
Purtscher-Like Retinopathy after Cocaine Abuse, Optic Coherence Tomography Findings: Case Report and Literature Review
*Corresponding Author(s):
Maria Larranaga CoresOphthalmology Service, La Paz University Hospital, Madrid, Paseo De La Castellana, 261, 28046, Madrid, Spain
Email:marialarranagacores@gmail.com
Abstract
Purpose: To report a case of Purtscher-like retinopathy after cocaine abuse, to describe the ophthalmic findings and its correlation with the Optical Coherence Tomography images, differential diagnosis and finally to discuss the possible causes that lead to these presentation.
Case Description: 39-year-old male who came to the Emergency Department presenting hemoptysis, symptoms consistent with acute renal failure and acute bilateral blurred vision after an episode of cocaine abuse. Funduscopy in both eyes revealed retinal haemorrhages, multiple bilateral peripapillary cotton wool spots and “Purtscher Fleckens”, and the Optic Coherence Tomography intraretinal fluid and bilateral papillary oedema. In addition, a hyper reflectivity of intermediate layers was appreciated in OCT, showing the capillaries that suffer from isquemia. The patient received a diagnosis of Purtscher-like retinopathy based on retinal findings and the absence of a history of trauma. He was urgently admitted to the nephrology unit due to thrombotic microangiopathy findings, in addition to alveolar hemorrhage, cardiomyopathy and acute subdural hematoma. The patient was diagnosed with atypical hemolytic uremic syndrome (aHUS), whose trigger may have been cocaine intake. Systemic Eculizumab was added to the hemodialysis and plasmapheresis treatment.
Keywords
Purtscher-like retinopathy; Optic Coherence Tomography; Substance abuse; Atypical hemolytic uremic syndrome; Thrombotic microangiopathy
Introduction
Purtscher's retinopathy was described for the first time in 1910 by Otmar Purtscher as an occlusive microvascular vasculopathy associated with severe head trauma. [1, 2] Since then, a similar retinal appearance has also been described in a variety of non-traumatic conditions, and the term ‘’Purtscher-like retinopathy’’ is used to describe it. [2] That group of conditions includes systemic disorders such as acute pancreatitis, fat embolism syndrome, renal failure, childbirth, connective tissue disorders, autoimmune processes or thrombotic microangiopathic diseases like hemolytic uremic syndrome (HUS), disseminated intravascular coagulation (DIC), or thrombotic thrombocytopenic purpura (TTP) [1-3] The estimated incidence both for Purtscher and Purtscher-like retinopathies is 0.24 persons per million per year. [1] It is presented bilateral in up to 60% of cases [1, 2].
Fundoscopy is characteristic, and includes cotton-wool spots and intraretinal haemorrhages in up to 90%, as well as the pathognomonic “Purtscher flecken”. [1] This characteristic finding is described as intraretinal whitening with a polygonal margin, but with a clear area (within 50 µm) between the affected retina and the adjacent arteriole; and it´s present in only half of the cases, although under diagnosis is suspected [1, 2]. All these abnormalities are confined to the posterior pole, within the macula and immediately nasal to the optical disk [2].
The most commonly accepted cause for Purtscher-like retinopathy is precapillary arteriole occlusion by micro embolization, resulting in microvascular infarction of the retinal nerve fiber layer; though other factors leading to blood-vessel damage may also contribute. [2] Complement disregulation in pathologies such as atypical hemolytic uremic syndrome (aHUS) (characterized by microangiopathic hemolytic anemia, thrombocytopenia, and acute kidney failure) has also been described as a potential cause of Purtscher´s retinopathy [3-9].
In recent years there has been increasing interest in the definition and interpretation of this retinopathy in Optical Coherence Tomography (OCT), in order to better understand the precise anatomical localization affected in the retina and which is the evolution it has once the appreciable findings on funduscopy have disappeared. The interest lies in correlating these persistent changes with final visual acuity, as well as establishing new prognostic factors based on OCT images at the initial time of diagnosis. Some of these OCT findings were also demonstrated in a number of conditions such as paracentral acute middle maculopathy (PAMM), suggesting that this is likely not a single diagnostic entity, but rather a final common pathway of ischemic condition [10-12].
Clinical Case
A 39-year-old male who went to the emergency room after drug use, presenting hemoptysis, malaise and blurred vision. Laboratory tests of the patient revealed hemoglobinemia (9.9 g/dL), thrombocytopenia (62,000/uL), leukocytes (12,300/uL), increased levels of blood lactate dehydrogenase (1895 U/L), creatinine (8.89 mg/dL), filtrate glomerular (7ml/min), and blood urea nitrogen (201 mg/dL). Blood pressure was 170/110 mmHg. As background, the patient reported regular consumption of snorted cocaine (200mg every 3 days), snorted ketamine, LSD, smoked THC, tobacco, benzodiazepines and alcohol.
On ophthalmologic examination, his best-corrected visual acuity measured on the Snellen scale was 20/25 in the right eye and 20/20 in the left eye. Anterior segment examination was unremarkable and intraocular pressures were within normal limits. Dilated fundus examination revealed multiple cotton wool spots and intraretinal hemorrhages confined to the peripapilary and the posterior pole and around the optic nerve, as well as “Purtscher flecken” (Figures 1A, 1B). Disc swelling and bilateral macular edema were observed.
He was diagnosed with alveolar hemorrhage, cardiomyopathy, and thrombotic microangiopathy: with Coombs-negative hemolytic anemia, thrombocytopenia, and rapidly progressive acute renal failure. The patient was hospitalized in the nephrology clinic and he was scheduled for hemodialysis and plasmapheresis. He also started treatment with methylprednisolone pulses.
Two days after admission, OCT was performed, revealing bilateral optic disc edema with mean retinal nerve fiber layer (RNFL) thickness of 182/160 microns (Figure 2), retinal thickening especially at the inner layer and bilateral macular neurosensory detachments (Figures 3A, 3B). High definition OCT section at the level of exudates showed increased thickness and hyper-reflectivity of internal layers compatible with the cotton-wool spots seen in the funduscopy (Figure 4). Due to the associated retinal findings and lack of a history of trauma, the patient was diagnosed with Purtscher-like retinopathy.
During admission, laboratory studies were expanded with the following results: negative ANCA, negative antiGBM, elevated complement activity, negative serologies, negative tumor markers, negative Shiga toxin and negative ADAMTS13 test (which excluded thrombotic thrombocytopenic purpura as a possible cause of thrombotic microangiopathy).
After an extensive differential diagnosis, the condition was finally diagnosed as atypical hemolytic uremic syndrome (aHUS) whose trigger was cocaine use. Eculizumab was added to the treatment with a favorable evolution, both at the systemic level and at the ophthalmic level (observed using OCT and serial funduscopies). One month after admission, vision was 20/20 in both eyes and there was a significant decrease in the number of exudates and hemorrhages on funduscopy and resolution of disc swelling and macular oedema in both eyes on OCT (Figures 2, 5A, 5B and 6A, 6B).
The patient consented to publication of the case and its images in writing.
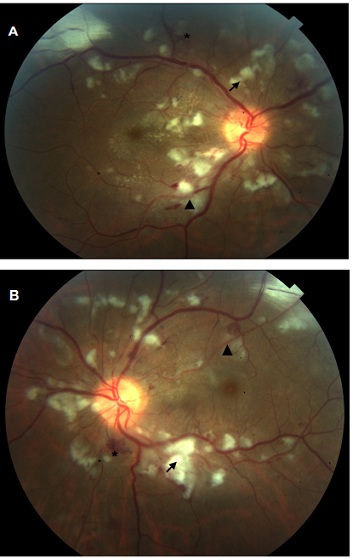 Figure 1: Fundus of right (A) and left (B) eyes showing intraretinal haemorrhages (asterisk), cotton-wool sports (arrow) and soft exudates with Purtscher flecken appearance (triangle).
Figure 1: Fundus of right (A) and left (B) eyes showing intraretinal haemorrhages (asterisk), cotton-wool sports (arrow) and soft exudates with Purtscher flecken appearance (triangle).
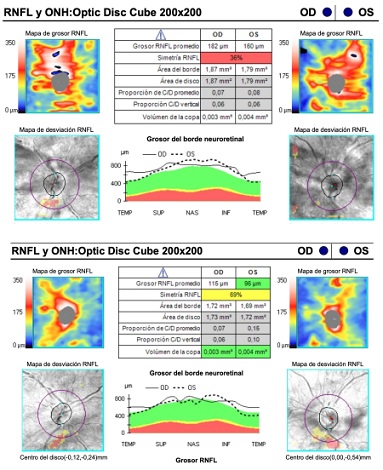 Figure 2: Superior: retinal nerve fiber layer OCT from the patient at admission showing optic disc swelling. Inferior: retinal nerve fiber layer OCT from the patient one month after treatment showing resolution of the optic disc swelling.
Figure 2: Superior: retinal nerve fiber layer OCT from the patient at admission showing optic disc swelling. Inferior: retinal nerve fiber layer OCT from the patient one month after treatment showing resolution of the optic disc swelling.
 Figure 3: High definition optical coherence tomography cross-sectional scan through the fovea of the right (A) and left (B) eye at admission. There is markedly increased hyper reflectivity of the outer plexiform. Serous macular detachment at optical coherence tomography in both eyes were presented.
Figure 3: High definition optical coherence tomography cross-sectional scan through the fovea of the right (A) and left (B) eye at admission. There is markedly increased hyper reflectivity of the outer plexiform. Serous macular detachment at optical coherence tomography in both eyes were presented.
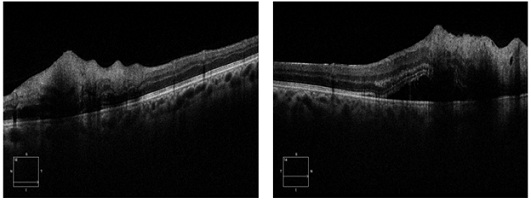 Figure 4: High definition optical coherence tomography section at the level of exudates. Increased thickness and hyper-reflectivity of internal layers compatible with the cotton-wool spots seen in the funduscopy.
Figure 4: High definition optical coherence tomography section at the level of exudates. Increased thickness and hyper-reflectivity of internal layers compatible with the cotton-wool spots seen in the funduscopy.
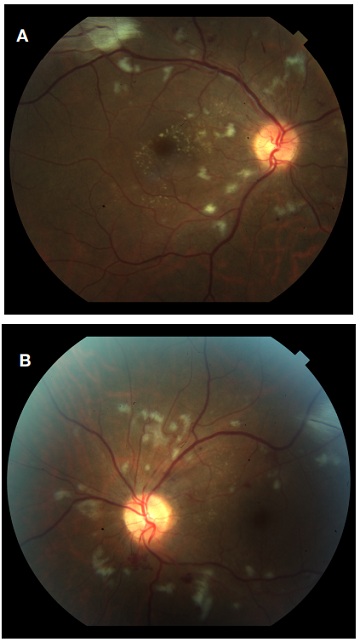 Figure 5: Funduscopy of the right (A) and left (B) eye one month after treatment. A decrease in the number of exudates and hemorrhages in the posterior pole can be seen, as well as almost disappearance of the flecken.
Figure 5: Funduscopy of the right (A) and left (B) eye one month after treatment. A decrease in the number of exudates and hemorrhages in the posterior pole can be seen, as well as almost disappearance of the flecken.
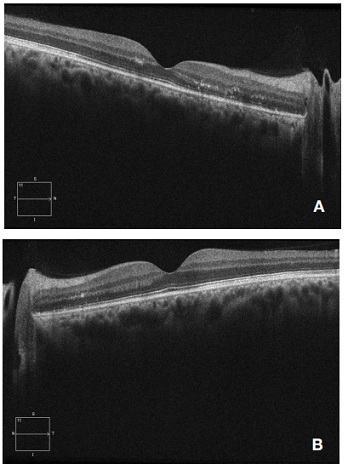 Figure 6: High definition optical coherence tomography cross-sectional scan through the fovea of the right (A) and left (B) eye one month after treatment. A resolution of the serous detachment is appreciated, although hyper reflective points persist in the middle layers.
Figure 6: High definition optical coherence tomography cross-sectional scan through the fovea of the right (A) and left (B) eye one month after treatment. A resolution of the serous detachment is appreciated, although hyper reflective points persist in the middle layers.
Discussion
Atypical hemolytic uremic syndrome (aHUS) is a rare and life-threatening condition presented in 5-10% of Hemolytic Uremic Syndrome (HUS) cases. [3, 5] A negative Shiga toxin test and the absence of diarrhea differentiates aHUS from typical HUS [5].
aHUS it´s characterized by a dysregulation of the complement alternative pathway that causes it’s chronic and uncontrolled activation. [5] This leads to vascular endothelial damage and complement aggregations: thickening of arterioles and capillaries, endothelial detachment, subendothelial accumulation of proteins, cell debris, and fibrin-platelet thrombi obstruction. [3,9] Activation of the complement system also results in formation of the membrane attack complex on cell surfaces, especially endothelial cells of the microcirculation such as the kidney or retina. [3] This leads to obstruction by fibrin-platelet thrombi and systemic multi-organ dysfunction. [3] The diagnostic suspicion is usually a thrombotic microangiopathy [3], like in our case. The triad of clinical features (microangiopathic hemolytic anemia, thrombocytopenia, and acute kidney injury) constitutes hemolytic uremic syndrome (HUS). [3, 6] Laboratory findings are usually; thrombocytopenia, anemia, elevated lactate dehydrogenase (LDH), elevated blood urea nitrogen (BUN), and declining kidney function [5].
Until 2013, the main treatment for aHUS was plasmapheresis, which improved platelet counts but did not stop the irregular complement activation that occurs in aHUS. [3] Therefore prognosis was poor: mortality and morbidity rates were high in the acute phase and more than half of the cases progressed to end-stage renal disease. [3] In recent years, the systemic use of the drug Eculizumab has been introduced in these patients, notably improving prognosis and increasing survival. [3] Eculizumab is a humanized monoclonal antibody that binds to C5, thus preventing the formation of the membrane attack complex [3].
aHUS does not usually present ophthalmological manifestations, but there have been described cases of venous and arterial occlusions, serous retinal detachments, vitreous and choroidal haemorrhages. [3] Since 2017, cases of Purtscher-like retinopathy associated with aHUS have also been reported [3-9].
Purtscher-like retinopathy is an occlusive microvasculopathy whose characteristic findings have been postulated to be secondary to thrombotic microangiopathy with endothelial damage and increased platelet consumption. [1-4] The most widely accepted theory to explain the cause of this condition in aHUS is micro embolization of C5 and leukocyte aggregates, causing arteriolar precapillary occlusion and microvascular infarct of the retinal nerve fiber layer and haemorrhages [3, 8, 9].
Fundus examination of Purtscher and Purtscher-like retinopathies typically includes cotton-wool spots (which may cover retinal arterioles and are caused by distal occlusion of the retinal capillaries alone by small emboli) [2], retinal hemorrhages (flame-shaped or dot-and-blot), and the pathognomonic “Purtscher-flecken” (which are typically polygonal and well delimited, never cover neighboring arterioles). [1, 6, 7, 9] There may be other signs at presentation, such as macular edema (22%), optic disk swelling (16%), or a pseudo cherry red spot (26%). [1, 6, 7, 9] Most cases are bilateral and the retinopathy usually presents between 24 and 48 hours after the underlying systemic disease; although unilateral cases have been reported, as well as cases in which retinopathy precedes systemic involvement [2, 6, 7].
As for the characteristic “Purtscher flecken”, they are located in the inner retina between arterioles and venules and leave a clear area that occupies an average of 50 µm between the affected retina and the adjacent arteriole, and that corresponds to the capillary free area on each side of the retinal arteries and precapillary arterioles. [2] The presence of this clear area suggests primary pathology is in pre-capillary arterioles. [2] Peripapillary capillaries have fewer arteriolar feeds and fewer anastamoses, so they may be more susceptible to embolic occlusion [2].
Cases of Purtscher-like retinopathy have also been described after drug use [13-17], as well as after infections such as Dengue [18], or in the context of acute alveolar hemorrhage [19].
Purtscher-like retinopathy does not have a specific treatment, and efforts should be focused on treating the underlying systemic process. [14] Vision recovers to a variable degree in most patients. [14] Agrawal A et al. described in 2006 poor prognostic factors such as coroidal hypoperfusion, involvement of the outer retinal layers, optic disc swelling and leakage on angiography, existence of previous similar episodes and severity of the systemic disease causing the condition [2, 14].
Some of the latest publications on this pathology try to relate the fundus findings with the images obtained in optical coherence tomography (OCT). [3, 10-12, 18, 19, 20] Purtscher’s retinopathy has been shown to represent two ischemic retinal findings on OCT: cotton-wool spots and Purtscher-flecken. [10] “Purtscher-flecken” can be characterized by cross-sectional and en face Spectral Domain OCT (SD-OCT) and represent localized areas of deep capillary ischemia; they lie in the inner nuclear layer, between arterioles. [10-12] They appear in OCT as hyper reflective areas. [10,11] Both the cotton-wool spots and the "Purtscher-flecken" have shown this hyper reflectivity that indicates ischemia in OCT, being the inner plexiform, the inner nuclear, and the outer plexiform layers the most affected in Purtscher and Purtscher-like retinopathies [10].
Important similarities in OCT findings have been appreciable between Purtscher's retinopathy and paracentral acute middle maculopathy (PAMM). [10-12] PAMM was originally described by Sarraf et al as an OCT-based diagnosis presented by hyper reflective band-like lesions in middle retinal layers (specifically the inner nuclear layer) caused by ischemic insult to retinal intermediate and deep capillary plexus. [10-12] As we said, this finding also represents the “Purtscher-flecken” found in Purtscher-like retinopathy. [10] In addition, OCT in Purtscher´s retinopathy shows hyper reflectivity in superficial layers (compatible with cotton wool spots).
Retinal edema or macular atrophy can also be seen in OCT. [14] An characteristic finding in OCT-angiography has been described in Purtscher´s retinopathy: a honeycomb-like hypo signal at the choriocapillaris layer, indicating the involvement and ischaemia of the choroid during the pathological process [20].
AIM Miguel et al described in 2013 five diagnostic Purtscher Retinopathy criteria: Purtscher flecken, retinal hemorrhages, cotton-wool spots (typically restricted to posterior pole), probable or plausible explanatory etiology and complementary investigation compatible with diagnosis. [1] They included in the group Purtscher Retinopathy patients who matched at least three of five criteria. Our patient matched all of them; if we assume the aHUS as a plausible explanatory etiology, (several case reports in the literature support this etiology [3-9]).
In the case of the patient presented here in this article, the final diagnosis was aHUS that could have been precipitated by cocaine intake. The main differential diagnosis considered given the patient's systemic condition was hypertensive retinopathy, which in severe stages also presents with cotton-wool exudates, intraretinal hemorrhages, macular edema, and optic nerve edema. [21] In favor of the diagnosis of hypertensive retinopathy were the following signs: crossing signs and the presence of hard exudates in the right eye forming a macular star. On the other hand, there were also several data in favor of the diagnosis of Purtscher-like retinopathy: the peripapillary distribution of exudates and hemorrhages, the improvement of the funduscopy when treatment with Eculizumab was initiated (despite the patient's systemic tensions not being controlled), and above all the presence of the pathognomonic Purtscher-flecken in our patients funduscopy.
For all these reasons, we consider that, although the final ophthalmological diagnosis of our patient was Purtscher-like retinopathy, it is likely that the funduscopy included characteristics common to those of hypertensive retinopathy that very probably our patient also presented. We remember that his systemic pressures on the day of admission were 170/110 and, associated with the significant renal failure that the patient presented, they did not remain stable in normal ranges throughout the patient's admission.
The main limitation of the case presented was the impossibility of performing a fluorescein angiography, given the patient's general condition throughout his admission.
Conflict of Interests
The authors declare no conflict of interest.
Funding
No funding or grant support.
References
- Miguel A, Henriques F, Azevedo L, Loureiro A, Maberley D (2012) Systematic review of Purtscher’s and Purtscher-like retinopathies. Eye. 27: 1-13.
- Agrawal A, McKibbin M (2006) Purtscher's and Purtscher-like Retinopathies: A Review. Survey of Ophthalmology. 51: 129-136.
- Santamaría ÁJF, Serret CA, Aguayo JA, García OG (2019) Optic coherence tomography angiography follow-up in a case of Purtscher-like retinopathy due to atypical hemolytic uremic syndrome. European Journal of Ophthalmology. 30: NP14-NP17.
- Benvenuto F, Guillen S, Marchiscio L, Falbo J, Fandiño A (2021) Retinopatía Purtscher-like en paciente pediátrico con síndrome urémico hemolítico: reporte de caso y revisión de la literatura. Archivos de la Sociedad Española de Oftalmología. 96: 607-610.
- Gange W, Haghighi A, Toy CB (2021) Purtscher-like Retinopathy Associated with Atypical Hemolytic Uremic Syndrome. RETINAL Cases & Brief Reports. Publish Ahead of Print.
- Patel MR, Bains AK, O’Hara JP, Kallab AM, Marcus DM (2001) Purtscher Retinopathy as the Initial Sign of Thrombotic Thrombocytopenic Purpura/Hemolytic Uremic Syndrome. Archives of Ophthalmology. 119: 1388-1389.
- Sánchez VJ, Castilla MM, Contreras DM, Rueda RT, Molina SFE, et al. (2018) Retinopatía Purtscher- like previa a fallo renal agudo. Archivos de la Sociedad Española de Oftalmología. 93: 198-201.
- Stroh I, Nguyen A, Channa R (2019) Purtscher-Like Retinopathy in Hemolytic Uremic Syndrome. JAMA Ophthalmology. 137: e183911.
- Ustaoglu M, Önder F, Solmaz N, Öztürk S, Ayer M (2017) Purtscher-Like Retinopathy Associated with Atypical Hemolytic Uremic Syndrome. Türk Oftalmoloji Dergisi. 47: 348-350.
- Lujan BJ, Coady PA, McDonald HR (2014) Spectral Domain Optical Coherence Tomography imaging of Purtscher-like Retinopathy.. RETINAL Cases & Brief Reports. 8: 283-285.
- Yalinbas D, Bozali E, Vural A, Kocak H, Erdogan H (2021) Purtscher-like Retinopathy Associated with Crimean-Congo Hemorrhagic Fever: A Case Report. Ocular Immunology and Inflammation. 30: 1016-1019.
- Niyousha MR, Hassanpoor N, Eftekhari A, Mousvi F (2018) Simultaneous paracentral acute middle maculopathy and purtscher-like retinopathy after acute febrile illness. Canadian Journal of Ophthalmology. 53: e184-e186.
- Aseem F, Zamora BG, Kauffman L, Miller PJ, John VJ (2018) Bilateral exudative retinal detachments due to thrombotic microangiopathy associated with intravenous abuse of Opana ER. American Journal of Ophthalmology Case Reports. 11: 72-74.
- Onaran Z, Akbulut Y, Tursun S, Ogurel T, Gökçinar N, et al. (2019) Purtscher-Like Retinopathy Associated with Synthetic Cannabinoid (Bonzai) Use. Turkish Journal of Ophthalmology. 49: 114-116.
- Kirgiz A, Kaldirim H (2017) Bilateral multiple exudative retinal detachments and macular edema in a patient diagnosed with synthetic cannabinoid (Bonzai) intoxication. International Ophthalmology. 39: 237-241.
- Rahman W, Thomas S, Wiselka M, Bibby K (2007) Cocaine-induced chorioretinal infarction. British Journal of Ophthalmology. 92: 150-151.
- Coban-Karatas M, Turunc T, Altan-Yaycioglu R (2012) Purtscher-like Retinopathy Related to Drug-induced Hypersensitivity Syndrome. Ocular Immunology and Inflammation. 20: 475-477.
- Lima L, Vianello S, Pimentel S, Costa de AG, Zett C, et al. (2017) Dengue Fever Presenting as Purtscher-like Retinopathy. Ocular Immunology and Inflammation. 26: 660-665.
- Huang LC, Williams BK, Ko AC, Yehoshua Z, Alabiad CR (2015) Purtscher-Like Retinopathy and Diffuse Alveolar Hemorrhage Caused by Soft Tissue Injection for Gluteal Augmentation. Journal of Clinical & Experimental Ophthalmology. 6: 402.
- Li B, Li D, Chen Y (2019) Purtscher-like retinopathy presented a honeycomb-like pattern in optical coherence topography angiography. BMC Ophthalmology. 19.
- Tsukikawa M, Stacey AW (2020) A Review of Hypertensive Retinopathy and Chorioretinopathy. Clinical Optometry. 12: 67-73.
Citation: Maria LC, Miguel PAB, Monica AD, Jesus PC, Jose Vicente DM, et al. (2023) Purtscher-Like Retinopathy after Cocaine Abuse, Optic Coherence Tomography Findings: Case Report and Literature Review. J Clin Stud Med Case Rep 10: 0151.
Copyright: © 2023 Maria Larranaga Cores, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

