 5: 002713.
- Wang F, Yin J, Ma Y, Jiang H, Li Y (2017) Vitexin alleviates lipopolysaccharide?induced islet cell injury by inhibiting HMGB1 release. Mol Med Rep 15: 1079-1086.
- Gupta SC, Tyagi AK, Deshmukh-Taskar P, Hinojosa M, Prasad S, et al. (2014) Downregulation of tumor necrosis factor and other proinflammatory biomarkers by polyphenols. Arch Biochem Biophys 559: 91-99.
- Abbasi MA, Taha MO, Zhilif MA, Disi AM (2013) β-Caryophyllene causes regression of endometrial implants in a rat model of endometriosis without affecting fertility. Eur J Pharmacol 702: 12-19.
- Aslam MS, Ahmad MS, Mamat AS (2015) Pharmacological potential of vitexin. Ind Res J Pharm & Sci 2: 114-122.
- Wang D, Sun-Waterhouse D, Li F, Xin L, Li D (2018) MicroRNAs as molecular targets of quercetin and its derivatives underlying their biological effects: A preclinical strategy. Crit Rev Food Sci Nutr 15: 1-13.
- Bonn-Miller MO, Loflin MJE, Thomas BF, Marcu JP, Hyke T, et al. (2017) Labeling Accuracy of Cannabidiol Extracts Sold Online. JAMA 318: 1708-1709.
- Ruth AC, Gryniewicz-Ruzicka CM, Trehy ML, Kornspan N, Coody G (2016) Consistency of Label Claims of Internet-Purchased Hemp Oil and Cannabis Products as Determined using IMS and LC-MS : A Marketplace Survey. J Regul Sci 3: 1-6.
- Vandrey R, Raber JC, Raber ME, Douglass B, Miller C, et al. (2015) Cannabinoid Dose and Label Accuracy in Edible Medical Cannabis Products. JAMA 313: 2491-2493.
- Sexton M, Shelton K, Haley P, West M (2018) Evaluation of Cannabinoid and Terpenoid Content: Cannabis Flower Compared to Supercritical CO2 Concentrate. Planta Med 84: 234-241.
- Bócsa I, Máthé P, Hangyel L (1997) Effect of nitrogen on tetrahydroCannabinol (THC) content in hemp (Cannabis sativa L.) leaves at different positions. J Int Hemp Association 4: 78-79.
- Hanus L, Dostalova M (1994) The effect of soil fertilization on the formation and the amount of Cannabinoid substances in Cannabis sativa L. in the course of one vegetation period. Acta Univ Palacki Olomuc Fac Med 138: 11-15.
- Sikora V, Berenji J, Latkovic D (2011) Influence of agroclimatic conditions on content of main Cannabinoids in industrial hemp (Cannabis sativa L.). Genetika 43: 449-456.
- Russo EB (2016) Current Therapeutic Cannabis Controversies and Clinical Trial Design Issues. Front Pharmacol 7: 309.
- Hemphill JK, Turner JC, Mahlberg PG (1980) Cannabinoid Content of Individual Plant Organs From Different Geographical Strains of Cannabis sativa L. J Nat Prod 43: 112-122.
- Latta RP, Eaton BJ (1975) Seasonal fluctuations in Cannabinoid content of Kansas Marijuana. Economic Botany 29: 153-163.
- Pa?a C, Kiliç T, Esendal E (2017) A research on determination of ontogenetic and diurnal variation of essential oil content and composition in Hypericum kazdaghensisii growing wild in Ida. Scientific Papers Series A. Agronomy 60: 364-369.
- Ayan AK, Yanar P, Cirak C, Bilgener M (2007) Morphogenetic and diurnal variation of total phenols in some Hypericum species from Turkey during their phenological cycles. Bangladesh J Bot 36: 39-46.
- Ross SA, ElSohly MA (1996) The volatile oil composition of fresh and air-dried buds of Cannabis sativa. J Nat Prod 59: 49-51.
- Janatová A, Fra?ková A, Tlustoš P, Hamouz K, Božik M, et al. (2018) Yield and Cannabinoids contents in different Cannabis (Cannabis sativa L.) genotypes for medical use. Indust Crops Products 112: 363-367.
- Singh Y, Bali C (2013) Cannabis Extract Treatment for Terminal Acute Lymphoblastic Leukemia with a Philadelphia Chromosome Mutation. Case Rep Oncol 6: 585-592.
- Saccà F, Pane C, Carotenuto A, Massarelli M, Lanzillo R, et al. (2016) The use of medical-grade Cannabis in patients non-responders to Nabiximols. J Neurol Sci 368: 349-351.
- Corral VL (2001) Differential Effects of Medical Marijuana Based on Strain and Route of Administration. J Cannabis Therapeutics 1: 43-59.
- Colizzi M, Bhattacharyya S (2017) Does Cannabis Composition Matter? Differential Effects of Delta-9-tetrahydroCannabinol and Cannabidiol on Human Cognition. Curr Addict Rep 4: 62-74.
- Crippa JAS, Crippa ACS, Hallak JEC, Martin-Santos R, Zuardi AW (2016) Δ9-THC Intoxication by Cannabidiol-Enriched Cannabis Extract in Two Children with Refractory Epilepsy: Full Remission after Switching to Purified Cannabidiol. Front Pharmacol 7: 359.
- Capasso R, Aviello G, Borrelli F, Romano B, Ferro M, et al. (2011) Inhibitory effect of standardized Cannabis sativa extract and its ingredient Cannabidiol on rat and human bladder contractility. Urology 77: 9-15.
- Comelli F, Giagnoni G, Bettoni I, Colleoni M, Costa B (2008) Antihyperalgesic effect of a Cannabis sativa extract in a rat model of neuropathic pain: mechanisms involved. Phytother Res 22: 1017-1024.
- Gallily R, Yekhtin Z, Hanuš, LO (2015) Overcoming the Bell-Shaped Dose-Response of Cannabidiol by Using Cannabis Extract Enriched in Cannabidiol. Pharmacology & Pharmacy 6: 75-85.
- Ligresti A, Moriello AS, Starowicz K, Matias I, Pisanti S, et al. (2006) Antitumor activity of plant Cannabinoids with emphasis on the effect of Cannabidiol on human breast carcinoma. J Pharmacol Exp Ther 318: 1375-1387.
- Ryan D, Drysdale AJ, Pertwee RG, Platt B (2006) Differential effects of Cannabis extracts and pure plant Cannabinoids on hippocampal neurones and glia. Neurosci Lett 408: 236-241.
- Scott KA, Dalgleish AG, Liu WM (2014) The combination of Cannabidiol and Δ9-tetrahydroCannabinol enhances the anticancer effects of radiation in an orthotopic murine glioma model. Mol Cancer Ther 13: 2955-2967.
- Wilkinson JD, Whalley BJ, Baker D, Pryce G, Constanti A, et al. (2003) Medicinal Cannabis: is delta-9-tetrahydroCannabinol necessary for all its effects? J Pharm Pharmacol 55: 1687-1694.
Citation: Nahler G, Jones TM (2018) Pure Cannabidiol versus Cannabidiol-Containing Extracts: Distinctly Different Multi-Target Modulators. J Altern Complement Integr Med 4: 048.
Copyright: © 2018 Gerhard Nahler, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

