
Usefulness of Medi Test Combi 9 in the Diagnosis of Hemoglobinuria in Limited Recourse Setting
*Corresponding Author(s):
Joseph Mabiala BodiDivision Of Emergency And Intensive Care, Department Of Pediatrics, University Hospitals Of Kinshasa, School Of Medicine, University Of Kinshasa, Congo, The Democratic Republic Of The
Email:josephbodi9@gmail.com
Abstract
Background
Detection of hemoglobin in fresh urine remains a challenge in resource limited setting since quantitative measurements of hemoglobinuria is not always technically feasible. This study aimed to evaluate the performance of 3 types of dipstick for the diagnosis of hemoglobinuria in children suffering Black water fever.
Methods
In a case-control study, 129 patients (43 inpatients with dark urine versus 86 outpatients with clear urine) were screened for hemoglobinuria. A spot urine sample from each subject was tested both with 3 dipsticks (Medi Test Combi 9, as well as 9 and 10 parameters Cypress) and a spectrophotometer using 3, 3′ dimethylbenzidine reagent. The performance of dipstick was compared and correlated to that of quantitative measurement for the haemoglobinuria values higher or equal to (1+).
Results
With the dipstick, the prevalence of hemoglobinuria in the 43 hemoglobinuria cases was 27% for (1+), 18% for (2+) and 53% for (3+). The corresponding hemoglobinuria values, based on the spectrophotometric quantification, were 0.061±0.0166mg/L, 0.3986±0.2612mg/dL and 0.5679±0.27688mg/L, respectively. The sensibility of Dipsticks was 100% for Medi test combi9 and 93% for both 9 and 10 parameters cypress diagnosis Dipsticks. Specificity and positive predictor value was 100%, for each of the dipsticks. The negative predictor value was 100% for Medi test combi9 and 96.6 % for both parameter 9 and 10 of cypress diagnosis.
Conclusion
Haemoglobinuria may truthfully be detected by each of the qualitative methods tested here. Each of these technics represents a reliable alternative method to detect hemoglobinuria in limited resource countries. The sensibility, specificity, positive and negative predictors value are maximal with Medi test combi9.
Keywords
Black water-patients; Diagnosis value; Dipstick; Hemoglobinuria; Spectrophotometer quantification
INTRODUCTION
Hemoglobinuria is a common observation in many diseases with acute intravascular hemolysis [1-5]. Back Water Fever (BWF) is a life-threatening form of severe malaria and the leading cause of hemoglobinuria in Sub-Saharan Africa. Besides, other diseases such as leptospirosis, hanta virus infections, septicemia with Clostridium perfringens, hemolytic uremic syndrome, enzymopathies such as G6PD-deficiency, and hemoglobinopathies can cause acute massive intravascular hemolysis, leading to hemoglobinuria [6-10]. Since massive hemoglobinuria is a real threat for renal function, early detection is crucial for quick management.
In a recent past, the assessment of hemoglobin in urine was performed using direct spectrophotometry [11], derivate photometry [12,13], chemoluminescence [14] and colorimetry methods [15-17]. Most technics performing quantitative detection require electricity and heavy laboratory infrastructure that are often unavailable or too expensive for laboratories in developing countries. Therefore, the detection of hemoglobin from fresh urine is a real challenge for the vast majority of laboratories in resources limited countries. An alternative could be the translation of simple quantitative technics applied for serum hemoglobin to the detection of hemoglobinuria. Unfortunately, these technics tend to be less reliable because of interferences of other components in the urine [17-19]. To date, qualitative detection using dipsticks remains the most commonly used method in limited resource setting. However, this approach has long been considered to be a highly subjective. Until recently, the ability of the technician to discriminate colors was a key component of the reliability of results. In addition, the threshold of detection in dipsticks varies depending on the authors and the protocols that is used [20,21]. Luckily, these limitations are now circumvented by the use of automatic tools that allow the lecture of the dipstick results using reflectometer [21]. For most of dipsticks, hemoglobin can be detected from the concentration of 0.05g/L and this threshold is significantly higher compared to serum hemoglobin detection (from 0.001g/L) [22,23].
Dipsticks in general can be considered as one of the most important advances in the current diagnosis in pediatric nephrology. In resources limited countries, dipsticks are the best way to approach the most frequent urinary diseases because of their minimal technical requirements and easy feasibility. Dipsticks have been used in the diagnosis in many diseases with urinary expression such as urinary infection, hematuria, proteinuria, myoglobinuria and the measurement hemoglobin components (e.g. urobilin, bilirubin), and pH [23-25]. Dipstick has also been previously used for the diagnosis of hemoglobinuria in children with dark urine [2-4]. Many types of dipsticks are used to directly diagnose urinary tract diseases at the bedside instead of delayed measurements on the bench. To date, no study has evaluated the performance of dipstick brands commonly used in the Democratic Republic of Congo (DRC). The present study aimed to evaluate 3 types of dipsticks commonly used in pediatric nephrology at University Hospitals of Kinshasa and assess their diagnostic value for hemoglobinuria in children presenting Blackwater fever, in comparison to the quantitative measure of hemoglobin in urine using the reagent 3,3′ dimethylbenzidine, considered as gold standard [26].
METHODS
Study design, participants and sites
All informations about this study were fully provided to parents in local languages. Written informed consent for participation in the study was obtained from their parents or legal representatives. A case-control study was conducted in 4 hospitals of Kinshasa City, the capital town of Democratic Republic of Congo (DRC), from January 2010 to December 2011 in 43 in- patients with black water fever (Cases) and 86 outpatients with uncomplicated malaria (Controls).
Case definition
Black Water Fever (BWF) was defined as the presence of haemoglobin in the dark urine, jaundice, anaemia following acute intravascular haemolysis (macroscopic haemoglobin), in patients with fever and Plasmodium falciparum malaria.
Uncomplicated Malaria (UM) when patients had fever or history of fever in previous 48 hours, with the presence of plasmodium falciparum in blood thick and film without severity signs. Two controls matched for age, sex and location area were selected for each case.
Hemoglobinuria was detected by the observance of color change on dipstick chromogen from yellow to blue. For the quantitative method, it was defined as the detection of hemoglobin in dark urine using the 3,3′dimethylbenzidine reagent and the spectrophotometer for lecture.
Laboratory measurements
Twenty mL of fresh urine were collected from each participant. Both urine dipstick test and urine spectrophotometer quantification of hemoglobin measurement were performed for each sample. The presence of haemoglobin in urine was first assessed using 3 types of dipsticks: Medi Test Combi9 (Machery Eur, Paris, France), Parameter 9 ( Cypress Diagnostics, 3201 Langdorp-Belgium) and Parameter 10 (Cypress Diagnostics, 3201 Langdorp-Belgium ), then quantified by spectrophometer (Thermo Genesis 10 BIO, New York, USA) using the 3, 3′-dimethylbenzidine reagent following the manufacturer’s procedure (26). The results of urine dipstick were read as either negative (yellow color) or positive (change in blue color) 1+, 2+, 3+, corresponding approximately to hemoglobin concentrations of respectively 0.061±0.0166mg/L, 0.3986±0.2612mg/dL and 0.5679±0.27688mg/L performed using quantitative measurement of hemoglobinuria with the 3,3′-dimethylbenzidine reagent (26). The diagnostic value (sensitivity, specificity, positive predictor value, negative predictor value) of dipstick ≥1+ and ≥2+ to detect hemoglobinuria was thus assessed and compared to quantitative method.
Urine analysis using dipstick
Procedure: Urine collection procedure and urine analysis were performed following the standard procedure for each dipstick type: (i) collect 5-10ml of fresh urine in clean recipient of 20ml; (ii) immerge the dipstick vertically in the recipient during one second; (iii) withdraw the dipstick; (iv) read the dipstick from yellow to different blue colors and the corresponding number of +; (v) close the test as positive if the color changes from yellow to blue, expressed by 1+, 2+ and 3+.
Spectrophotometric quantification of hemoglobin in urine using the 3,3′dimethylbenzidine reagent Reagents: Glacial acetic acid MM=60.05, reference 310 K19356463, Merck; pure potassium cyanide at 99%, MM=65.12, reference P/4600/50, Fisher Chemicals; ethanol at 95°; pure potassium ferricyanide at 99%, MM=329.26 (cat.num P/4880/50, Fisher Chemicals); pure sodium hydrogen carbonate pro-analysis 99%, MM=84.01 (cat.num S/4200/60, Fisher Chemicals); 3,3’-dimehylbenzidine (o-toluidine) MM=212.29 (Merck); hydrogen peroxide at 3%, MM=33.01 (Lot : 000042, Cooper)
Working solutions preparation: R1 solution is the mixture of 1 volume of glacial acetic acid and 4 volumes of ethanol 95%. R2 solution, is the stock solution of 3,3’-dimehylbenzidine at 0.2g/l, obtained by dissolving 2g of the R2 is dissolved into 100ml of R1 and kept into a brown recipient. R3 solution is concentrated of 0.04g/l, freshly prepared by diluting 1/5 of the reagent R2. R4 is the solution of hydrogen peroxide at 2%, obtained by diluting 2/3 of the solution concentrated at 3% with distilled water. Hemoglobin solution is a centrifugation supernatant obtained after lysis of 2ml of packed Red Blood Cells (RBC) suspended in 3 ml physiological saline. This pellet is from a sample of 5ml of blood collected in a tube with EDTA, after decanting the plasma and washes the RBC. Determination of hemoglobin solution concentration was done using the Drabkin method [27]. The optical density read was transferred to a calibration curve obtained previously to determine the hemoglobin concentration of the solution. Calibration curve realization: made from a hemoglobin solution of 1g/L determined by the Drabkin method and diluted ½, ¼, 1/8 (Figure 1). The chromogen was formed with 10ml of each dilution of the hemoglobin solution, mixed with a solution consisting 1000µl of R3 and 1000µl of R4. The chromogenic absorbance was determined with the spectrophotometer (Thermo Genesis 10 BIO, NEW YORK, USA) at 630nm wavelength against a blank reagent formed by a mixture V/V of R3 and R4 reagents. Absorbance was obtained for different concentrations of the hemoglobin solution.
Hemoglobin measurement in urine using the reagent 3, 3’-dimehylbenzidine: 20ml fresh urine were collected from patients in a sterile recipient, transferred and analyzed in the laboratory. Urine samples with nitrite test positive using dipsticks were excluded.
Measure of hemoglobin in urine: 1000µl R3 and 1000µl of R4 were mixed in a test tube. After 5 minutes, 10µl of urine sample were added in the tube. After incubation at room temperature for 10 minutes, lecture was performed using optic density of coloration obtained with the spectrophotometer at 630nm against a blank reagent formed by a mixture V/V of R3 and R4 reagents. Two measures were done for each sample and the mean was calculated. The optic density was reported to the standard curve to get the concentration of hemoglobin in urine for each sample.
Data management and analysis
Data were recorded using Epi-Info 7 version 2002 and analysed with SPSS, version 18. All records were crosschecked with the original data sheets before analysis. Qualitative hemoglobinuria expressed as 1+, 2+, 3+ was subdivided into three sub-groups: corresponding to quantitative hemoglobin sub-group measured by spectrophometer. The diagnostic value (sensitivity, specificity, positive Predictor value, negative predictor value) of the dipstick 1+ or greater was thus studied. Chi-squared or Fisher exact tests were used to evaluate associations between the number of (+) s on the dipsticks and the concentration on quantitative measurement of haemoglobin in urine. Confidence intervals were built. Two tailed p=0.05 was adopted as the significant threshold.
Ethics statement
The Ethics Committee of Public Health School of University of Kinshasa approved the protocol under the number ESP/CE/027B/2011.
RESULTS
A total of 129 Congolese children were recruited, including 43 with haemoglobinuria and 86 without haemoglobinuria. Sixty-eight were females (52.7%) and 61 males (47.3%). The baseline data is provided in the table 1. Mean age was 8.75 years (SD=3.73) for cases and 8.55 years (SD=3.77) for controls and 8.62 (DS=3.84) years for the whole study population. Using Combi 9 dipstick, we identified (1+) hemoglobinuria in 27% of patients, (2+) in 18% and (3+) in 53%. When evaluated with the spectrophotometer, the average hemoglobinuria value was 0.061±0.0166mg/L in patients with (1+), 0.3986±0.2612mg/dL in those with (2+) and 0.5679±0.27688mg/L in those with (3+).
|
Case (n=43) |
Control (n=86) |
Total (n=129) |
OR (IC95%) |
P |
|
|
Distribution for age |
|||||
|
- ≤5 years |
8 (18.6) |
20 (23.3) |
28 (21.7) |
1 |
|
|
- >5 years |
35 (81.4) |
66 (76.7) |
101 (78.3) |
1.33 (0.53-3.32) |
0.676 |
|
Sex n (%) |
|||||
|
- Male |
21 (48.8) |
40 (46.7) |
61 (47.3) |
1.10 (0.53-2.28) |
0.803 |
|
- Female |
22 (51.2) |
46 (53.5) |
68 (52.7) |
1 |
|
|
Season |
|||||
|
- Rainy |
38 (88.4) |
51 (59.3) |
89 (69.0) |
5.22 (1.87-14.56) |
<0.001 |
|
- Dry |
5 (11.6) |
35 (40.7) |
40 (31.0) |
1 |
|
|
Plasmodium |
|||||
|
- Falciparum |
37 (86.0) |
73 (84.9) |
110 (85.3) |
1.10 (0.39-3.12) |
0.860 |
|
- Falciparum-malariae |
6 (14.0) |
13 (15.1) |
19 (14.7) |
1 |
|
|
Parasitemia (parasites/microliter) |
|||||
|
- Low |
33 (76,7) |
43 (51.8) |
76 (61.3) |
3.31 (1.41-7.78) |
0.005 |
|
- High |
10 (23.2) |
40 (48.2) |
48 (38.7) |
1 |
Table 1: Socio-demographic features of patients in the study population.
In comparison to the quantitative method, the sensibility was 100% for medi test combi 9 and 93% for both parameter 9 and 10 from cypress diagnosis. Specificity and positive predictor value were 100% for all types of dipsticks. The negative predictive value was 100% for medi test combi 9 and 96.6% for both parameter 9 and 10 of cypress diagnosis.
Figure 1 shows the correlation between the concentration of hemoglobin in dark urine and optic density obtained by spectrophotometric measurement using the equation y=0.544x, in which y represents the optic density and x the concentration of hemoglobin in urine.
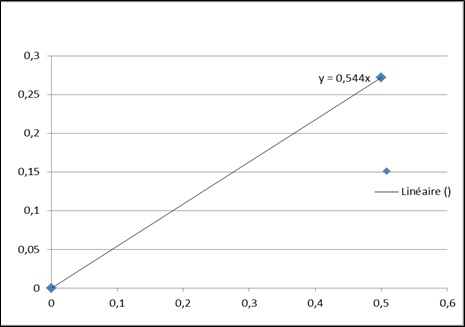 Figure1: Standard curve with correlation between optic density and the concentration of hemoglobin in urine.
Figure1: Standard curve with correlation between optic density and the concentration of hemoglobin in urine.
Note: This figure below was established to show the correlation between the concentration of hemoglobin in dark urine and optic density obtained by spectrophotometric measurement using the equation y=0.544x. y represents the optic density an x, the concentration of hemoglobin in urine.
The dipstick Medi test combi®9 presented with maximal (100%) for sensibility, specificity, positive predictor and negative predictor value (Table 2).
|
Dip stick |
Sensibility (%) |
Specificity (%) |
PPV (%) |
NPV (%) |
|
MeditestComb®9 Parameters 9 Parameters 10 |
100 93 93 |
100 100 100 |
100 100 100 |
100 96.6 96.6 |
Table 2: Specificity, sensibility, PPV and PNV of dip stiks used in the study.
Note: The dip stick Medi test combi®9 presented maximal (100%) for sensibility, specificity, positive predictor and v negative predictor value.
Table 3 shows that dipstick medi test combi9, discriminates low concentration of hemoglobin in urine. At 1+, the concentration of hemoglobin was: 0.0618±0.0166g/L with CI: 0.607-0.629 and p=0.001.
|
Medi test Combi9 |
Quantitative Hemoglobinuria (mg/L) |
CI (95%) |
P |
|
+ (12) |
0.0618± 0.0166 |
0.607-0.629 |
0.001 |
|
++ (8) |
0.3986±0.2612 |
0.3767-0.4206 |
NS |
|
+++(23) |
0.5679±0.27688 |
0.4656-0.6703 |
NS |
|
Total: 43 |
0.4031±0.27688 |
0.3169-0.4894 |
|
Table 3: Correlation between Dip stick medi test combi9 interpretation and quantitative concentration of hemoglobin confirmed by spectrophometer.
Note: Dipstick medi test combi9, detect low concentration of hemoglobin in urine. At 1+, the concentration of hemoglobin was: 0.0618±0.0166g/L with CI0.607-0.629 and p=0.001.
DISCUSSION
Every single method has its strength and weaknesses or limits, and users should be aware of these specificities when choosing a technic and when doing the interpretation of results. Various tests are used in the detection of hemoglobin in urine [12,18-20,22,24]. The purposes of this study was to test 3 dipsticks used in pediatric nephrology at University Hospital and evaluated their diagnostic value in children suffering Blackwater fever at the University hospitals of Kinshasa in comparison to the quantitative measure (using the reagent 3, 3′ dimethylbenzidine as gold standard).
Among the 3 dipsticks assessed during this study, we have observed a good correlation between the spectrophotometer and the Medi test Combi9® dipsticks for the detection of haemoglobinuria. Previous studies reported that majority of dipsticks could detect hemoglobin when the concentration was above 0.05g/L for urine or above 0.001g/L in case of blood [22,23]. With Dipsticks medi test Combi9®, we were able to detect hemoglobinuria at the concentration of 0.0618±0.0166g/L (p=0.001), which correlated to 1+ of on the dipsticks. Interestingly, this average detection threshold was within the range of 0.05 set by previous studies [22,23]. Given that this tool was able to detect even lower levels of hemoglobinuria, we anticipate that Medi 9 Combi9® can be used for early stage detection of haemoglobinuria. This tool well used is the solution in limited resources countries to measure hemoglobin in urine. It can significantly help in the situations of limited resources with the absence of equipped laboratories, absence of electricity, or reagents. Even in rural area, dipsticks can be easily used. A guideline for the screening of hemoglobinuria in resources limited setting should recommend the use of dipsticks.
Basically, the best screening test should be both very sensitive and very specific. Unfortunately, only very few tests satisfy these requirements [28]. Based on the specificity, sensibility, positive and negative predictor value of the Medi test Combi9® dipstick in the current study, all at 100%. Our results suggest that Medi test Combi9® dipstick is an adequate screening tool for hemoglobinuria [29-31]. Another quality of Medi test combi9 in the diagnosis of hemoglobinuria is the high positive and negative predictor value which is important in the precise and concise diagnosis [24,32]. Previous attempts to establish correlation between urinary bilirubin detection with dipstick and liver function showed that positive dipstick detection of bilirubinuria was not a reliable reflect of liver function [33,34]. In the light of our result, we can conclude that dipstick detection of hemoglobinuria, although not informative for defective liver function, should be considered as a useful reflect for massive intravascular hemolysis as observed in Blackwater fever. Since majority of LMIC are exposed to malaria, sickle cell disease and other diseases causing massive intravascular hemolysis, and threatening kidneys, Medi test Combi9® dipstick should be recommended as screening tool for haemoglobinuria in this setting in case of anaemia associated to one of these diseases, even when hemoglonuria is not clinically obvious yet.
It has been reported that the concentration of ascorbic acid (vitamin C) influences dipstick result for hemoglobinuria [35]. A good dipstick for hemoglobinuria should simultaneously assess ascorbic vitamin allowing hemoglobin results to be criticized with regards to the ascorbic vitamin. Thus, dipstick assessing both hemoglobin and ascorbic acid in urine, such as the Combi9 dipstick used in this study, should be recommended.
CONCLUSION
Strengths of the study
Because of the high frequency of disease with hemoglobinuria in DRC, early screening efforts should be encouraged in patients with dark urine. This study reports that dipstick: Medi test Combi9® is the best due to his sensibility, specificity, positive and negative predictors value. This dipstick can be strongly recommended in the screening of fresh dark urine for detection of hemoglobin in urine.
Limitations of the study
It is a case-control study of consecutively recruited patients with a single measurement of hemoglobinuria in urine and a relatively small sample size, thus limiting the generalization of the results to the entire population of patients with dark urine. In addition, the types and the number of dipstick were also underrepresented limiting the generalization of results obtained. We need to test all dipstick used in the field for more evidence.
The authors declare that they have no competing interests.
AUTHORS’ CONTRIBUTIONS
CNN, PMT, MNA and JMB, conceived, designed, deployed and directed the case-control study at the Department of Paediatrics at Kinshasa University Hospital and wrote the manuscript. RLL carried out patient recruitment and follow-up, sample collection, storage and transport. NZM performed the measurement of hemoglobin in urine, using spectrophotometer with the reagent 3,3′ diméthylbenzidine. KH, JV, AL and LT brought very precious corrections. PKK and PPA analyzed data. AO edited the English corrections. EKS and NMN corrected the manuscript before submission. All authors read and approved the final manuscript.
ACKNOWLEDGMENT
The authors thank all the children, guardians and parents who participated in this present study. We thank prof. Mvumbi Lelo and prof. Kamango Erick for the spectrophotometer at the Department of Molecular Biology at Faculty of Medicine at University of Kinshasa. We thank professor Fons Verdonck for getting scholarship to finish writing paper in Belgium.This research project was supported by the Katholieke Universiteit de Leuven, Belgium through the program of scholarship for young Congolese researchers (Bourses chaires scientifiques pour jeunes congolais) obtained through prof Jan Dequeker.
REFERENCES
- Bodi MJ, Nsibu NC, Longenge LR, Aloni MN, Akilimali PZ, et al. (2013) Blackwater fever in Congolese children: A report of clinical, laboratory features and risk factors. Malar J 12: 205.
- Aloni MN, Nsibu CN, Meeko-Mimaniye M, Ekulu PM, Bodi JM (2012) Acute renal failure in Congolese children: A tertiary institution experience. Acta Paediatr 101: 514-518.
- Gobbi F, Audagnotto S, Trentini L, Nkurunziza I, Corachan M, et al. (2005) Blackwater fever in children in Burundi. Emer Infect Dis 7: 1118-1120.
- Rogier C, Imbert P, Tall A, Sokhna C, Spiegel A, et al. (2003) Epidemiological and clinical aspects of blackwater fever among African children suffering frequent malaria attacks. Trans R Soc Trop Med Hyg 97: 193-197.
- Lawrence SG (1993) G6PD deficiency as protection against falciparum Malaria: An epidemiologic critique of population and experimental studies. American Journal of Physical Anthropology 36: 153-178.
- Janus J, Moerschel SK (2010) Evaluation of anemia in children. Am Fam Physician 1: 1462-1471.
- Bruneel F, Gachot B, Wolff M, Bedos JP, Regnier B, et al. (2002) Blackwater fever. Presse Med 32: 1329-1334.
- Olga PS, Correa DF (2003) Black water fever: A case-report. Rev Cubana Med Trop 55: 50-53.
- Rother RP, Bell L, Hillmen P, Gladwin MT (2005) The clinical sequelae of intravascular hemolysis and extracellular plasma hemoglobin: A novel mechanism of human disease. JAMA 293: 1653-1662.
- Arruda MM, Rodrigues CA, Yamamoto M, et al. (2010) Paroxysmal nocturnal hemoglobinuria: From physiopathology to treatment. Rev Assoc Med Bras 6: 214-221.
- Frank C, Brauckmann C, Palos M, Arsene CG, Neukammer J, et al. (2017) Comparison of potential higher order reference methods for total haemoglobin quantification-an interlaboratory study. Anal Bioanal Chem 409: 2341-2351.
- Taulier A, Levilain P, Lemonnier A (1986) Intérêt de la spectrophotométrie dérivée pour le dosage de l’hémoglobine plasmatique et urinaire: Comparaison avec la méthode utilisant la correction d’Allen. Ann Biol Clin 44: 242-248.
- Copeland BE, Dyer PJ, Pesce AJ (1989) Hemoglobin determination in plasma or serum by first derivative recording spectrophotometry: Evaluation of the procedure of Soloni, Cunningham, and Amazon. Am J Clin Pathol 92: 619-624.
- Olsson T, Bergström K, Thore A (1984) Chemiluminescent immunosorbent assay of serum myoglobin based on the luminol reaction. Clin Chim Acta 138: 31-40.
- Ahlquist DA, Schwartz S (1975) Use of leuco-dyes in the quantitative colorimetric microdetermination of hemoglobin and other heme compounds. Clin Chem 21: 362-369.
- Slaunwhite D, Clements J, Tuggey RL, Reynoso G (1979) Leucomalachite green assay for free hemoglobin in serum. Am J Clin Pathol 72: 852-853.
- Fairbanks VF, Ziesmer SC, O’Brien PC (1992) Methods for measuring plasma hemoglobin in micromolar concentration compared. Clin Chem 38: 132-140.
- Chalmers AH, Snell LE (1993) Estimation of plasma and urinary hemoglobin by a rate spectrophotometric method. Clin Chem 39: 1679-1682.
- Goyal MM, Basak A (2009) Estimation of plasma hemoglobin by modified kinetic method using 0-Tolidine. Indian J Clin Biochem 24: 36-41.
- Gauer GF (1985) Clinicopathologic evaluation of early renal disease in dogs. Comp Cont Educ Pract Vet 7: 32-38.
- Pages JP, Troillet TJL (1990) Les protéinuries. Prat Med Chir Anim Comp 25: 585-597.
- Jensen AL, Kjelgaard HM (2006) Method comparison in the clinical laboratory. Vet Clin Pathol 35: 276-286.
- Le Bricon T (2001) Exploration biologique de la protéinurie au laboratoire d'analyses: Aspects quantitatifs. Ann Biol Clin 59: 701-715.
- Pels RJ, Bor DH, Woolhandler S, Himmelstein DU, Lawrence RS (1989) Dipstick urinalysis screening of asymptomatic adults for urinary tract disorders. II. Bacteriuria. JAMA 262: 1221-1224.
- Patel HP (2006) The abnormal urinalysis. Pediatr Clin North Am 53: 325-337.
- Assoumanou MG, Akpona S (2011) Dosage de l’hémoglobine urinaire par un réactif 3,3′ diméthylbenzidine : Mise au point technique. Int J boil chem sci 5: 479-89.
- Drabkin DL (1946) Spectrophotometric studies; the crystallographic and optical properties of the haemoglobin of man, in comparison with those of other species. J Biol Chem 164: 703-723.
- Nendaz MR, Perrier A (2004) Sensibilité, specificité, valeur prédictive positive, valeur prédictive négative d’un test diagnostique. Revue des maladies respiratoires 21: 390-393.
- Bodi J, Nsibu C, Aloni M, Lukute GN, Kunuanuna TS, et al. (2014) Blackwater fever associated with acute renal failure among Congolese children in Kinshasa. Saudi J Kidney Dis Transplant 25: 1352-1358.
- Kunuanunua TS, Nsibu CN, Gini-Ehungu JL, Bodi JM, Ekulu PM, et al. (2013) Acute renal failure and severe malaria in Congolese children living in Kinshasa, Democratic Republic of Congo. Nephrol Ther 9: 160-165.
- Aloni MN, Nsibu CN, Meeko-Mimaniye M, Ekulu PM, Bodi JM (2012) Acute renal failure in Congolese children: A tertiary institution experience. Acta Paediatr 101: 514-518.
- Woolhandler S, Pels RJ, Bor DH, Himmelstein DU, Lawrence RS (1989) Dipstick urinalysis screening of asymptomatic adults for urinary tract disorders. I. Hematuria and proteinuria. JAMA 262: 1214-1219.
- Foley KF, Wasserman J (2014) Are unexpected positive dipstick urine bilirubin results clinically significant? A retrospective review. Winter 45: 59-61.
- Binder L, Smith D, Kupka T, Nelson B, Glass B, et al. (1989) Failure of prediction of liver function test abnormalities with the urine urobilinogen and urine bilirubin assays. Arch Pathol Lab Med 113: 73-76.
- Ko DH, Jeong TD, Kim S, Chung HJ, Lee W, et al. (2015) Influence of vitamin C on urine dipstick test results. Ann Clin Lab Sci 45: 391-395.
Citation: Bodi JM, Ngole M, Nsibu CN, Longenge RL, Monkoti MM, et al. (2021) Usefulness of Medi Test Combi 9 in the Diagnosis of Hemoglobinuria in Limited Recourse Setting. J Nephrol Renal Ther 7: 044.
Copyright: © 2021 Joseph Mabiala Bodi, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

