
The Use of Intravenous Tenecteplase in Acute Ischemic Stroke after Idarucizumab Reversal of Dabigatran Anticoagulant Effects; Experience from a Regional Hospital Directed by Video Telemedicine Stroke Specialist
*Corresponding Author(s):
Tunde Maiyaki IbrahimDepartment Of Medicine, Goulburn Valley Health, Graham Street, Shepparton, Vic 3630, Australia
Email:imaiyaki@yahoo.com
Abstract
Novel Oral Anticoagulants (NOACs) are widely used in patients with non-valvular atrial fibrillation to prevent Acute Ischemic Stroke (AIS) and other thrombo- embolic phenomena. The management of AIS with Intravenous Thrombolysis Therapy (IVT), also known as fibrinolytic therapy in people who are taking NOACs poses a great challenge due to increased risks of hemorrhage and intracranial bleeding. We present the case of a 77-year-old man on dabigatran for atrial fibrillation who woke up with an extensive right middle cerebral artery ischemic stroke and had his anticoagulant effects reversed with idarucizumab before successful thrombolysis with intravenous tenectaplase.
Introduction
The Novel Oral Anticoagulants (NOACs)- the factor Xa (rivaroxaban and apixaban) and direct thrombin (dabigatran) inhibitors- are widely used to prevent embolic events, especially Acute Ischemic Stroke (AIS), in patients with non-valvular Atrial Fibrillation (AF) [1]. Despite NOACs use in AF and venous-thromboembolic phenomenon about 1-2 % of individuals with atrial fibrillation and 0·1-0·2% of those with venous thromboembolism who are receiving one of the NOACs (dabigatran, rivaroxaban, or apixaban) are likely to develop AIS yearly. Additionally, 0·2-0·5% of individuals with atrial fibrillation who are receiving one of the NOACs can be expected to experience an intracranial hemorrhage [2]. An important part of the management of AIS involves the intravenous administration of a recombinant tissue Plasminogen Activator (rtPA) to achieve reperfusion1.To improve outcomes in patients who experienced a potential disabling AIS (National Institutes of Health Stroke Scale (NIHSS) score >4), the American Heart Association, American Stroke Association, European Stroke Organization, and Stroke Foundation Australia recommend the use of IV rtPA within 4.5 to 9 hours of symptom onset. This reperfusion method, however, is challenging in AIS patients who are already taking a NOAC because of increased risks of NOACs-associated bleeding especially spontaneous intracranial hemorrhage (SICH) [1-4]. Despite this risk the treatment options are very practicable even in regional health facilities linked to the video assisted telehealth center through which stroke specialist can remotely direct patient care. Therefore, IV rtPA is still most practicable treatment option even in situation of prior NOAC use. Although some stroke experts advise for it use only if last dose of NOAC was 48hrs or more before the stroke or the specific coagulation parameter are normal which can be achieved by reversal of anticoagulation [1,4,5]. This case report is to show clinicians the real-world successful use of IV rtPA to treat stroke patient after reversal of NOAC in a regional hospital using telemedicine.
Case Presentation
A 77-year-old male farmer was admitted with an extensive right middle cerebral artery ischemic “wake up stroke” in the context of non-valvular AF on dabigatran (110mg BD) about 13 hours after the patient was last known to be well. On the background of significant cardiovascular risk factors of hypertension, type 2 diabetes mellitus and dyslipidemia giving CHADSVASC score of 3 prior to his stroke. He was also on metoprolol 25mg BD, metformin 1g BD and fenofibrate 145mg daily respectively.
The main neurological features were right-sided headache and left-sided hemiparesis, hemisensory loss, hemi spatial neglect, hemianopia, and motor aphasia with NIHSS score of 10. He was hemodynamically stable with BP=150/70mmHg and in AF with average heart rate of about 90/min. The patient had the last dose of dabigatran 16 hours before presentation. The initial CT stroke imaging protocol (Figures 1&2) revealed acute right frontal and temporal lobe infarcts with ischemic penumbra extending to involve the right temporoparietal lobe and thrombus in the M2 segment of the right Middle Cerebral Artery (MCA).Base line blood glucose was 5.9mmol/L, international normalized ratio (INR) of 1.2, activated Partial Thromboplastin Time (aPTT) of 31 seconds, prothrombin time (PT) of 16.7 seconds, platelets count of 247/nl, HbA1c of 6.2% (44mmol/mmol Hb),and hemoglobin of 139g/L. The patient had grade 4 chronic kidney disease with serum creatinine of 151 μmol/l and eGFR 38 ml/min. The lipid profile was essentially normal and ECG showed rate controlled atrial fibrillation.
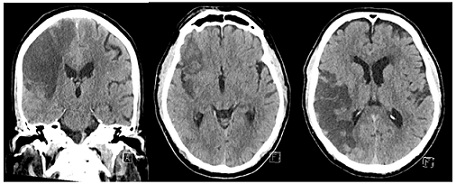 Figure 1: Initial CT showing acute right frontal and temporal lobe infarction and right temporoparietal lobe ischemia.
Figure 1: Initial CT showing acute right frontal and temporal lobe infarction and right temporoparietal lobe ischemia.
 Figure 2: Initial perfusion CT scan showing reduced cerebral blood flow, increased cerebral blood volume, and prolong mean transient time in right frontal and temporal lobe, right temporoparietal lobe, and attenuation of the M2 segment of the right middle cerebral artery (MCA).
Figure 2: Initial perfusion CT scan showing reduced cerebral blood flow, increased cerebral blood volume, and prolong mean transient time in right frontal and temporal lobe, right temporoparietal lobe, and attenuation of the M2 segment of the right middle cerebral artery (MCA).
As per-protocol the case was discussed with the Victorian Stroke Telemedicine (VST) neurologist and patient was subsequently thrombolyzed with IV tenecteplase (0.25mg/kg body weight) after reversal of dabigatran with idarucizumab 5mg single dose. He was admitted on the intensive care unit and acute early stroke rehabilitation was initiated.
The serial NIHSS scores at two- and twenty-four-hours post-thrombolysis were 10 and 17, respectively suggesting worsening neurological status. A non-contrast CT brain 24-hours post IVT showed extensive right middle cerebral artery territory established infarct and interval cerebral edema but with no evidence of Hemorrhagic Transformation (HT). The NIHSS score and clinical assessments remained unchanged on day 5 post-IVT but repeat CT (Figure 3) showed established extensive infarct with a small HT in the right frontal lobe. Although the recovery was slow our patient improved remarkably (ambulating with a four-wheel frame) over a 4-month period, with a NIHSS score of 5 at 4 months.
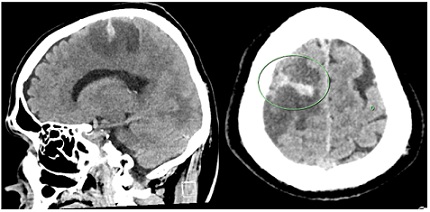 Figure 3: CT scan on day 5 post IVT showing established extensive Rt MCA infarct and small hemorrhagic transformation in the right frontal lobe.
Figure 3: CT scan on day 5 post IVT showing established extensive Rt MCA infarct and small hemorrhagic transformation in the right frontal lobe.
Discussion
We described a successful IVT with tenecteplase after reversal of dabigatran with idarucizumab in a patient with permanent non valvular AF on dabigatran who presented with wakeup AIS. Although our patient's parameters (NIHSS score, and radiologically) showed initial deterioration he had NIHSS score reduction of five with neurological and functional improvement over four- months after thrombolysis. The outcomes (small HT and reduction in NIHSS score) in our patients are consistent with that reported in earlier studies on this subject of using IVT after dabigatran reversal with idarucizumab [6,7]. Kermer, et al., in a meta-analysis of 80 AIS patients treated with IVT after dabigatran reversal with idarucizumab showed clinical improvement in stroke severity and quality of life in 78% of the patients [6]. Frol, et al, in their comprehensive review of 251 AIS patients reported HT (8%), SICH (8%) and a mean reduction of six from the initial NIHSS score after IVT use post stroke after dabigatran reversal with Idarucizumab [7]. This rate of HT and SICH was reported to be like those who had no prior anticoagulant. HT is a spontaneous complication in some patients with AIS even without the use of thrombolytic agents such as idarucizumab. This risk is increased by hypertension, hyperglycaemia, stroke severity, and fibrinolytic therapy [8]. Our patients had all these factors therefore the small haemorrhagic transformation is not surprising as this could have occurred without the use of thrombolysis. The small volume of the HT in our patients despite the large size of the infarct might be due to good blood pressure (150/90) and blood sugar level control (7mmol/l). The mechanism of HT in AIS is not very clear but postulated mechanism include increase in vascular permeability caused by an alteration in the blood-brain barrier which occur because of increased thrombin production in AIS [8-10]. Additionally, the anti coagulation associated with NOAC use in AIS patients increases the extravasation of blood into the infarcted tissue, leading to hemorrhagic transformation [8]. With the successes reported for IVT with rtPA in stroke patients post dabigatran reversal with Idarucizumab studies are needed to evaluate the use of rtPA in stroke patients on anti-activated factor X agents (apixaban,rivaroxaban and edoxaban) and reversal with Andexanet alfa which is recently approved by FDA as a reversal agent for apixaban and rivaroxaban [11]. Our patient’s good outcome was not only credited to the IVT but also the early initiation of comprehensive stroke management and rehabilitation. Finally this study shows that stroke patients can be successfully thrombolyzed in regional and possibly rural hospitals with adequate facilities such as neuroimaging scans and access to high speed internet connection to facilitate telemedicine guidance and direction from a stroke specialist located in other institution.
Conclusion
This case report demonstrates a safe and effective IVT use after reversal of dabigatran effect using idarucizimab in a regional health facility. This treatment and comprehensive management lead to improved stroke outcomes.
References
- The European Stroke Organisation (2021) NOAC reversal before systemic thrombolysis in ischaemic stroke: 2021 ESO guidelines update. Basal, Switzerland.
- Hankey G J, Norrving B, Hacke W, Steiner T (2014) Management of acute stroke in patients taking novel oral anticoagulants. Int J Stroke 5: 627-632.
- MIMS Neurology (2021) NOAC reversal before systemic thrombolysis in ischaemic stroke: 2021 ESO guidelines update.
- Australian Clinical Guidelines for Stroke Management - Chapter 3 & 4 of 8: Acute medical and surgical management, and secondary prevention - Stroke Foundation Australia (2019).
- Powers WJ, Rabinstein AA, Ackerson T, Adeoye OM, Bambakidis NC, Becker K, et al. (2019) Guidelines for the early management of patients with acute ischemic stroke: 2019 update to the 2018 guidelines for the early management of acute ischemic stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 50: 344-418.
- Kermer P, Eschenfelder CC, Diener HC, Grond M, Abdalla Y, Abraham A, et al. (2020) Antagonizing dabigatran by idarucizumab in cases of ischemic stroke or intracranial hemorrhage in Germany-Updated series of 120 cases. Int J Stroke 15: 609-618.
- Frol S, Sagris D, Pretnar OJ, Šabovic M, Ntaios G (2021) Intravenous thrombolysis after dabigatran reversal by idarucizumab: A systematic review of the literature. Front Neurol 12: 666086.
- Spronk E, Sykes G, Falcione S, Munsterman D, Joy T, et al. (2021) Hemorrhagic Transformation in Ischemic Stroke and the Role of Inflammation. Frontiers in Neurology 12: 1-15.
- Seiffge DJ, Meinel T, Purrucker JC, Kaesmacher J, Fischer U, et al. (2021) Recanalisation therapies for acute ischaemic stroke in patients on direct oral anticoagulants.J Neurol Neurosurg Psychiatry 92: 534-541.
- Chen B,Cheng Q, Yang K, Lyden PD (2010) Thrombin mediates severe neurovascular injury during ischemia. Stroke. 41: 2348-2352.
- Siegal DM, Curnutte JT, Connolly SJ, Lu G, Conley PB, et al. (2015) Andexanet Alfa for the Reversal of Factor Xa Inhibitor Activity. N Engl J Med 373: 2413-2424.
Citation: Ogeleka P, Ibrahim TM (2022) The Use of Intravenous Tenecteplase in Acute Ischemic Stroke after Idarucizumab Reversal of Dabigatran Anticoagulant Effects; Experience from a Regional Hospital Directed by Video Telemedicine Stroke Specialist. J Clin Stud Med Case Rep 9: 0140.
Copyright: © 2022 Pascal Ogeleka, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

