
Targeting Multiple Hallmarks of Aging with Proxylane, Ginkgo biloba leaf extract and Niacinamide Improves Healthspan by Reversing the Aged Skin Phenotype
*Corresponding Author(s):
Franck JuchauxL’Oréal Recherche Et Innovation, Chevilly-Larue, France
Email:franck.juchaux@loreal.com
Abstract
Aging is an intrinsic and inevitable process, characterized by a progressive decline in physiological functions. While unavoidable, it is nonetheless modifiable, a fundamental principle of the longevity approach. Unlike conventional anti-aging strategies, longevity targets the root causes of aging at the cellular level to sustainably maintain skin health and improve skin appearance. In this context, we have developed a specific combination of Proxylane, Ginkgo biloba leaf extract, and niacinamide, designed to target five key hallmarks of skin aging.
To demonstrate its efficacy, we used a skin model reconstructed from aged donor cells, faithfully reproducing an aged phenotype. Our study revealed that this combination effectively reversed the aged phenotype. More specifically, it increased epidermal thickness, drastically reduced the presence of senescent cells in the epidermis, restored autophagy, and stimulated the expression of essential proteins. These included collagens IV, VII, and perlecan, for strengthening the dermo-epidermal junction, as well as collagen I, elastin, and fibrillin, for the dermal extracellular matrix. The combined treatment showed superior results compared to each active ingredient taken individually, highlighting the value of simultaneously targeting multiple pathwaysof aging.
These results clearly demonstrate in vitro the effectiveness of our combination in mitigating age-related issues by addressing five key hallmarks of aging, thus confirming its potential to further improve skin visible aging signs as part of a comprehensive longevity strategy.
Introduction
Aging, an intrinsic process characterized by a progressive decline in physiological functions, is inevitable. But although unavoidable, it is nonetheless modifiable. McCain et al. provided the first evidence of this possibility of modifying aging in 1935, who demonstrated that caloric restriction increased lifespan in animals [1]. However, extending lifespan without maintaining good health and optimal vitality is not desirable. Thus, the very essence of longevity research lies in influencing lifespan while preserving good health for longer, a concept known as healthy longevity.
Aging is intimately linked to the alteration of fundamental biological mechanisms, commonly referred to as hallmarks of aging. These mechanisms have a direct impact on molecular, cellular and tissular functions and are central to the aging process itself. By actively acting on these biological markers, it becomes possible to influence the aging trajectory and thus prolong the period during which an organism remains healthy [2,3]. To date, fourteen distinct markers of aging have been identified: genomic instability, telomere shortening, epigenetic alterations, loss of proteostasis, impaired macroautophagy, dysregulation of nutrient sensing, mitochondrial dysfunction, cellular senescence, changes in the extracellular matrix, stem cell depletion, impaired intercellular communication, chronic inflammation and dysbiosis, and psychological isolation [4].
The skin, like other organs of the body, is not immune to this phenomenon. In response to internal factors (hormonal imbalances, genetic predisposition, etc.) and External Factors (UV radiation, pollution, etc.), it undergoes significant morphological changes. These include a decrease in the thickness and barrier function of the epidermis, flattening of the dermo-epidermal junction (DEJ), and dermal atrophy with degradation of the extracellular matrix and its structural proteins such as collagen, elastin, and fibrillin. These changes lead to a loss of skin texture and volume, altering the skin's appearance and manifesting as wrinkles, fine lines, and a loss of firmness and elasticity [5-9].
For years, the “anti-aging” approach to dermo-cosmetic skincare has focused on treating the visible signs of aging (wrinkles, fine lines, xerosis, age spots, etc.). However, these visible signs are only a superficial state, the consequence of invisible and deeper changes affecting molecular, cellular and tissue functions. The concept of skin longevity therefore aims to target the underlying causes of aging in order to sustainably maintain skin health and improve skin appearance.
Most of the pathways of aging described at the organismal level must be directly applicable to the skin, although for some, such as the recently added psychological isolation [4], the direct link is more difficult to establish due to a lack of specific studies. However, there is currently no clear consensus neither on the number of hallmarks to consider in the skin nor the hallmarks that should be primarily targeted; some authors consider seven hallmarks [10], while others consider twelve hallmarks [11]. Furthermore, it is also important to consider that the significance of these hallmarks will depend on the targeted age. In young individuals, where there are no visible signs of aging and where morphological skin changes are not yet present, it is preferable to apply a preventive strategy by targeting hallmarks associated with early signs, such as DNA damage, epigenetic modifications, or metabolic alterations. Conversely, in older adults, where signs of aging are clearly visible and morphological changes are well established, it is more appropriate to target later biological mechanisms such as cellular senescence [12]. Finally, it is also important to note that all these markers are interconnected and that targeting one of them can influence one or more others. Under these conditions, it is difficult to precisely identify the most important hallmarks in skin aging, and there is currently no clear consensus on the ideal number of hallmarks to target simultaneously. Although some authors emphasize the importance of multiple targeting [13], to our knowledge, published data on this type of comprehensive approach are limited.
In this context of longevity, we were particularly interested in developing a treatment for mature skin and hypothesized that it is necessary all 3 categories of hallmarks (primary, antagonistic, and integrative) and to target at least 5 hallmarks. Primary markers drive the aging process; antagonistic markers represent a response to primary alterations, while integrative markers ultimately explain the aged phenotype [4].
In skin, numerous cellular dysfunctions accumulate over time that culminate in mature skin. Explaining morphological alterations, mature skin contains many senescent cells, and micro inflammation is well established. This micro inflammation is partly linked to the release of SASP (Senescence-Associated Secretory Phenotype) by senescent cells, but also to environmental stressors [14]. Furthermore, mature skin is also characterized by the presence of many degraded or misfolded proteins and many altered or non-functional mitochondria. These phenomena are linked to an alteration in autophagy and mitophagy mechanisms [15-19]. Cellular metabolism is thus strongly impacted, following these mitochondrial lesions, but also following the decrease in the production of NAD+, an essential cofactor of the electron transport chain [20,21]. To maintain healthy mature skin and toimprove improve skin-aging signs it is important to target the following hallmarks: cellular senescence, micro inflammation, mitochondrial dysfunction, extracellular matrix alterations, and loss of proteostasis.
We therefore designed a specific combination of active ingredients targeting these five essential biological dysregulated pathways associated to aging. To do this, we selected three active ingredients: Proxylane (PX, hydroxypropyltetrahydropyrantriol), Niacinamide (vitamin B3), and a Ginkgo biloba leaf extract (SEN, SILAB).
Proxylane is a xylose-derived sugar (C-xylosid) developed by L'Oréal [18]. Widely used in skincare, particularly for mature skin [22-24], it promotes the synthesis of glycosaminoglycans (GAGs), especially heparan sulfate [25-27], and improves the structure of the dermo-epidermal junction (DEJ) by stimulating the synthesis of proteoglycans and type VII collagen [28,29]. Proxylane is also described as promoting epidermal repair by stimulating keratinocyte proliferation and migration [30] and as promoting the synthesis of proteins and essential components of the extracellular matrix. Proxylane is therefore a prime active ingredient for targeting hallmarks of extracellular matrix (ECM) changes.
Niacinamide is a natural substrate of NAMPT (nicotinamide phosphoribosyltransferase), an enzyme involved in NAD+ synthesis [30,31]. Nicotinamide has also been shown to activate autophagy and mitophagy [32,33] and reduce inflammation [32-34]. Thus, through niacinamide, three of the four important markers of skin aging can be targeted: loss of proteostasis, mitochondrial dysfunction, and micro inflammation.
Ginkgo biloba leaf extract is obtained by enzymatic hydrolysis, which enriches it with cyclitols. Ginkgo biloba is a tree that does not age, which has piqued the curiosity of many scientists [35]. Ginkgo biloba extracts are widely described in the literature for their effects on senescence and inflammation [36], a finding confirmed with our own extract (unpublished data). The use of this extract therefore allows us to target a key hallmark of skin aging: cellular senescence.
The objective of this in vitro study was to demonstrate the potential of targeting multiple aging biomarkers using proxylane, niacinamide, and a Ginkgo biloba leaf extract, in order to target five hallmarks of skin aging and reverse the aged phenotype of reconstructed skin at the molecular, cellular and tissue levels, with the aim of improving skin features of aging.
Materials and Methods
Culture and treatments
Primary cultures of human fibroblasts and keratinocytes were established using healthy skin biopsy samples obtained from aged donors (age: 57 and 42 years, respectively), as previously described [37]. Human skin tissue was collected according to the principles of the Declaration of Helsinki and its use was declared to the French Research Ministry. The donors provided written informed consent according to the 2014 French bioethics law (law number: 94–954 of July 29, 1994).
First, a dermis equivalent was reconstructed by seeding human aged dermal fibroblasts in a scaffold composed of collagen, glycosaminoglycans and chitosan (LabSkin matriX®, LabSkin Creations, Lyon, France). After 10 days of culture under optimized cell culture conditions for ECM neo-synthesis, as previously described [38], the medium was changed and replaced by a culture medium containing or not (control), 0.15% of Ginkgo biloba leaf extract (SEN), or 0.3% Proxylane (PX) or 0.012% Niacinamide (B3) or the combination of all, PX: SEN: B3 (0.3 : 0.15 : 0.012). Dermis equivalents were then cultured for additional 11 days with renewal of medium and treatments every 2-3 days.
At the end of the incubation period, culture medium was changed, keratinocytes from aged donor were seeded on the top of each dermis equivalent and the tissues were cultured for 1 day (without actives). Culture medium was then changed, treatments were applied as described previously and the tissues were cultured in immersion for 7 days with renewal of culture medium and treatment every 2 days. The culture medium was refreshed; the tissues were placed at the air/liquid interface and cultured for 7 days in the absence of treatment. At the end of the incubation period, the culture medium was replaced, the treatments were applied systemically as described previously, and the tissues were cultured for an additional 7 days. Reconstructed tissues with cells from young donors (5 Yo) were cultured in parallel and used as a control, and all conditions were performed in n=3. At the end of the incubation, the tissues were fixed and embedded in paraffin for histological and immunohistological analysis.
Histological analysis and epidermal thickness measurement
To evaluate the global cutaneous structure of samples, Haematoxylin-phloxin-saffron (HPS) staining was performed. Paraffin sections of 5 μm of each condition were cut. After dewaxing and rehydration, the samples were stained with HPS. After rinsing, the sections were dehydrated before the mounting of the slides with a hydrophobic mounting medium. Epidermal thickness was obtained with a Euclidean distance map. Pixels corresponding to the epidermis were selected from other pixels. Images were converted to 8-bit binary image. Images corresponding to the area of interest were converted to a 16-bit distance map. To each epidermis pixel (nonzero) in the distance map binary image a value equal to its distance from the nearest background pixel (zero) was assigned. The epidermis basal line was selected and then applied on the distance map. The mean intensity of the basal line corresponds to the mean distance between the basal line and the stratum corneum. Data are expressed in μm. For all data, the statistical significance was assessed running one-way Student's test, and asterisks indicate statistically significant differences as follows: *P < 0.05, **P < 0.01 and ***P < 0.001.
In situ immunolabeling and images analysis
We analyzed paraffin-embedded sections using immunohistochemistry (Collagen I and P16INK4A) or using immunofluorescence (Collagen VII, Perlecan, Heparan sulfate, Elastin, Fibrillin-1 and LC3B). Following heat-mediated antigen retrieval treatment (incubation for 15 min at 95°C in citrate buffer [Dako, Glostrup, Denmark]), phosphate-buffered saline containing 4% of bovine serum albumin was used to block non-specific binding. Subsequently, the sections were incubated with the primary antibody of interest (anti-collagen I and anti-elastin from Novotec, Bron, France; anti-collagen VII, anti-P16, anti-LC3B from Santa Cruz Biotechnology, Santa Cruz, CA, USA; anti-fibrillin-1, anti-perlecan from Sigma, Saint Louis, USA; anti-Heparan sulfate from Cliniscience, Nanterre, France) diluted in phosphate-buffered saline containing 4% of bovine serum albumin overnight at room temperature. After incubation, for immunohistochemistry analysis, sections were incubated for 1 h with EnVisionTM anti-mouse/rabbit-HRP secondary antibody (EnVision+ System-HRP; Dako) and DAB+ substrate solution was applied to the sections to reveal the color of the antibody staining. Next, slides were counterstained by immersing them in 25% Harris Hematoxylin counterstaining solution. As a negative control, incubation with the primary antibody was replaced by incubation with the corresponding immunoglobulin G class. For immunofluorescence analysis, after incubation, the sections were incubated for 1 h with an AlexaFluor-568-conjugated anti-mouse/rabbit secondary antibody (Molecular Probes, Invitrogen, Carlsbad, CA, USA) and subjected to nuclear counterstaining using 4′,6-diamidino-2-phenylindole. As a negative control, we prepared a condition in which the primary antibody was replaced by the corresponding immunoglobulin G class.
Immunostained specimens were observed using a Zeiss Axio Observer D1 microscope (Zeiss, Oberkochen, Germany). For the markers of interest, positively red stained-tissue areas were automatically detected and segmented from other pixels. The surface area of interest was measured automatically. Data were normalized by the DEJ length and dermal area for dermal-epidermal markers and dermal markers, respectively, and are expressed as percentage of density. For all data, the statistical significance was assessed running one-way Student's test, and statistically significant differences are indicated by asterisks as follows: *P < 0.05, **P < 0.01, ***P < 0.001 and ****P < 0.0001.
Results
Reproduction of an aged-skin phenotype
Significant morphological changes were observed in reconstructed skins with cells from aged donors, compared to those reconstructed with cells from young donors (Figure 1). In aged skins, the epidermis was thinner (-30%, Table 1) and the expression of P16INK4A protein was very strongly increased (x194, Table 1), indicating a high proportion of senescent cells. Concurrently, a significant decrease in the expression of LC3B protein was also observed (-96%, Table 1), indicating an accelerated decrease in the autophagy process. The immunohistology analysis also revealed a decrease in Heparan sulfate in both the epidermis and dermis (-76%, Table 1), as well as a clear decrease of collagen VII and Perlecan in the dermal-epidermal junction area (-70% and -39% respectively, Table 1). A clear decrease of elastic proteins elastin and fibrillin (-54% and -23% respectively, Table 1) was also observed, as well as, to a lesser extent, a decrease in collagen I (-10%, Table 1). All these modifications are coherent with an aged phenotype including epidermal and dermal atrophy, alteration of the dermo epidermal junction DEJ, and alteration of the ECM.
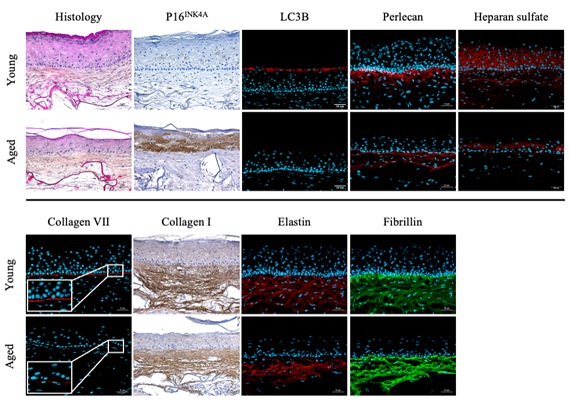 Figure 1: Aging-induced cutaneious structural changes - In situ immunolabeling.
Figure 1: Aging-induced cutaneious structural changes - In situ immunolabeling.
Human full thickness skins were reconstructed using cells extracting from a young donor (5 Yo) or from an aged donor (57 Yo). Histological observations were carried out after Haematoxylin-phloxin-saffron (HPS) staining and the expression of P16INK4A, LC3B, PGC-1a, Collagen VII, Perlecan, Collagen I, Elastin and Fibrillin were evaluated through immunohistology and image analysis.
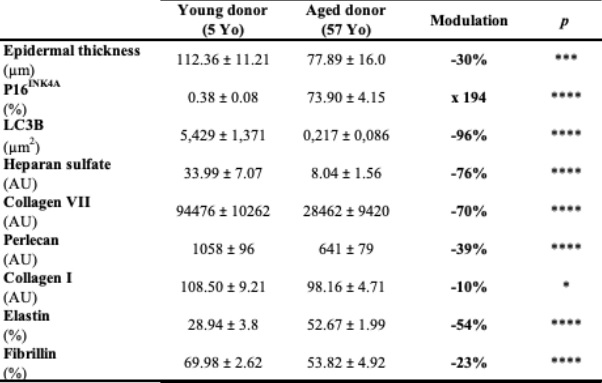 Table 1: Aging-induced cutaneious structural changes - image analysis.
Table 1: Aging-induced cutaneious structural changes - image analysis.
Human full thickness skins were reconstructed using cells coming from a young donor (5 Yo) or from aged donor (57 Yo). Histological observations were carried out after Haematoxylin-phloxin-saffron (HPS) staining and the expression of P16INK4A, LC3B, PGC-1a, Collagen VII, Perlecan, Collagen I, Elastin and Fibrillin was evaluated through immunohistology and images analysis. Statistical significancy: P < 0.05, **P < 0.01, ***P < 0.001 and ****P < 0.0001.
Effects of Gingko biloba leaf extract, proxylane and niacinamide
Ginkgo biloba leaf extract, tested at 0.15%, significantly inhibited P16INK4A expression (-37%, Figure 2B), thus indicating a decrease in senescent cells. No significant effect was observed on the other markers studied, except for a decrease in collagen VII expression. Proxylane, tested at 0.3%, clearly and significantly stimulated epidermal thickness (+29%, Figure 2A). In parallel, a reduction in the proportion of senescent cells, visualized by a decrease in P16INK4A expression, was also observed (-37%, Figure 2B). Additionally, PX also stimulated the expression of heparan sulfate (+98%, Figure 2E) as well as the expression of the DEJ (Dermal-Epidermal Junction) markers collagen VII and perlecan (+59% and +47% respectively, Figure 2D & 2F). Finally, Proxylane also stimulated the expression of proteins constituting structural fibers: collagen I, elastin, and fibrillin (+9%, +35%, and +13% respectively, Figure 2G-2I). Niacinamide, tested at 0.012%, significantly inhibited P16INK4A expression (-33%, Figure 2B) while stimulating the expression of the autophagy marker LC3B (+156%, Figure 2C). On the other hand, a significant improvement in the expression of perlecan, elastin, and fibrillin was observed (+29%, +48%, and +27% respectively, Figure 2D, 2H, 2I) without modulating epidermal thickness or the expression of collagens I and VII (Figure 2).
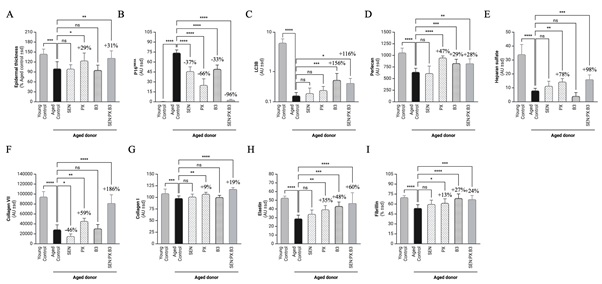 Figure 2: Effect of Ginkgo biloba leaf extract, Proxylane, Niacinamide and their combination on morphological changes in aged-reconstructed skins.
Figure 2: Effect of Ginkgo biloba leaf extract, Proxylane, Niacinamide and their combination on morphological changes in aged-reconstructed skins.
Human full thickness skins were reconstructed using cells coming from aged donor (57 Yo) and treated or not (Control) with 0.15% Ginkgo biloba leaf extract (SEN) or 0.3% Proxylane (PX) or 0.012% Niacinamide (B3) or the combination of all (SEN:PX:B3 0.15:0.3:0.012). Skin reconstructed with cells from young donor (5 Yo) were cultured in parallel and used as control. Histological observations were carried out after Haematoxylin-phloxin-saffron (HPS) staining and the expression of P16INK4A, LC3B, PGC-1a, Heparan sulfate, Collagen VII, Perlecan, Collagen I, Elastin and Fibrillin was evaluated through immunohistology and images analysis. Statistical significancy: P < 0.05, **P < 0.01, ***P < 0.001 and ****P < 0.0001.
Effect of the combination of Gingko biloba leaf extract, Proxylane and Niacinamide
By combining the 3 active ingredients, the overall skin structure was very significantly improved. Epidermal thickness was clearly increased (+31%, Figures 2A, 3), senescent cells, expressing the P16INK4A marker, were almost eliminated (-96%, Figures 2B, 3), highlighting an additive effect between the 3 active ingredients, and the expression of LC3B was increased, indicating a restauration of autophagy process (+116%, Figure 2C, 3).
An additive effect was also found in heparan sulfate and elastin expression (+98% and +60% respectively, Figures 2E, 2H, 3), while for collagens VII and I, a synergistic effect was found, meaning that the effect of the mixture was significantly greater than the effect of each of the active ingredients tested alone (+186% and +19% respectively, Figures 2F, 2G, 3). Finally, an increase in the expression of perlecan and fibrillin was also demonstrated, but in this case, the observed effect was equivalent to that of Proxylane or Niacinamide (+28% and +24% respectively, Figures 2D, 2I, &3).
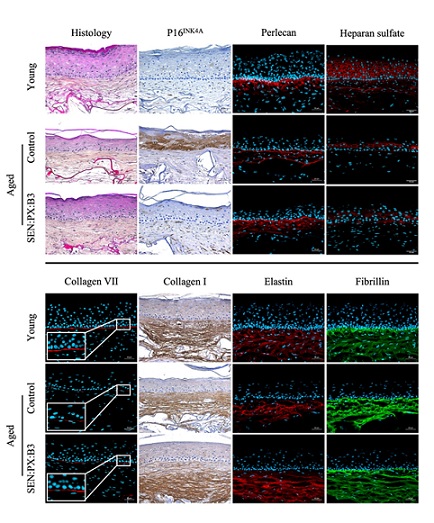 Figure 3: Representative images of aged-reconstructed skins treated or not with Ginkgo biloba leaf extract, Proxylane, Niacinamide and their combination on morphological changes in aged-reconstructed skins - In situ immunolabeling.
Figure 3: Representative images of aged-reconstructed skins treated or not with Ginkgo biloba leaf extract, Proxylane, Niacinamide and their combination on morphological changes in aged-reconstructed skins - In situ immunolabeling.
Discussion
Drawing inspiration from the strategies employed in the field of geroscience and longevity, we applied the concept of longevity specifically to the skin. The aim of this study was to demonstrate, in the context of mature skin, the necessity of targeting multiple hallmarks, particularly cellular senescence, micro-inflammation, mitochondrial dysfunction, extracellular matrix changes and loss of proteostasis, to reset age-related disorders and to restore a stable physiological state, similar to that observed in younger skin. In this way, by acting on the root causes of skin aging, we should be able to restore skin structure, increasing thus its healthspan, and to ultimately improve its appearance. We therefore selected three active ingredients, Proxylane, Niacinamide, and a Ginkgo biloba leaf extract, to address these five hallmarks. We then evaluated them individually and in combination in a reconstructed human skin model designed to reproduce an aged phenotype through the use of cells from older donors.
Firstly, this study allows us to correlate the importance of the hallmarks we targeted with morphological changes in the skin. Indeed, in reconstructed skin models using aged cells, a strong increase in P16INK4A protein expression was observed, thus highlighting a high proportion of senescent cells [36-38], while the expression of LC3B protein was strongly inhibited. The loss of LC3B expression is correlated with a decrease in the autophagy process [39]. Finally, our results also show a significant decrease in the expression of Heparan sulfate, collagen I, elastin, and fibrillin, major proteins of the extracellular matrix that play a role in the formation of structural fibers. The alteration of these hallmarks leads to skin atrophy, which presents a thinner epidermis, an altered dermal-epidermal junction (loss of collagen VII), and dermal atrophy. All these elements thus underscore the relevance of the tissue model used in our study. As our initial objective was to demonstrate benefits of the combination of ingredients on the tissue structure, we did not measure the evolution of inflammatory markers, even though micro-inflammation is a major hallmark.
In our study, we also confirmed the effect of Ginkgo biloba leaf extract on p16 expression, a feature of senescent cells, which is consistent with our internal data (data on file) and the work of Admar et al. [32], describing the benefits of Ginkgo biloba extracts, particularly Gingeklin, for treating inflammation and senescence-related disorders. In our study, although the observed inhibition ofsenescence feature remains clear (37% inhibition), it does not reverse the aged phenotype. Indeed, no other modulation was observed on the other parameters studied. An inhibition of collagen VII expression was even observed, although this inhibition should be interpreted with caution given the low basal expression level of collagen VII in aged skin. These results reinforce our hypothesis that addressing a single hallmark is not enough to effectively act on mature skin where the phenotype is well established and the signs of aging are clearly visible,. These senescent cells secrete numerous inflammatory factors and proteases (SASPs) that maintain micro-inflammation and contribute to the degradation of major structural proteins in the extracellular matrix [33,34].
Similarly, treatment with niacinamide alone did not properly reverse the aged skin phenotype, even though a clear inhibition of the proportion of senescent cells was observed and the expression of LC3B, perlecan, elastin, and fibrillin was increased. Niacinamide was selected because it is described as having beneficial effects on micro-inflammation, loss of proteostasis, and mitochondrial dysfunction [28-30,33]. In our study, we did not measure the level of inflammation, nor were we able to directly measure the effects on energy metabolism. Nevertheless, by measuring the expression of LC3B, we can highlight effects related to loss of proteostasis. Although we measured only an effect on two markers, the observed results remain consistent with those described in the literature. Regardless, these results demonstrate once again that niacinamide alone does not reverse the aged skin phenotype.
In our study, Proxylane was the molecule that, when tested alone, showed the best efficacy by inhibiting senescence and restoring the expression of heparan sulfate, collagen VII, perlecan, elastin, fibrillin, and collagen I, and by increasing epidermal thickness. While the effects on epidermal thickness as well as the effects on the expression of proteins of located at the dermal-epidermal junction or in the dermal extracellular matrix were confirmed [22-25], those on senescence were unexpected based on its published mode of action. Currently, we do not know whether this decrease in P16INK4A expression observed with Proxylane is due to a direct or indirect effect. These results challenge our theory that it is necessary to target, at a minimum, the following five hallmarks: cellular senescence, microinflammation, mitochondrial dysfunction, extracellular matrix alterations, and loss of proteostasis. Indeed, in this study, Proxylane appears to have no effect on loss of proteostasis or mitochondrial dysfunction, while a clear reversal of the phenotype was observed. However, it is important to remind that we initially selected Proxylane for its effects on the extracellular matrix, but our internal data (data on file) also suggested an effect on stem cell exhaustion, to which we can now add an effect on senescent cells. Thus, several hallmarks are indeed targeted by Proxylane, even if they were not initially intended. Furthermore, considering the interconnectedness of hallmarks, we can assume that the effect on senescent cells contributes to limiting inflammation [34].
Furthermore, even though Proxylane showed significant effects, it was only by combining the three active ingredients that we obtained the best efficacy, mainly in terms of senescence, collagen VII, collagen I, and elastin. Indeed, senescent cells expressing the P16INK4A marker had almost disappeared after treatment with the trio, and the expression of collagen VII and collagen I was synergistically increased with the trio. Collagen VII is considered an important biomarker in skin healthspan [35].
The results of this study clearly demonstrate the benefits of our combination in limiting age-related skin changes by targeting five hallmarks of aging and thus confirm its value in a longevity strategy aimed at improving aging signs of mature skin [36,39].
Unlike conventional anti-aging treatments, the objective of this approach is to address the underlying causes of aging in order to help the skin regain its structure and thus sustainably modulate its appearance. Our study clearly demonstrates the benefits of combining proxylane, niacinamide, and Ginkgo biloba leaf extract to improve skin-aging features, by strengthening the skin's structure. It should be noted, however, that our study has limitations, the limited number of biomarkers in each of the targeted hallmarks as it is an in vitro study; clinical evidence in mature skin will be necessary to confirm this promising results.v
Conclusion
By targeting several root causes of aging this new combination of Proxylane, Niacinamide, and Ginkgo biloba leaf extract, reverses the phenotype of aged skin in an in vitro 3D modelto, thus demonstrating its high potential to improve healthspan of mature skin.
References
- McCay CM, Crowell MF, Maynard LA (1989) The effect of retarded growth upon the length of life span and upon the ultimate body size. Nutrition 5: 155-1571.
- Moqri M, Herzog C, Poganik JR, Justice J, Daniel B, et al. (2023) Biomarkers of aging for the identification and evaluation of longevity interventions. Cell 186: 3758-3775.
- López-Otín C, Blasco MA, Partridge L, Serrano M, Kroemer G, et al. (2023) Hallmarks of aging: An expanding universe. Cell 186: 243-278.
- Kroemer G, Maier AB, Cuervo AM, Gladyshev VN, Ferrucci L, et al. (2025) From geroscience to precision geromedicine: Understanding and managing aging. Cell 188: 2043-2062.
- Wang Z, Man MQ, Li T, Elias PM, Mauro TM, et al. (2020) Aging-associated alterations in epidermal function and their clinical significance. Aging (Albany NY). 2: 5551-5565.
- Farage MA, Miller KW, Maibach HI (2017) Degenerative Changes in Aging Skin. In: Farage M, Miller K, Maibach H (eds.). Textbook of Aging Skin. Springer, Berlin, Heidelberg.
- Huang S, Strange A, Maeva A, Siddiqui S, Bastien P, et al. (2023) Quantitative nanohistology of aging dermal collagen. Front Aging 4:1178566.
- Jevtic M, Löwa A, Novácková A, Kovácik A, Kaessmeyer S, et al. (2020) Impact of intercellular crosstalk between epidermal keratinocytes and dermal fibroblasts on skin homeostasis. Biochim Biophys Acta Mol Cell Res 1867: 118722.
- Coleman SR, Grover R (2006) The anatomy of the aging face: volume loss and changes in 3-dimensional topography. Aesthet Surg J 26: 4-9.
- Chin T, Lee XE, Ng PY, Lee Y, Dreesen O, et al. (2023) The role of cellular senescence in skin aging and age-related skin pathologies. Front Physiol 14: 1297637.
- Dréno B, Benoit I, Perrier E, Radman M, et al. (2024) Beyond the genome: protecting the proteome may be the key to preventing skin aging. Eur J Dermatol 34: 355-360.
- Jin S, Li K, Zong X, Eun S, Morimoto N, et al. (2023) Hallmarks of Skin Aging: Update. Aging Dis 14: 2167-2176.
- Eckhart L, Tschachler E, Gruber F (2019) Autophagic Control of Skin Aging. Front Cell Dev Biol. 2019; 7: 143.
- Sreedhar A, Aguilera-Aguirre L, Singh KK (2020) Mitochondria in skin health, aging, and disease. Cell Death Dis 11: 444.
- Zhang C, Gao X, Li M, Yu X, Huang F, et al. (2023) The role of mitochondrial quality surveillance in skin aging: Focus on mitochondrial dynamics, biogenesis and mitophagy. Ageing Res Rev 87: 101917.
- Chini CCS, Tarragó MG, Chini EN (2017) NAD and the aging process: Role in life, death and everything in between. Mol Cell Endocrinol 455: 62-74.
- McReynolds MR, Chellappa K, Baur JA (2020) Age-related NAD+ Exp Gerontol 134: 110888.
- Cavezza A, Boulle C, Guéguiniat A, Pichaud P, Trouille S, et al. (2009) Synthesis of Pro-Xylane: a new biologically active C-glycoside in aqueous media. Bioorg Med Chem Lett 19: 845-849.
- Bouloc A, Roo E, Moga A, Chadoutaud B, Zouboulis CC, et al. (2017) A Compensating Skin Care Complex Containing Pro-xylane in Menopausal Women: Results from a Multicentre, Evaluator-blinded, Randomized Study. Acta Derm Venereol 97: 541-542.
- Wang ST, Neo BH, Betts RJ (2021) Glycosaminoglycans: Sweet as Sugar Targets for Topical Skin Anti-Aging. Clin Cosmet Investig Dermatol 14: 1227-1246.
- Vassal-Stermann E, Duranton A, Black AF, Azadiguian G, Demaude J, et al. (2012) A New C-Xyloside induces modifications of GAG expression, structure and functional properties. PLoS One 7: 47933.
- Pineau N, Carrino DA, Caplan AI et al. Biological evaluation of a new C-xylopyranoside derivative (C-Xyloside) and its role in glycosaminoglycan biosynthesis. Eur J Dermatol 21: 359-370.
- Pineau N, Bernerd F, Cavezza A, Dalko-Csiba M, Breton L, et al. (2008) A new C-xylopyranoside derivative induces skin expression of glycosaminoglycans and heparan sulphate proteoglycans. Eur J Dermatol 18: 36-40.
- Sok J, Pineau N, Dalko-Csiba M, Breton L, Bernerd F, et al. (2008) Improvement of the dermal epidermal junction in human reconstructed skin by a new c-xylopyranoside derivative. Eur J Dermatol 18: 297-302.
- Deloche C, Minondo AM, Bernard BA, Bernerd F, Salas F, et al. (2011) Effect of C-xyloside on morphogenesis of the dermal epidermal junction in aged female skin. An ultrastuctural pilot study. Eur J Dermatol 21: 191-196.
- Muto J, Naidu NN, Yamasaki K, Pineau N, Breton L, et al. (2011) Exogenous addition of a C-xylopyranoside derivative stimulates keratinocyte dermatan sulfate synthesis and promotes migration. PLoS One 6: 25480.
- Tan CL, Chin T, Tan CYR, Rovito HA, Quek LS, et al. (2019) Nicotinamide Metabolism Modulates the Proliferation/Differentiation Balance and Senescence of Human Primary Keratinocytes. J Invest Dermatol 139: 1638-1647.
- Bierman JC, Laughlin T, Tamura M et al. Niacinamide mitigates SASP-related inflammation induced by environmental stressors in human epidermal keratinocytes and skin. Int J Cosmet Sci 42 :501-511.
- Boo YC (2021) Mechanistic Basis and Clinical Evidence for the Applications of Nicotinamide (Niacinamide) to Control Skin Aging and Pigmentation. Antioxidants (Basel) 10: 1315.
- Marques C, Hadjab F, Porcello A, Lourenço K, Scaletta C, et al. (2024) Mechanistic Insights into the Multiple Functions of Niacinamide: Therapeutic Implications and Cosmeceutical Applications in Functional Skincare Products. Antioxidants (Basel) 13: 425.
- Wang L, Cui J, Jin B, Zhao J, Xu H, et al. (2020) Multifeature analyses of vascular cambial cells reveal longevity mechanisms in old Ginkgo biloba Proc Natl Acad Sci USA 117: 2201-2210.
- Adnan M, Rasul A, Hussain G, Hussain G, Shah MA, et al. (2020) Ginkgetin: A natural biflavone with versatile pharmacological activities. Food Chem Toxicol 145: 111642.
- Marques C, Hadjab F, Porcello A, Lourenço K, Scaletta C, et al. (2024) Mechanistic Insights into the Multiple Functions of Niacinamide: Therapeutic Implications and Cosmeceutical Applications in Functional Skincare Products. Antioxidants (Basel) 13: 425.
- Low E, Alimohammadiha G, Smith LA, Costello LF, Przyborski SA, et al. (2021) How good is the evidence that cellular senescence causes skin ageing? Ageing Res Rev 71: 101456.
- Kim J, Kim H, Seo WY, Lee E, Cho D, et al. (2024) Collagen Type VII (COL7A1) as a Longevity Mediator in Caenorhabditis elegans: Anti-Aging Effects on Healthspan Extension and Skin Collagen Synthesis. Biomol Ther (Seoul) 32: 801-811.
- Safwan-Zaiter H, Wagner N, Wagner KD (2022) P16INK4A-More Than a Senescence Marker. Life (Basel) 12: 1332.
- Germain L, Rouabhia M, Guignard R, Carrier L, Bouvard V, et al. (1993) Improvement of human keratinocyte isolation and culture using thermolysin. Burns 19: 99-104.
- Dos Santos M, Metral E, Boher A, Rousselle P, Thepot A, et al. (2025) In vitro 3-D model based on extending time of culture for studying chronological epidermis aging. Matrix Biol 47: 85-97.
- Murase D, Kusaka-Kikushima A, Hachiya A, Fullenkamp R, Stepp A, et al. (2020) Autophagy Declines with Premature Skin Aging resulting in Dynamic Alterations in Skin Pigmentation and Epidermal Differentiation. Int J Mol Sci 21: 5708.
Citation: Juchaux F, Menthière SDe, Simonnet J-T, Aguilar L, Kovylkina N, et al. (2025) Targeting Multiple Hallmarks of Aging with Proxylane, Ginkgo biloba leaf extract and Niacinamide Improves Healthspan by Reversing the Aged Skin Phenotype. J Clin Dermatol Ther 12: 0161.
Copyright: © 2026 Franck Juchaux, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

