
Using Over-the-Counter and Other Prescription Medications to Potentiate Opiates in the USA: Literature Review. Medical and Public Health Aspects of OTC Medication Misuse
*Corresponding Author(s):
Valeriy ZSchool Of Behavioral Sciences, California Southern University, Costa Mesa, United States
Tel:+16263897415,
Email:Zvonarevval@gmail.com
Abstract
Opiates are generally indicated for the treatment of various pain syndromes; however, if alternate treatments have been exhausted and a physician elects to prescribe opioids, then both the physician and patient should be aware of the risks associated with use. More that 10% of patients prescribed opioids go on to misuse their prescription, and opioids account for three-quarters of all fatal prescription drug overdoses. Opioid boosters are drugs, herbs, or substances that are utilized to intensify the effects of the opioid. The use of potentiates goes back to as early as the 1600s where it is reported that morphine was sometimes combined with cocaine and alcohol to intensify its opioid effects. Today, the use of potentiates is aggravating the issue of opioid abuse. While there are many approaches by which opioid effects can be intensified illicitly (e.g. snorting, or crushing tablets, evaporating liquid opioids via vapes, mixing liquid opioids with sodium bicarbonate, consuming grapefruit juice, etc), this article is intended to discuss most medications that are utilized as opioid boosters or potentiates. The article is not intended to provide a complete list of all potential boosters, however, we believe that it is an educational material to enhance our understanding and combat the abuse of opioids.
Keywords
Diphenhydramine; Herbs; Hydrocodone; Opioid abuse
INTRODUCTION
The opioid crisis was declared to be a national emergency in the United States by President Trump. Unfortunately, it seems that the president overlooks the contribution that pharmaceuticals are making in the development of this national epidemic. In the 1990s pharmaceutical companies gave assurances that people would not become addicted to opioids, which led to a significant outburst in these drugs being prescribed to patients. Naturally, the increased rate of prescriptions resulted in the increased misuse of opioids, and hence addiction. The United States, with only 5% of the world population, consumes over 80% of the opioids produced globally. The American Society of Addiction Medicine classifies opioids as both illicit drugs such as heroin and licit drugs such as prescribed pain relievers (e.g. oxycodone, codeine, hydrocodone, fentanyl and morphine) [1]. This Society declared that in 2015 there were 20.5 million Americans aged 12 years and above who had substance use disorder, and among them Two million were related to continuous use of prescribed pain relievers and 591,000 to illicit heroin use with 23% of first-time heroin users developing opioid addiction. There is a close statistical relation between the use of heroin and over-the-counter (OTC) medication. It was revealed that four out of five heroin users in the US started out by misusing OTC medication. Currently, 40% of users are potentiating their opiates with enhancers. Commonly used OTC enhancers include grapefruit juice, cimetidine, codeine, diphenhydramine, quinine, vitamin supplements, gabapentin and clonazepam, among others. The study aims to provide the first literature review on the topic. In this case, our research will be aimed at finding the ways opiate drug abusers are using OTC and other prescribed medications to potentiate intoxication effects of the opiates, and also to identify the main reason people are potentiating opiates. The study will also investigate other drugs that are used to potentiate opiates. We believe there is a need for further research among street drug users with the aim of clarifying our understanding of opiates and their use. This study is motivated by the previously published literature and by the needs of society, and it will analyze data regarding the use of opiate boosters (OTC and other prescription medications) in the USA along with user feedback to provide information about the evolving abuse and suggest new approaches to combat opioid misuse.
BACKGROUND
National Opioid Overdose Epidemic statistics demonstrate that drug overdoses in the US are the major cause of deaths due to accidents. In 2015, there were 52,404 registered overdose cases in the country with opioid overdose accounting for 38% (20,101 cases) of the total number of accidents. Within the decade from 1999 to 2008, the use of opiate boosters [2], overdose death rates and OTC treatment admissions quadrupled. Addicts wanted to increase the power of opiates using medication and non-medication substances. In many cases, the misuse of other psychoactive substances could lead to potentiating of the opioid-related euphoria resulting in addictive behavior. However, this phenomenon plays only a secondary role compared to opioid (especially heroin) addiction. Intravenous drug users (IVDU) are striving to stay euphoric for a long time. However, it’s not always possible. This is either because opiates are very expensive to be used permanently or due to the fact that these drugs are not available in some countries, like Great Britain. Because of this, users are now potentiating opiates with enhancers or boosters. According to the National Institute of Drug Abuse, men are more affected by the use of opioids than women [3]. However, women using these drugs are more dependent on them than men. The number of women affected by these drugs still remains significant. Currently, a high level of OTC overdose is observed with females and is increasing by 400% compared to males with an increase by 237% [4]. More and more adolescents between 12 to 17 years of age are also being affected by overdose. There are over 276,000 adolescents using OTC drugs for non-medical purposes; 21,000 have used heroin in the past, while 5,000 adolescents are still using this drug. Therefore, heroin still remains a common opioid drug in this age category. Ethical and racial differences are also characteristic in the use of drugs in the US. According to the National Institute on Drug Use and Health, 38. 2% of white young adults are using illicit substances. They are followed by 30. 6% of Afro-American and finally by 27. 5% of young Hispanic adults [5]. This study was carried out among college students of Midwestern University, as different races are represented among them. It should be noted that as people growolder, their addiction to drugs is only increasing. The Substance Abuse and Mental Health Services Administration found that 28. 5% of those between 18 to 21 years used drugs, while this figure amounted to 43. 3% with young people between 21 and 25 years of age [6]. Table 1 presents the most common opiate agents.
|
Opioids |
|||
|
Drug |
Mechanism of action |
Indication |
Notes |
|
Heroin |
Strong µ agonist |
Manage pain, suppress cough, antimotility for diarrhea Contraindications: phenothiazine, MAO, Inhibitors, and tricyclic antidepressants (depressant effects); alcohol and benzodiazepine (respiratory effects); hepatic metabolism
|
Often abused IV; not legal in US |
|
Morphine |
Strong µ agonist |
Prototypical opioid; various routes of admin |
|
|
Fentanyl |
Strong µ agonist |
Rapid onset and offset with small dose; CV stability; 100x more potent than morphine |
|
|
Methadone |
Strong µ agonist |
Also used for opioid/heroin withdrawal; racemic mixture of NMDA antagonist and mu agonist |
|
|
Meperidine |
Strong µ agonist |
No binary SE, doesn’t constrict sphincter of Oddi; seizures |
|
|
Codeine |
Moderate µ agonist |
Less potent than morphine |
|
|
Hydrocodone |
Moderate µ agonist |
Most prescribed opiate; often combined with NSAIDs/acetaminophen |
|
|
Oxycodone |
Moderate µ agonist |
Similar to hydrocodone |
|
|
Tramadol |
Weak µ agonist |
Synthetic codeine; lower addiction risk; can cause seizures and serotonin syndrome |
|
|
Buprenorphine |
µagonist; ? antagonist |
Analgesic: deterrent, detoxification |
High affinity, low efficacy at mu receptor → partial agonist |
|
Nalbuphine |
? agonist; µ antagonist |
Treats opioid-induced pruritus |
Originally hoped to be less addictive, less side effects than other opioids → no |
|
Naloxone |
µ antagonist (short acting) |
Treat opioid addiction, overdose, and toxicity; reverses mu agonist effects; increases respiratory rate within 1-2 min |
Better for opioid overdose; half-life is 1 hour |
|
Naltrexone |
µ antagonist (long acting) |
Treats alcoholism; lasts 24 hours after moderate dose |
|
Table 1: Most common opiate agents.
Opiate metabolism: Active and inactive metabolites are produced during the metabolism of opioids. It is established that active metabolites are more potent compared to the parent compound. Before getting into the systemic circulation, most of the opioids have to undergo extensive first-pass metabolism, which occurs in the liver. These phases are modification reactions (Phase 1) and conjugation reactions (Phase 2). Phase 1 metabolism occurs by CYP pathways, while Phase 2 occurs by conjugation or both. CYP3A4 and CYP2D6 enzymes are involved in Phase 1 metabolism. Over 50% of all drugs are metabolized by the CYP3A4 enzyme. Because of this, there is a high likelihood of drug-drug interactions when opioids are metabolized by the CYP3A4 enzymes. Another enzymes, CYP2D6 is involved in the metabolism of fewer drugs. There is substantial interaction potential in each of these opioids with other drugs that are commonly used. This is especially important, if drugs are inducers, substrates or inhibitors of CYP3A4. Glucuronidation is the most important Phase 2 reaction. UGT2B7 enzyme is important in opioid metabolism via glucuronidation of the following medications: hydromorphone, morphine, and oxymorphone. The metabolism of several key opioids to assist in the interpretation of toxicology testing results is presented in (Figure 1).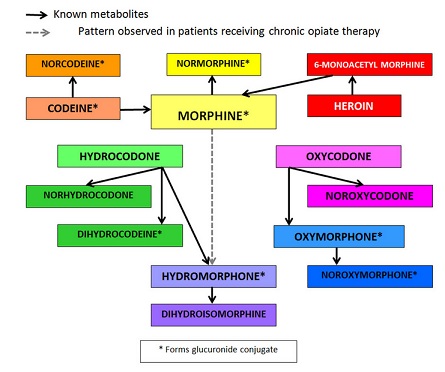
Figure 1: Opiate metabolism.
Source: Medical Pharmacology. Chapter 13: Pain Management opioids https://www.pharmacology2000.com/Central/Opioid/New_Opioid3.htm
POTENTIATORS
A potentiator is a compound, herb, or other medication that is utilized to expand the impacts of a substance. For many years, individuals have been combining substances with an end goal to accomplish a more noteworthy “elevated” effect. Indeed, even with apparently innocuous ordinary substances, unintended results may emerge , and consolidating substances expands the potential for addiction and overdose. The utilization of potentiators is intensifying the issue of sedative maltreatment. The intravenous (IV) injection and oral (PO) use of different OTC medications for non-clinical purposes is a new and widespread drug trend. In addition to rewarding effects, the non-clinical use of substances such as tropicamide, H2-blockers and heroin claim a role for pharmacological interactions in influencing drug association in a poly abuse pattern. The most common opiate potentiators are identified in (Table 2).
|
Opiate Potentiator Group |
Example |
Mechanism of Action |
|
Antihistamines
|
Diphenhydramine (Benadryl) Promethazine Chlorpheniramine Cyclizine Doxylamine succinate |
H1 - antagonists |
|
H2 - antagonists |
Cimetidine |
It competitively inhibits histamine binding to histamine H2 receptors.It also blocks the activity of cytochrome P450, which might explain proposals for its use as apotentiator. |
|
CYP enzymes inhibitors
|
Grapefruit juice
|
Grapefruit contains compounds known asfuranocoumarins that block the CYP3A4 enzymes. |
|
NMDA antagonists
|
Memantine
DXM (dextromethorphan)
Magnesium sulfate |
Memantine blocks the effects of glutamate, a neurotransmitter in the brain that leads to neuronal excitability and overstimulation of neurons. DXM is a nonselective serotonin reuptake inhibitor; sigma-1 receptor agonist; as an NMDA receptor antagonist, produces effects similar to ketamine and phencyclidine. Active metabolite is 3-methoxymorphinan, which produces local anesthetic effects in animal models with a potency above dextrorphan, but below dextromethorphan itself. Magnesium: unclear mechanism of action. Inhibits the NMDA receptors limiting the effect of excitotoxic neurotransmitters such as glutamate and increasing the seizure threshold. Mg causes cerebral vasodilation and decreases cerebral ischemia. Smooth muscle relaxant. |
|
Muscle-relaxants
|
Magnesium
Orphenadrine
Cyclobenzaprine
Carisoprodol
Baclofen |
Magnesium acts by binding to and lowering the activity of NMDA receptors through blocking of the calcium channels that are coupled with these receptors. It also lowers the synthesis of substance P and potentiates morphine action at the dorsal horn, specifically at the presynaptic region. This results in an increase in the action of morphine when co-administered with magnesium. Orphenadrine appears to block muscarinic acetylcholine receptors as well as NMDA receptors in the CNS. Cyclobenzaprine is a centrally acting skeletal muscle relaxant. Carisoprodol is a centrally acting skeletal muscle relaxant that does not directly relax skeletal muscles. A metabolite of carisoprodol, meprobamate, has anxiolytic and sedative properties. Baclofen, on the other hand, acts as an agonist at GABA B receptor. This receptor overlaps in expression with those of opioids. By binding to this receptor, this drug produces a synergy in action with opioids due to potentiation of the analgesic and hypnotic effects of opioids. |
|
Antimalarial medications
|
Quinine
|
Quinine inhibits nucleic acid synthesis, protein synthesis, and glycolysis inPlasmodium falciparum and could bind with hemazoin in parasitized erythrocytes. However, the precise mechanism of the antimalarial activity of quinine sulfate is not completely understood. |
|
CCK-inhibitors |
Proglumide
|
CCKa and CCKb- inhibitor. It enhances analgesia produced by opioid drugs and could prevent or even reverse the development of tolerance to opioid drugs [7,8]. |
|
Opiate receptor agents |
Loperamide
Propoxyphene
|
Loperamide: agonist at mu-opioid receptors; slows gut motility. Poor CNS penetration (low addictive potential in therapeutic dose). Propoxyphene: It acts as a mu-opioid receptoragonist, noncompetitive α3β4 neuronal nicotinic acetylcholine receptor antagonist and weak serotonin reuptake inhibitor. Dextromethorphan is a weak opioid like codeine and known to cause dependency among recreational users. |
|
Other CNS medications
|
Clonidine
Benzodiazepines Clomipramine
Barbiturates Stimulants |
Clonidine: alpha 2 agonist. It blocks sympathetic outflow of norepinephrine through stimulation of alpha 2 receptors in the brain resulting in sympathetic tone reduction. Benzodiazepines: facilitate GABAa action by increasing frequency of Cl channel opening. Clomipramine: TCA antidepressant Barbiturates: bind to the GABA receptor complex and decrease the rate of GABA dissociation. Stimulants:dextroamphetamine induces the release of dopamine within the mesocorticolimbic system, a major component of the brain reward system resulting in measurable behavioral changes such as euphoria. Methylphenidate blocks the dopamine transporter causing an increase in dopamine concentration at the synapse. |
|
COX inhibitors |
Naproxen
|
Reversibly inhibits cyclooxygenase-1 and 2 (COX-1 and 2) enzymes, which results in decreased formation of prostaglandin precursors. |
Table 2: Most common opiate potentiators.
Other substances with opiate potentiating effect are presented in (Table 3).
|
Substance |
Use |
Mechanism of Action |
Possible Potentiation of Opiate Drugs |
|
Cat’s claw |
Used in the treatment of arthritis, infections, autoimmune conditions,cancer management. |
Components of Cat’s claw increase the number of white blood cells in the body thus they boost immunity, NMDA properties. |
Cat’s claw increases methadone effect [9]. |
|
Baking soda |
Treatment for acid reflux and GERD. |
Baking soda neutralizes the acid in the stomach responsible for irritation. End products of the reaction are water, salt, and carbon dioxide, which do not irritate the stomach lining. |
Oral ingestion only; affects pH of the stomach to allow more opiates to be absorbed into the blood.
|
|
Rhodiola |
Decreases stress, improves brain function, treatment of depression, management of diabetes and cancer. |
Rhodiolaprovides human cells with a property that makes them resistant to destruction, thus they develop resistance to most of the attacks against it. In regard to brain function, the herb helps in sustaining neurotransmitters like serotonin, dopamine and norepinephrine, which are responsible for memory, speed, concentration and memorizing. |
Rhodiolarosea L. increases both development and expression of morphine dependence [10].
|
|
Ashwagandha |
Management of stress, anxiety, attention deficit disorder, bipolar disorder, diabetes, high cholesterol levels, male infertility. |
1. Increases production of insulin, thus controls blood sugar levels in blood. 2. Stimulates cell apoptosis. 3. Decreases stress and anxiety. 4. Boosts testosterone. 5. Stimulates the breakdown of cholesterol and triglycerides. |
Clinical studies show potentiation and tolerance reversal [11].
|
|
Black seed (Nigella sativa) |
Treatment of headache, nasal congestion, gas colic, diarrhea, asthma, cough, lowering blood pressure. |
Black seed stimulates production of white blood cells. Components also slow down allergic reactions in the body, thus acting as antihistamines, which is essential in such respiratory conditions like asthma. This substance was reported to be a potentiator, but confirmedmechanisms are missing. |
N. sativa and its major active compound, thymoquinone (TQ), a calcium channel blocking agent,reduces withdrawal syndrome and opioid dependency [12]. |
|
Hordenine |
Athletic performance, weight loss. |
The herb stimulates the central nervous system thus increasing parameters like heart rate, blood pressure, and breathing rate, which are essential in athletic performance Hordenine has alkaloid properties, which play a part in digestion thus effecting weight management. |
Unclear potentiating effect; however, this phenolic alkaloid could cause false positives in morphine immunoassays of the beer drinkers urine [13].
|
|
Tribulusterrestris |
Enhancing libido, keeping the urinary tract healthy and reducing swelling. |
The herb contains compounds that are considered diuretics. Effective kidney filtration provides frequent passing of urine, thus flushing the urinary tract and resulting in a healthy system. Possible testosterone booster. Pain reliever in traditional medicine. |
Mild MAO-B inhibitor. It was concluded that T. terrestris has a mild analgesic effect, but exact pharmacological properties are unclear [14]. |
|
DLPA |
Treatment of vitiligo, depression, pain, alcohol withdrawal |
The essential amino acid stimulates production of dopamine, which is a chemical responsible for regulating moods in the brain and effective in the management of depression. DLPA reacts with UV light to enhance the skin to produce more pigment, thus essential in the management of vitiligo. Components are also combined with other amino acids in the body to relieve symptoms of alcohol withdrawal. |
Markedly potentiates opiate analgesia [15].
|
|
Curcumin |
Treatment of inflammation, prevention of cancer, prevention of Alzheimer’s disease. |
Curcumin reduces the action of chemicals responsible for inflammation. The compound neutralizes free radicals in the body, which are responsible for abnormal growth of cells. It also stimulates other cells to produce antioxidant enzymes. Curcumin increases the number of hormones responsible for the growth of neurons in the brain, as well as prevents degenerative processes there. |
According to various reports, 2g taken with piperine potentiates the potentiator [16]. It was suggested that curcuminmay attenuate opioid tolerance and dependence by suppressing CaMKIIα activity. |
|
Dimethylsulfoxide (DMSO) |
Treatment of bladder inflammation, skin conditions, management of inflammatory pain. |
The drug facilitates absorption of other drugs across membranes. It also possesses anti-inflammatory effects, thus managing pain. When used topically, the drug resolves skin breakages like herpes zoster and blisters from cancer treatment. |
DMSO increases absorption of opiates taken orally thus potentiating their analgesic effect in the body due to the increased availability of the binding proteins [17]. |
Table 3: Other medications and herbal medicines.
OPIATE POTENTIATORS
General overview
Opiates could be mixed with other substances to potentiate or increase their effects. According to [18], opiate potentiating means enhancing the effects of opioids or opiates by mixing them with another drug or substance. Although using potentiates boosts the effects of opiates, it is risky and could be associated with life-threatening side effects. Therefore, opioid potentiators must be used with extreme caution because they are relatively safe when used as prescribed by a doctor. According to previous data, the research related to prescription or OTC drug abuse is still in its early stages, which is why the problem has yet to be solved. According to the National Institute on Drug Abuse (NIDA), men are more affected by the use of opioids than women. However, women who use these drugs are more dependent on them than men are. There are over 276,000 adolescents using OTC drugs for non-medical purposes, and currently overdosing with OTC medication is high among women: it has increased by 400% compared to an increase of 237% in men. According to several studies, the use of cocaine, heroin, and other abused drugs are well researched in the USA, but opioid potentiators are always overlooked. Participants were 586 users of these drugs from the streets. The methodology of this research indicated that methadone was extensively used (71. 9%) and sold (64. 7%), when compared to oxycodone (OxyContin), oxycodone/acetaminophen (Percocet) and hydrocodone/acetaminophen (Vicodin), which were used by about 35% and sold by about 34% of the sellers. We will try to describe how people from all over the nation mix opiates with different medications and various substances. More large-scale studies need to be carried out to confirm and better describe the extent of opiate enhancer misuse in the USA and elsewhere.
Gabapentinoids
The first paper that was helpful in this study appeared to be one that explained issues that surrounded the abuse of opiates and their enhancers. In a survey carried out in substance misuse clinics, 22% of respondents admitted abusing gabapentin or pregabalin; of these, 38% abused gabapentinoids to potentiate the “high” obtained from methadone. The possible mechanism of action for the potentiation of an opiate “high” could be related to gabapentin’s ability to increase the analgesic effect of opiates. It was also hypothesized that the increased analgesic effect of gabapentin and morphine could be contributed to by the increase in gabapentin serum concentration that results from the two medications being given together. When combined with opiates, the risk of respiratory depression and drug-related mortality increases. Gabapentin users reported a range of subjective symptoms including euphoria, enhanced sociability, state of relaxation, sedative or opiate-like comedown, psychedelic and MDMA-like effects. In a subsequent national (US) assessment of medical harm resulting from gabapentin and opioid co-abuse, ~24% of patients with sustained co-prescription of gabapentin and opioids had at least three prescription claims exceeding established dosage thresholds; as compared to the 3% and 8% of patients prescribed gabapentin or opioids alone, respectively [19].
Muscle relaxants
Numerous research articles were published with the purpose of explaining the relationship between various muscles relaxants and opioids, and the interactions that were expected to lead to potentiating of the opioids’ activity. Table 4 summarizes the most recent information regarding the use of muscle relaxants in co-administration with opiates. Major effects, including boosting effect, are described in detail.
|
Muscle relaxant |
Opiate agent |
Description |
|
Baclofen, meprobamate, carisoprodol, chlorzoxazone, methocarbamol, tizanidine, metaxalone, orphenadrine, and cyclobenzaprine. |
Oxycodone, methadone,buprenorphine |
Authors demonstrated in their study that through inhibiting metabolism of opioids in vitro, skeletal muscle relaxants sufficiently increaseduration of the opioids’ action [20]. |
|
Baclofen |
Fentanyl, oxycodone, morphine, and buprenorphine |
Researchers demonstrated that the overlap that existed in the expression of opioid receptors and GABA receptors had significant importance in the interaction that existed between opioids and baclofen. When administered together, these drugs showed synergistic activity in the production of analgesia [21]. |
|
Magnesium sulfate |
Various |
Magnesium acts through the antagonism of the various molecules’ activity at the N-methyl-d-aspartate receptors. This antagonism was implicated in its role as a muscle relaxant. Additionally, the antagonism was demonstrated to play a fundamental role in increasing the analgesic effect of opioids in the management of pain of chronic inflammatory origin, and pain that was acute in nature [22]. |
|
Magnesium sulfate |
Various |
Authors emphasized the fact that magnesium had the ability to potentiate the activity of opioids in such a way that low doses were needed to achieve the desired effect and made it likely to be abused by opioid addicts. In addition to the above, its antagonistic role at the N-methyl-d-aspartate receptors and its abundance in the body coupled with the risks associated with the use of opioids, such as potential for tolerance development, addiction, disorders of consciousness, and constipation that are chronic among others, there was an idea to use magnesium either as an adjunct to opioids or as their replacement especially against chronic pain or against migraines [23,24]. |
|
Magnesium sulfate |
Morphine |
According to the article presented, neuropathic pain that is associated with an excess stimulation of NMDA receptors obtains poor response in the use of morphine. Coadministration of an antagonist of the NMDA receptors, such as magnesium with morphine was shown not only to restore, but also to increase potency of morphine in managing the neuropathic pain [25]. |
|
Magnesium sulfate |
Morphine Tramadol Oxycodone |
Parenteral administration of magnesium sulfate in its micronized form was demonstrated to increase the antinociceptive activity of opioids in different types of pain [26].
|
|
Carisoprodol |
Opiates + benzodiazepines |
This is a centrally acting relaxant of muscles, which combined with opioids or even benzodiazepines significantly raise effects of these drugs. Researchers described this concept as the Holy Trinity, where abusers were combining a muscle relaxant, such as carisoprodol, benzodiazepine, and an opioid analgesic as a method of augmenting the “high” that they obtained from using either a benzodiazepine or opioid alone [27,28]. |
|
Carisoprodol |
Opiates + benzodiazepines |
Carisoprodolwas implicated as having a very high potential for abuse. The drug undergoes biotransformation in the hepatocytes through N-dealkylation and hydroxylation to form the primary metabolite, which is meprobamate. This connects and modulates the activity of GABAA receptors producing sedative-hypnotic effects within the central nervous system in a manner similar to that of the opioids [29,30]. |
Table 4: Summary of various muscle relaxants analyzed in previous studies. Major effects are reported.
While the above drugs interact in a way that could be employed to potentially lower the amount of opioid analgesics that are used particularly against chronic pain, their augmentation or potentiation of this activity could be exploited by abusers to attain a new level of euphoric reaction. When combined with opioids, the hypnotic effect is heightened thus it's potential for abuse. Carisoprodol exists in a combination formulation with aspirin at a strength of 200 mg/325 mg. This drug is administered orally and absorption occurs within 1.5 hours to 2 hours, when the concentration of drug in plasma attains its peak value. The half-life of the primary metabolite is 10 hours, and this is the molecule that is responsible for sedative-hypnotic activity. Having not been classified as a controlled drug, this drug is readily available particularly in certain online pharmacies, where it has a label of “no prescription needed” [31]. This increases its potential for abuse. Other than carisoprodol, baclofen is available in a tablet form with a strength of 10 milligrams. This is often administered in a standard regimen of three times a day, with the strength ranging from a minimum of 5 mg to a maximum of 25 mg. After oral administration, peak plasma concentration is attained after 1 hour to 3 hours, and it is eliminated within 3 hours to 4 hours. When given with opioids, it potentiates their activity, and has the potential for inducing withdrawal symptoms. Magnesium, at a strength of 500 mg is often given to patients, who are on opioid analgesics to lower the dose of opioids. Because of a half-life that lasts longer than 12 hours, the tablet could be administered once daily. Being a supplement, this drug is readily available and could easily be abused when combined with opioids.
BENZODIAZEPINES
Benzodiazepines are strictly controlled substances, and as such carry a risk of abuse on their own. More than 30% of opioid overdoses also include benzodiazepines. Both opioids and benzodiazepines are able to sedate users, suppress breathing, and impair cognitive function. A description of the combined use of benzodiazepines with opiates is presented in (table 5).
|
Agents Reviewed |
Description |
|
Diazepam + Methadone |
Diazepam was not able to inhibit the metabolism of methadone. No differences were reported in plasma levels of methadone or its metabolites. |
|
Diazepam Buprenorphine Fentanyl |
Eleven patients, who had previously received buprenorphine, suffered sudden respiratory depression requiring manual ventilation of their lungs followed by doxapram infusion. |
|
Diazepam Methadone |
The pupil constriction induced by diazepam + methadone combinations was greater than that induced by comparable doses of methadone alone. Diazepam 40 mg significantly increased opioid subjective effects, when compared to either of the drugs alone. |
|
Diazepam Methadone: 50 or 60 mg (mean = 56 mg), 100% and 150% of normal dose. |
No evidence of drug-drug interaction between the two agents. |
|
Methadone +Diazepam |
Rhabdomyolysis-induced acute renal failure due to methadone-diazepam overdose. |
Table 5: Review of opioid agents and benzodiazepine use.
BARBITURATES
Barbiturates are one of the most widely used potentiators for opioids. They are central nervous system depressants, and their mode of action involves reduction of nerve activity resulting in muscle relaxation. They also reduce blood pressure, breathing and heart rate and could be habit-forming [32]. All barbiturates are known to affect gamma-aminobutyric acid, i.e. a chemical neurotransmitter used by the nerves to communicate with each other. They are mainly administered for the treatment of headaches, seizures, and insomnia. Examples of common barbiturates available within the United States include butalbital, phenobarbital, secobarbital, pentobarbital, butobarbital and amobarbital. The most important problem with the use of potentiators with opioids is that it results in over-sedation, which manifests through inability to respond to any form of stimuli or wake up and sometimes causes users to slip into a coma. In addition, combination sometimes also results in changes in breathing patterns characterized by depressed breathing, which results in a state characterized by insufficient oxygen in the brain [33]. Effects of the potentiators on the euphoric impact of opioids depend on the method by which they are combined. One of the ways potentiators are taken to increase their euphoric impact is through the rectal route, whereby their effect was reported to increase by 10 percent when administered through this route. Another methods commonly used by the abusers of opioids involves heating the opioids with the potentiates to obtain a liquid with higher concentration that is then ingested through different forms. Potentiation of opioids are linked to a larger percentage of deaths associated with the abuse of opioids. Unfortunately, the impact of potentiation of opioids with various barbiturates wasn’t well described in previous studies. Table 6 summarizes the existing data about the impact of opioids’ potentiation by selected barbiturates.
|
Medication/Substance |
Dosage/Pattern of Use |
Effects Described |
|
Secobarbital withcodeine |
100mg |
Increased respiratory depression [34] |
|
Butalbital with codeine |
Over 60mg |
Hypotension, confusion, tachycardia or bradycardia, false feeling of wellbeing, dizziness, headache, nausea and vomiting, weakness, dyspnea and erratic CNS stimulation symptoms [35]. |
|
Pentobarbitol with morphine |
50mg/kg i.p. |
Increased sleeping time [36]. |
|
Phenobarbital with morphine |
60mg |
Reduced short-term tolerance to morphine [37]. |
|
Butalbital with codeine |
50mg |
Increased analgesia [38]
|
Table 6: Impact of opioids’ potentiation by selected barbiturates.
ANTIHISTAMINES
Antihistamines, such as promethazine, are characterized by misuse potential among patients utilizing opioids. Promethazine and codeine cough syrup, in combination with soda or juice, have a history of abuse and are referred to as “lean,” “purple drank” or “Sizzurp”. As CNS depressants, the effects of these drugs could increase the risk for euphoria, intoxication, respiratory depression, and death when combined with opioids. Cimetidine: According to [39], cimetidine functions by inhibiting the cytochrome P-450 enzymes, which are relevant in the metabolism of opioids, as well as of other drugs. Hence, it increases the duration of action of the opioids, causing an increase in the euphoric state. This drug is a histamine H? receptor antagonist, which significantly reduces the amount of hydrochloric acid in the stomach. The dosage of cimetidine varies depending on the type of disease under treatment. However, the most used dosage is 800 mg, which is mostly available over the counter and works effectively for a maximum of a single hour [40]. This agent exhibits various effects including the risk of acute liver injury resulting from the heavy workload for the same, headache, dizziness, gynecomastia, as well as somnolence. Furthermore, in cases of overuse it could also result in diarrhea, nausea, vomiting, confusion, hallucinations, disorientation, decreased sexual ability in men, abdominal pain, easy bruising, irregular heartbeat, as well as jaundice. Cimetidine is classified as a category B drug in pregnancy; hence, its use could be acceptable. Approximately 48%-75% of the drug is expelled in the urine, while less than 2% is excreted in bile. (Cimetidine 800 mg is available over the counter, which makes this a great opiate potentiator. According to several sources, it works for about an hour. Cimetidine is likely to affect the metabolism of codeine to morphine.
Diphenhydramine inhibits histamine, and also increases the analgesic, as well as the mood properties associated with opiates to a tiny degree. Essentially, this agent inhibits a subset of CYP2D6. Diphenhydramine is considered a histamine H? receptor blocker in the respiratory tract, gastrointestinal smooth muscles, as well as in the blood vessels. The drug is fundamentally used in relieving various allergic symptoms such as itching, rash, watery eye, running nose, sneezing, and cough. The drug also is useful in the prevention and treatment of nausea, vomiting, and dizziness during motion sickness. Diphenhydramine helps to relieve some side effects of antipsychotic medications [41]. The drug’s dosage is 25 mg-50 mg orally which is given every 6 to 8 hours. Its onset of action is between 5 minutes to 30 minutes. The drug’s effects include dry mouth, dizziness, abdominal pain, drowsiness, constipation, confusion, restlessness, irregular heartbeat, difficulty in passing urine, blurred vision, euphoria, as well as palpitation among several others. The drug increases the risk of falls and over sedation in the elderly patient making it a high-risk medication. According to previous data, this agent increases the analgesic and mood properties of opiates to a small degree. Administration of the drug alongside opiates results in the reduction of itchiness and better effects for the patient [42]. Importantly, taking more diphenhydramine than what is clinically necessary could result in hepatic injury.
PROMETHAZINE
Promethazine is an H? receptor blocker. In fact, it is notable that any of the other sedative anticholinergic antihistamines tends to work towards reducing various side effects of opiates and potentiation of analgesia [43]. Most importantly, this agent is strictly administered after the administration of opiates. The drug is used in allergic conditions including nausea and vomiting, postoperative sedation, motion sickness, preoperative sedation, as well as obstetric sedation. Promethazine is administered in 25 mg orally in a frequency of every 4-6hours.It has an onset of 3-5minutes when given via intravenous route and 20 minutes when given orally. Some of promethazine’s neuropsychiatric side effects include drowsiness, blurred vision, sedation, confusion, disorientation, euphoria, as well as catatonic states [44]. Notably, promethazine's administration using the intravenous route (especially when abused) could result in severe tissue injuries such as gangrene, thrombophlebitis, and burning. Consequently, the preferred method of administering this drug is deep intramuscular injection. Moreover, an IV infusion could be given. Promethazine is considered as a category C drug in pregnancy; hence, it ought to be cautiously used in the cases where its benefits would outweigh its risks in the patient.
CHLORPHENIRAMINE MALEATE (CPM)
This drug acts by blocking the histamine receptors on the respiratory smooth muscles; hence, antagonizing their constrictor effect [45]. Furthermore, it is notable that this sedating anticholinergic antihistamine tends to work towards reducing various side effects of opiates and potentiating of analgesia. Essentially, this medication is strictly administered after the administration of opiates. The aforementioned drug helps as a nasal decongestant, as well as in the relief of various symptoms of allergy or opiate withdrawal including running nose, sneezing, watery eyes, rash, cough, as well as itchiness exhibited in the eyes, throat, nose, and skin [46]. The dosage involves 10ml orally every 4-6hours, which does not surpass 60ml in a period of 24hours. This drug is available over the counter. Various neuropsychiatric effects of the agent include drowsiness, blurred vision, dizziness, confusion, disorientation, insomnia, sedation, as well as euphoria. Furthermore, the drug enters breast milk and is consequently contraindicated during the period of breastfeeding. After analyzing several sources about opiate abuse, it was noted that CPM was also associated with neonatal abstinent syndrome.
CYCLIZINE
This drug is an H? receptor antagonist, which works by blocking the H? histamine receptor. It is notable that as any of the others sedating anticholinergic antihistamines, it tends to work towards reducing various side effects of opiates and potentiating of analgesia. The drug is applicable in the treatment, as well as prevention of motion sickness, nausea, vomiting, as well as vertigo [47]. The drug's recommended dose is 50 mg orally that should be administered thrice a day. Research postulates that the onset of action of the drug is approximated at 2 hours with a four-hour duration of action in a patient. The drug ought not to be administered concurrently with other sedatives, anticholinergics, or tranquilizers. Cyclizine is classified under category B in pregnancy. Moreover, after analyzing several sources about opiate abuse, it was noted that cyclizine is also associated with neonatal abstinent syndrome.
DOXYLAMINE SUCCINATE
According to several sources, this agent is commonly used and works to enhance the effects of opiates effectively. Further, the drug is an antihistamine used in the prevention of both nausea and vomiting during pregnancy for the women who fail to respond to conservative management. Besides, it is also essential in relieving various withdrawal symptoms. The mechanism of the prevention of morning sickness and drowsiness is not yet known [48]. The drug’s dose is 10 mg for Diclegis and 20 mg for Bonjesta. Doxylamine exhibits various side effects including dry mouth, blurred vision, headache, somnolence, vertigo, fatigue, malaise, anxiety, hypersensitivity, urinary retention, as well as insomnia. These effects are exacerbated by concomitant use of doxylamine and other sedatives. Research also indicates that women should avoid breastfeeding while undergoing therapy with this agent. Table 7 provides additional details regarding co-administration of opiates with antihistamines.
|
Opiate of Potentiation |
Medication Used as Potentiation |
Mechanism of Action and Dosages |
Effects |
|
Morphine, fentanyl and nalbuphine |
Cimetidine
|
Cimetidine functions by inhibiting the cytochrome P-450 enzymes, which function in the metabolism of opioids, as well as other drugs. Hence, it increases the duration of action of the opioids causing an increased euphoric state. Dosage is 800mg. |
Risk of acute liver injury resulting from the heavy workload for the same, headache, dizziness, gynecomastia, as well as somnolence. |
|
Morphine |
Diphenhydramine
|
The drug inhibits histamine and also increasing the analgesic, as well as the mood-altering properties associated with opiates to an unusually small degree. The drug’s dosage is 25mg-50mg orally, which is given every 6 to 8 hours.
|
Dry mouth, dizziness, abdominal pain, drowsiness, constipation, confusion, restlessness, irregular heartbeat, difficulty in passing urine, blurred vision, euphoria, as well as palpitation |
|
Morphine |
Promethazine |
Works towards reducing various side effects of opiates and potentiation of analgesia. Dose is 25mg orally in a frequency of every 4-6hours and with an onset of 3-5minutes when given via intravenous route and 20 minutes when given orally. |
Dry mouth, constipation, drowsiness, blurred vision, sedation, confusion, disorientation, euphoria, extrapyramidal symptoms, irregular heartbeat, urinary retention as well as catatonic states. |
|
Opium |
Chlorpheniramine maleate (CPM)
|
Acts by blocking the histamine receptors on the respiratory smooth muscles; hence, antagonizing their constrictor effect. The dosage involves 10ml orally every 4-6hours, which does not surpass 60ml in a period of 24hours.
|
Dry mouth, constipation, drowsiness, blurred vision, dizziness, confusion, disorientation, euphoria, extrapyramidal symptoms, irregular heartbeat, hypotension, insomnia, sedation, whizzing, thickening of bronchiole secretions, as well as euphoria. |
|
Methadone |
Cyclizine Droperidol
|
Works by blocking the H1 histamine receptor. The drug's recommended dose is 50mg orally that should be administered thrice daily. |
Drowsiness, xerostomia, headache, nausea, dermatitis, urinary retention, as well as diplopia. |
|
Methadone |
Doxylamine Succinate
|
Works to enhance the effects of opiates . Further, the drug is an antihistamine used in the prevention of both nausea, as well as vomiting during pregnancy for the women who fail to respond to conservative management.The drug’s dosage is 10mg for Diclegis and 20mg for Bonjesta.
|
Dry mouth, blurred vision, abdominal pain, headache, somnolence, vertigo, fatigue, malaise, anxiety, hypersensitivity, urinary retention, as well as insomnia. |
Table 7: Antihistamines and opiates potentiation.
ANTIPSYCHOTICS
In total, 11 atypical antipsychotic (AA) medications were approved by the Food and Drug Administration since the drug class was introduced in the 1970s [49]. However, in the recent past, there was an increase in reports indicating the off-label use of the drugs outside areas approved by FDA. Quetiapine, a type of AA, was identified as the most abused of the class [50]. Other than areas approved by FDA and health professionals, abusers are using the drugs in enhancing the effects of illicit substances such as marijuana or controlling the adverse effects of the substances. The goal of abusers is to self-medicate the withdrawal symptoms or using it in combination with other drugs against the directions of FDA. For instance, in 2008 when the first case of abusing quetiapine was reported, a 48-year-old man who had been addicted to alcohol was prescribed with a daily dose of 100 mg/d, but increased it to 1,000 mg/d to deal with withdrawal symptoms. Basically, the drug is misused and abused by taking it in excessive levels, and for purposes for which it is not meant. The drug has a special group of individuals that abuse it and in special places. According to, reports published between 1966 to 2012 indicated males in their 30s form the largest number of individuals abusing the drugs. Further individuals with a history of misusing anxiolytics, hypnotics, and sedatives were eight times more likely to misuse and abuse AAs . Most of the users are prisoners, who were jailed as a result of substance abuse; and therefore, they seek ways to deal with the withdrawal symptoms they were facing after being stopped from drugs. According to, prisoners abuse AAs to have uninterrupted sleep. In a study conducted to determine the effects ofolanzapine on morphine-induced emesis and dysregulation of sleep associated with chronic pain; it was established that the AAs decreased morphine-induced nausea and vomiting, and completely alleviated the disturbance of sleep induced by sciatic ligation of nerves [51]. They have difficulty sleeping as a result of mental illnesses they have such as anxiety, and once they find out that taking extra doses of AAs helps them to relaxn, they increase its demand in prisons. Data collected by the Drug Abuse Warning Network (DAWN) between 2005 to 2011 indicated that nationally the number of individuals making emergency department (ED) visits as a result of abusing quetiapine in the US increased by 90%. With the statistics on the upward trend every other year, concerns over misuse and abuse of AAs grew further making it necessary to put in place deterrent efforts to control the situation. Though the abusers of AAs are doing it in hideouts, misuse and abuse of the drugs are revealed through different means. First, the drugs released to the market are not reaching the intended users as a result of most of the drugs ending up in the hands of abusers. According to the quetiapine black market in the US grew with 25 mg tablets of the drug selling for 3-8 dollars. On the other hand, it was also established that the drug assumed different names while on the streets to avoid the attention of security agencies. All these are indications of a drug being misused. The abuser takes the drug in different forms. For instance, the drugs could be taken as a tablet, an injection after dissolving the tablet and also smoked by others after combining the drug with other substances. It works faster when taken as an injection as a result of directly entering the blood system leading to immediate effect. Misuse and abuse of the drugs lead to harmful effects that are posing a threat to the health of abusers. According to [52], misuse and abuse of AAs lead to dry mouth, abdominal pain, constipation, dizziness, asthenia, and stomach upset. Further, the drugs when used in excess lead to elevated transaminase levels, rapid weight gain and diabetes [53]. Though rarely reported, in extreme cases, abuse of AAs leads to death as a result of overdose or wrong combination with other drugs. Clozapine and olanzapine were found to have the most extreme side effects while lurasidone, aripiprazole and ziprasidone had low risks compared to others. These effects contribute to increasing healthcare costs that became a major concern within the US healthcare industry. Further, the abuse of these drugs was found to be addictive, which forms the basis of abusing AAs. Many side effects also led to the worries of the drug being taken off-label as a measure of control due to the rising levels of misuse. According to, there is a discussion over the unwelcome possibility of restricting access to the drugs. Though such a move would have good intentions of managing the situation that is slowly running out of control, it would serve as a serious blow to the millions of mentally ill-patients that depend on the drug in their treatment. The drugs have been found to be quite effective, when used in the right areas as guided by FDA and by prescriptions of healthcare professionals.
COMBINED USE OF OPIATES AND AAS
Many of the side effects of AAs were the result of overdose and combination with other drugs not prescribed by healthcare professionals. One of the drugs commonly combined with AAs is the opiates. The purpose for such combinations is to boost the effectiveness of opiates, which are mainly used in the treatment of medium to extreme levels of pain that may not respond well to other forms of medication. According to, approximately 75% of suicide attempt visits and misuse and abuse visits in the ED were a result of quetiapine in combination with other drugs with pharmaceuticals being the most frequently diagnosed combination. The general observation is that when combined with other drugs, the effects experienced are more severe even though the abusers carry out the combination to boost the effectiveness of the drugs. The effects of combining AAs with opiates are harmful to the health of the abusers. Combined drugs are taken in different ways. According to, the drugs are administered either intravenously or orally. When taken intravenously, the drugs act faster compared to when they are administered orally. Abusers prefer this way of administration, as it skips hepatic metabolism leading to higher doses being quickly absorbed into the bloodstream. Besides the two forms of administration, smoking was also utilized as a means through which abusers administer the combined drugs. According to, a case of a patient who preferred marijuana and crushed quetiapine tablets with marijuana was reported. This was done with the hope of experiencing a relaxed night, as abusers believe, by abusing AAs they will be able to address their mental illness. However, as established in the effects of abusing AAs, the abusers are putting their health at risk.
AAs are drugs previously not considered to have the potential of abuse but have currently been subjected to misuse and abuse. Abusers of the drugs are using it to deal with withdrawal symptoms. A majority of the abusers are found in prisons, where they use the drugs for getting relaxed sleep. In other instances, the drugs are used in combination with other drugs as a way of boosting the effectiveness of the other drugs. Whether abused on their own or in combination with other drugs, side effects include weight gain or diseases such as diabetes and addiction. In extreme cases, abuse of AAa resulted in death and is therefore generally a threat to the well being of the abusers. Increasing rates of misuse and abuse haveled to discussion of the unwelcomed move of making it difficult to access the drugs, as they are a hot product on the black market, where they are being misused. Though such a move will be good for controlling the side effects, it will be a big concern to the millions of people with mental illnesses that depend on the drug for the treatment.
STIMULANTS
Entails a review of literature with a focus on studies in which researchers assessed the effectiveness of combining morphine and amphetamine or its analog methylphenidate with the view of increasing the effectiveness of morphine in producing analgesia [54]. Authors indicate that experiments involving human and animal subjects demonstrated that combining methylphenidate or d-amphetamine with morphine results in increasing effectiveness of morphine in producing analgesia. According to, animal studies carried out in the research that they reviewed demonstrated that drugs that cause psycho-stimulation such as d-amphetamine or methylphenidate had analgesic features that were intrinsic, and this unique feature contributed to their ability to improve morphine’s analgesic capability. They also mention that studies analyzed that involved human subjects indicated that analgesia produced by opioids such as morphine was enhanced by amphetamines. Additionally, amphetamines not only lowered somnolence but also increased individual’s cognitive ability, and this was a basis for their use in attaining improved analgesia with opioids. When cognitive ability and alertness of a patient increases, the dose of an opioid used to produce analgesia could be increased. This provides space for increasing analgesia without fear of cognitive effects of the opioid, and this could only be achieved through the combination therapy as reported by [55] and in the literature review. The article indicates that the combination of an opioid analgesic and either methylphenidate or d-amphetamine could be used to enhance analgesia and cognition of patients with the goal of improving their quality of life, especially when the patients were going through debilitating pain that prevented them from living with the highest possible quality of life. The second study is by [56]. According to these researchers, addicts to various substances of abuse and recreational drug users often combined a wide variety of drugs with the goal of attaining the highest possible level of “high.” The term “speedball” was used to refer to a combination of an opiate and a stimulant of the psychomotor system with the intention of generating effects that were greater. However, the use of this combination resulted in multiple deaths [57]. In the history of combining an opiate with a psycho-stimulant, heroin and cocaine were the most widely used combination according to [58] and as reported. However, methamphetamine became increasingly popular over the past few years, and as such was used in combination with opiates and other substances of abuse. Despite the rapid increase in the latter combination, research was limited in this field, and as such, the authors carried out this study to establish the interaction between opiates and methamphetamine. They assessed interactions that are behavioral in nature between morphine and methamphetamine, while utilizing a wide range of dosages for each of these drugs. Rats were used as the animal subjects for testing these interactions. The researchers found out that a combination of morphine and methamphetamine generated behavior stimulation that was significantly higher when a comparison was made in the use of each of these drugs separately. However, they discovered that the extent of interaction between morphine and methamphetamine depended on dosages of each drug used, and the type of behavior that was assessed. They concluded that combining these drugs resulted in interactions, in behavior, and in a manner that was complex. They also indicated that combining morphine, opiates and methamphetamine resulted in greater effects than when each of these drugs was used separately. The third study that is the focus of this paper was carried out by [59]. The study aim was to evaluate the prevalence and trends of concurrently using opioids and stimulants by the adults that were suffering from attention-deficit hyperactivity disorder (ADHD). Researchers also intended to evaluate the factors that characterized the long-term use of this combination among these patients. They carried out a cross-sectional study in which data was extracted for the period between the years 1999 and the year 2010 from 29 different states within the United States of America. They used multivariable models of regression in determining trends regarding the prevalence of long-term use of a combination of an opioid and a stimulant, and risk factors that led to such concurrent use. They found there was an increase in the concurrent use of an opioid and a stimulant in the long-term period among adults who had ADHD. They also found out that the predisposing factors that led to the concurrent use of this combination, other than being 40 years or older, was being a non-Hispanic white, being a resident of the southern and northeast regions of the United States of America, and being diagnosed with substance abuse disorder. Those who were depressed had an anxiety disorder, pain that was chronic, chronic obstructive pulmonary disease, and cardiovascular conditions had the highest likelihood of using this combination. The researchers concluded that concurrent utilization of an opiate and a stimulant combination in the long-term period among adults who were suffering from ADHD, was a common occurrence. As such, co-administration of opioids and stimulants in the long-term management of pain associated with ADHD would require prioritizing the comprehension of risks associated with this process. The final article under consideration is an opinion paper on the need to adopt concurrent use of opioids and stimulants in relieving pain while preventing complications associated with opioid use by [60]. The author begins by indicating that morphine was historically used with great success together with dextroamphetamine to achieve greater levels of pain relief by one and a half and two times the pain-relieving effect that would normally be achieved when morphine was used alone. Other than the use of dextroamphetamine, the article points out that cocaine and morphine were successfully used to manage pain in patients who were in agony for illnesses that were at the advanced stages as per the book by [61]. Indicated that opioids had numerous side effects that included constipation, fatigue, sedation, suppression of hormones,falls, and mental exhaustion. Those required lowering of the dosage of opioids used while attaining pain relief. In order to do this, there was need to add a stimulant that would ensure sufficient pain relief to the patient while lowering the dose of opioids, so that they could experience as few as possible of these side effects. The question,is how this is achieved in the body. In this article, is indicated that numerous neurochemical pathways were involved in relieving pain. These pathways included the adrenergic system, gamma-aminobutyric acid (GABA-nergic) pathway, and the serotonergic pathway as quoted from [62]. Stimulants worked by targeting the adrenergic system with a focus on noradrenaline, while morphine and other opioids targeted the endorphin receptors. The combination of these two modalities of action ensured that a greater impact was attained in relieving pain than when each of these drugs was used alone. Table 8 describes various effects and findings due to simultaneous use of opiates and stimulants.
|
Medications or substances |
Doses, pattern of use |
Effects described |
|
Morphine |
It exists in numerous forms. These include tablets that are meant for immediate release. For patients who are opioid-naïve, it is used to manage pain, and this tablet is given in a dose of between 15 milligrams and 30 milligrams every four hours for as long as the healthcare professional deems necessary. These patients could also be given oral solution or a suppository of morphine that has either 10 milligrams or 20 mg of the drug. They could also be injected with the drug through various routes for acute pain. According to the reviewed articles, intravenous morphine could be given for acute pain, when an antagonist is available. In this case, the dosing is between 2mg and 10mg for an adult weighing 70 kg in body weight. Such acute pain includes post-operative severe pain. Additionally, morphine is used to manage pain that is chronic. According to the reviewed articles, this pain could either be related to cancer or other chronic illnesses that are non-cancer related. |
Morphine acts through endorphin receptors, where it blocks the ascending pathways of pain and thus alters the natural response that the body has towards pain. In this process, it generates analgesia. However, it also produces depression of the respiratory system, significant sedation, constipation, fatigue, dullness of mental activity, hormonal suppression and falls [63-69]. |
|
Methamphetamine |
This is available in the strength of 5 milligrams administered in tablet form orally at an initial dose of 5mg every 24 hours or in divided doses for the treatment of attention-deficit hyperactivity disorder (ADHD). It is used in combination with opioids like morphine to produce marked analgesia, as indicated in the reviewed articles. |
It directly stimulates the central nervous system by causing dopamine release and the release of other catecholamines. Therefore, it augments analgesia produced by morphine or other opioids. |
Table 8: Simultaneous use of opiates and stimulants.
The use of opioids brought about fears regarding their potential to increase the likelihood of the user becoming dependent or addicted to the opioid. This led to studies being conducted on the combination of other forms of central nervous stimulants and opiates like morphine. The aim of these studies was to demonstrate that such combinations help in attaining sufficient pain relief with minimal effective dose of opiates likes morphine. This not only lowers the side effects experienced when high doses of opiates are used, but also reduces the likelihood of addiction. Other than these, the long-term effect and likelihood of use of such combinations should be assessed for its benefits and risks for either ADHD or pain.
PHARMACOKINETIC ENHANCERS
Pharmacokinetic enhancers are used to boost the effectiveness of another drug. When the two drugs are given together, the pharmacokinetic enhancer interferes with the metabolism of the other drug which allows the targeted drug to remain in the body longer and at a higher concentration. While this principle has clinical utility in many settings, it could be abused to potentiate the effects of opioids. Below is a list of some common pharmacokinetic enhancers that could be utilized as opioid potentiators.
HIV MEDICATIONS
Many HIV medications are strong inhibitors of the cytochrome P450 enzymes, specifically CYP3A4. This inhibition of CYP3A4 leads to a subsequent increase in the serum concentration of the opioid due to its decreased metabolism. Ritonavir, atazanavir (ATV), indinavir and cobicistat are a few examples of these drugs. Some authors describe HIV drugs as potentiators of opiate drugs. Medications or substances described in the articles include buprenorphine, atazanavir, methadone and antiretroviral medications [70-73]. The majority of these articles discuss pharmacokinetic interactions between antiretroviral drugs and opiates. For instance, in their article, [74] examined drug interactions between buprenorphine and the protease inhibitors atazanavir and ritonavir. According to the authors, HIV drugs have an effect on treating opioid dependence. Their study concluded that ATV administration for the treatment of HIV disease in opioid-dependent patients produced a pharmacokinetic interaction characterized by increased metabolite and buprenorphine concentrations. Also, McCance-Katzsummarizes the present information regarding interactions between methadone and antiretroviral medications. According to the article , buprenorphine has a significant pharmacokinetic interaction with antiretrovirals although not a pharmacodynamic relationship; hence, simultaneous use of such drugs is unable to treat opioid-dependence in HIV-infected patients. Studies referred to in these articles demonstrated that HIV drugs had positive effects, when used simultaneously with opiates. Also, the articles indicated that opiates were highly recommended to HIV-infected people since most of them are likely to have chronic pain. According to the article by, HIV-infected persons were recommended to use opioid doses below the 50-90 morphine milligram equivalents (MMEs) range to be effective and avoid the risk of overdose, which could adversely affect the patient. Drugs like methadone are said to be opioid agonists and are known to prevent opiate dependence. HIV drugs have a pharmacological effect of reducing opiate addiction. Also, when HIV drugs are used simultaneously with opiates, they have a positive effect on the HIV treatment outcomes. It is clear from the articles that simultaneous use of opiates with HIV drugs help in reducing opioid dependence.
ANTIFUNGAL AGENTS
Certain antifungal agents such as itraconazole, ketoconazole and posaconazole are strong inhibitors of cytochrome P450 enzymes. This inhibition leads to a subsequent increase in the serum concentration of the opioid due to its decreased metabolism. Medications described in the literature include: fluconazole (FLC), itraconazole, voriconazole, ketoconazole, terbinafine, tolnaftate, caspofungin, micafungin, anidulafungin, nystatin and amphotericin B, and methadone. The articles indicate that most antifungals like itraconazole are available in the form of capsules, intravenous and oral solutions. Also, fluconazole is available in capsules, tablets, powdered, or injectable forms that consist of 2 mg of fluconazole and 9 mg of sodium chloride diluents. They are used for several days ; for instance, a person takes one tablet three times a day. One of the articles discussing the pharmacological effects of antifungals on potentiating opiatesis. According to the article, interaction of antifungals with opiates increases blood concentration of the opioids, thus, increasing their effectiveness. According to [75], antifungal drugs like azoles have inhibitory effects. Interaction between antifungal drugs and opiates could change the pharmacokinetics of the drugs. Azole agents block the in vitro metabolism of methadone (opioid). This initiates a longer presence of opiates in the body thus increasing its effectiveness. Antifungal medications have the pharmacological effect of potentiating opiates. Also, the article, “What Is Opiate Potentiation” mentions the effects of antifungals on opiate potentiation. However, there is a significant lack of information regarding the use of these medications in opiate potentiating. Antifungal drugs have an effect of potentiating opiates by preventing opiate tolerance, as described in the article. According to their article, if antifungal drugs are used simultaneously with opioids after a patient develops drug dependence of opiates, a potentiating effect is created, thus making the opiates become more effective [76]. The article, “Ketoconazole increases cocaine and opioid use in methadone-maintained patients” [77] discusses simultaneous use of antifungal drug (ketoconazole) and opiate (methadone). The authors referred to a study to determine whether use of ketoconazole increased opioid and cocaine use in methadone-maintained patients. Findings of the study indicated the increased use of opioids on humans who used methadone when subjected to chronic ketoconazole treatment. The side effects of voriconazole included reversible disturbances in vision such as a blurred vision, bright spots or altered color discrimination [78]. Ketoconazole led to a lower incidence of failure to achieve complete resolution than was observed with placebo, but the results were statistically heterogeneous and could not be explained by subgroup analyses of dose, mode of delivery or conflict of interest [79].
ANTIBIOTICS
Certain antibiotics such as clarithromycin are strong inhibitors of cytochrome P450 enzymes. This inhibition leads to a subsequent increase in the serum concentration of the opioid due to its decreased metabolism. These articles discuss interactions that opioids have with antibiotic drugs. For instance,“Non-analgesic effects of opioids: interactions between opioids and other drugs [80] describes interaction that opioids have with other drugs. According to the article, there are various antibiotics that have interactions with opioids. From the article, it is clear that, when antibiotics are used with opioids, they increase effectiveness of the opioid drugs, as well as treating the opioid-related adverse effects. The article clarifies that almost all antibiotics, when combined with opioids, have a potentiating effect. The article gives an example of warfarin, which could be potentiated with antibiotics by inhibiting intestinal flora that produces vitamin K. Also, when warfarin is combined with antibiotics, hepatic metabolism of warfarin is inhibited, thus, increasing its effectiveness. Interaction of opioid analgesics and other drugs results in pharmacological effects that rely on the interacting agents, and the mode of administration. The study presented confirms that antibiotics are often combined with opioids in patients undergoing surgical procedures to increase the strength of the opioids in relieving pain. An example is erythromycin, which increases the effect of opioids when combined with the drugs. The review by [81] reveals that antibiotics have no early effect on pain, and a slight effect on pain in the days following.There was only a modest effect with a number of children with tympanic perforations, contralateral otitis episodes and abnormal tympanometry findings at two to four weeks and at six to eight weeks, compared with placebo in children with acute otitis media (AOM). However, when combined with opioids, the rate at which they relieve pain increases. This clearly indicates that antibiotics have a potentiating effect on opioids. Furthermore, supports opiate potentiation by the antibiotics. According to the article, antibiotics inhibit CYP34A thus having a similar mechanism of action as antifungals, which produces similar potentiation effects. In addition, when opiates are combined with antibiotics the intended method of metabolism is altered thus increasing the ‘high’, as well as the potency of the drug. As a result, the effect of the opioid drug increases. However, when used simultaneously, there appears to be an adverse reaction such as an overdose; and it could result in drug abuse, which could further be considered hazardous. Therefore, patients should only use the two drugs simultaneously following a doctor’s prescription to avoid possible risk factors for both medicines.Table 9 summarizes existing findings on simultaneous use of these medications.
|
Group of Drugs (substances) |
Medications or substances described in literature |
Doses and pattern of use |
Effects mentioned in literature
|
|||
|
HIV Medications |
Buprenorphine, atazanavir (ATV), methadone, raltegravir and antiretroviral medications |
Doses or pattern of use of these medications differ depending on the drug itself. For example, atazanavir is not orally taken, instead the patient is injected with it two times a day. Raltegravir is administered two times a day. Nonetheless, the number of drugs taken by HIV patients was reduced from about 20 tablets a day in 1996 to one tablet in 2006. The recommended dose for buprenorphine and methadone should be below the 50-90 morphine milligram equivalents (MMEs) range. |
ATV administration for the treatment of HIV disease in opioid-depended produces a pharmacokinetic interaction characterized by increased metabolite and buprenorphine concentrations. Drugs like methadone are known to beopioid agonists and to prevent opiates dependence. |
|||
|
Antifungals |
Medications or substances described in articles: fluconazole (FLC), itraconazole, voriconazole, ketoconazole, terbinafine, tolnaftate, caspofungin, micafungin, anidulafungin, nystatin and amphotericin B, and methadone. |
Most antifungals like itraconazole are available in the form of capsules, intravenous and oral solutions. Also, fluconazole is available in capsules, tablet, powdered or injectable forms that consist of 2 mg of fluconazole and 9 mg of sodium chloride diluent. They are used for several days, for instance; a person takes one tablet three times a day.
|
Interaction of antifungals with opiates increases blood concentration of the opioid, thus increasing their effectiveness. Antifungal drugs like azoles have an inhibitory effect. Also, interaction between antifungal drugs and opiates could change the pharmacokinetics of the drugs. Lastly, when antifungal drugs are used with opiates, they could block an enzyme (CYP3A4) from assisting to metabolize the opioid drug; therefore, increasing the amount of the drug in the blood . |
|||
|
Antibiotics |
Medications or substances described in articles include codeine, oxycodone, erythromycin, phenytoin, carbamazepine, ciprofloxacin, aspirin and barbiturates.
|
Erythromycin; Ciprofloxacin in various doses |
Erythromycin increases and rifampicin decreases the effects of opioids, while cimetidine could enhance the effects of opioids by increasing their duration of action. Also, carbamazepine, phenytoin and barbiturates could enhance the metabolism of opioids that rely on hepatic metabolism . Ciprofloxacin increases the effect of warfarin. Also, ciprofloxacin inhibits warfarin's metabolism ; may alternate opiate metabolism [82] |
|||
Table 9: Pharmacokinetic enhancers and opiates potentiation.
OTHER SUBSTANCES
According to different studies and patient reports about the effects of tropicamide, these are remarkable in claiming the increased efficacy of heroin along with decreasing and delayed withdrawal symptoms. For example, Spangolo et al. describe a 22-year-old heroin-addicted woman having a 2-year history of IV injection with an eye drop solution containing 1% tropicamide, which is an antimuscarinic agent. She reported injecting tropicamide, because it attenuated symptoms and signs of opiate withdrawal, and also had hallucinogenic and euphorigenic effects. Acute tropicamide intoxication could lead to anticholinergic syndrome, hyperthermia, tremors and convulsions. Chronic tropicamide-related problems include cardiovascular toxicity, psychosis, renal or liver failure, severe weight loss and infections [83].
Sharma et al. developed a research study showing that inhibition of COX-1 enzyme potentiates opioid-induced antinociception in an animal model of central nociception with a chief objective of exploring the antinociceptive pharmacodynamic effects of opioids, when combined with non-steroidal anti-inflammatory drugs (NSAIDs),and when central nociception is taking place. The study reported that co-administration of naproxen (non-selective COX inhibitor) with opioids potentiated the antinociceptive effect. It appears that inhibition of COX-1, rather than COX-2, plays an important role in synergistic antinociceptive effects of combination therapy. This particular study was selected because it introduces the same idea as that is used by the patients abusing the opioids. The main limitation of the study was in the fact that it was conducted on animals, rather than on human beings [84].
CONCLUSION AND RECOMMENDATIONS
The main reason why people are potentiating opiates is to make the drugs effect more powerful. Additionally, the literature overview indicates that drug providers and other professionals, who are allowed to prescribe opiates, provide most of the potentiating drugs to users. Use of opioid enhancers represents a serious health risk. More large-scale studies are needed to be carried out to confirm and better describe the extent of opiate enhancer misuse in the USA and elsewhere. Specific prevention programs should be implemented for the general population, as well as for the high-risk population of poly drug abusers. Health and other professionals should be rapidly informed about this new and alarming trend in misuse. As a part of the recommendation, it is essential for the American healthcare systems to review, the way these potentiating drugs should be prescribed to patients, especially to those below the age of 18 years.
REFERENCES
- Chevy C (2011) American Society of Addiction Medicine, Public Policy Statement: Definition of Addiction.
- Alpha O (2008) The Ultimate Opiate Potentiation Thread.
- National Institute on Drug Abuse (2018).
- Bethesda MD (2015) National Institute on Drug Abuse. Drugs of Abuse: Opioids.
- McCabe SE, Morales M, Cranford JA, Delva J, McPherson MD et al., (2007) Race/ethnicity and gender differences in drug use and abuse among college Students. J Ethn Subst Abuse 6: 75-95.
- SAMHSA (2018) Age and gender-based populations. SAMHSA - Substance Abuse and Mental Health Services Administration.
- Watkins LR, Kinscheck IB, Mayer DJ (1984) Potentiation of opiate analgesia and apparent reversal of morphine tolerance by proglumide. Science 224: 395 -396.
- Walters J (2017) America's opioid crisis: how prescription drugs sparked a national trauma.
- Lynch ME (2005) A review of the use of methadone for the treatment of chronic noncancer pain. Pain Res Manage 10: 133-144.
- Mattioli L, Perfumi M (2011) Evaluation of Rhodiolarosea L. extract on affective and physical signs of nicotine withdrawal in mice. Journal of Psychopharmacology 25: 402 -410.
- Caputi FF, Acquas E, Kasture S, Ruiu S, Candeletti S et al., (2018) The standardized With aniasomnifera Dunal root extract alters basal and morphine-induced opioid receptor gene expression changes in neuroblastoma cells. BMC complementary and alternative medicine 18: 9.
- Adnan LH, Bakar NH, Mohamad N (2014) Opioid dependence and substitution therapy: thymoquinone as potential novel supplement therapy for better outcome for methadone maintenance therapy substitution therapy. Iran J Basic Med Sci 17: 926 -928.
- Singh A, Granley K, Misrha U, Naeem K, White T, et al., (1992) Screening and confirmation of drugs in urine: Interference of hordenine with the immunoassays and thin layer chromatography methods. Forensic Science International 54: 9-22.
- Heidari MR, Mehrabani M, Pardakhty A (2007) The analgesic effect of Tribulusterrestris extract and comparison of gastric ulcerogenicity of the extract with indomethacin in animal experiments. Ann NY Acad Sci 1095: 418-427.
- Russel Al, McCarthy MF (2000) DL-phenylalanine markedly potentiates opiate analgesia - an example of nutrient/pharmaceutical up-regulation of the endogenous analgesia system. Med Hypotheses 55: 283 -288.
- Hu X, Huang F, Szymusiak M, Liu Y, Wang ZJ, et al., (2015) Curcumin Attenuates Opioid Tolerance and Dependence by Inhibiting Ca2+/Calmodulin-Dependent Protein Kinase II α J Pharmacol Exp Ther 352: 420 -428.
- Moradi S, Charkhpour M, Ghavimi H, Motahari R, Ghaderi M, et al., (2013) Gap junction blockers: a potential approach to attenuate morphine withdrawal symptoms. Journal of Biomedical Science 20: 77.
- Elkins C (2018) What Is Opiate Potentiation.
- Peckham AM, Fairman KA, Sclar DA (2018) All-cause and drug-related medical events associated with overuse of gabapentin and/or opioid medications: a retrospective cohort analysis of a commercially insured US population. Drug Saf 41: 213 -228.
- Moody DE, Fu Y, Fang WB (2018) Inhibition of In vitro Metabolism of Opioids by Skeletal Muscle Relaxants. Basic Clin Pharmacol Toxicol 123: 327-334.
- Meir R, Totsch S (2017) Baclofen and opioid synergism in mice: new insights and potential treatments. The Journal of Pain 8: 26.
- Bujalska Zadro?ny M, Tatarkiewicz J, Kulik K, Filip M (2017) Magnesium enhances opioid-induced analgesia - What we have learnt in the past decades. Eur J Pharm Sci 99: 113-127.
- Banerjee S, Jones S (2017) Magnesium as an Alternative or Adjunct to Opioids for Migraine and Chronic Pain: A Review of the Clinical Effectiveness and Guidelines. Ottawa, ON: Canadian Agency for Drugs and Technologies in Health.
- Moody DE, Fu Y, Fang WB (2018) Inhibition of In Vitro Metabolism of Opioids by Skeletal Muscle Relaxants. Basic Clin Pharmacol Toxicol 123: 327-334.
- Begon S, Pickering G, Eschalier A, Dubraya C (2002) Magnesium Increases Morphine Analgesic Effect in Different Experimental Models of Pain. Anesthesiology 96: 627-632.
- Bujalska Zadro?ny M, Kulik K, Ordak M, Sasinowska Motyl M, G?si?ska E, et al., (2016) Micronized Organic Magnesium Salts Enhance Opioid Analgesia in Rats. PLoS ONE 11: e0161776.
- Horsfall JT, SpragueJ (2017) The Pharmacology and Toxicology of the ‘Holy Trinity’. Basic Clin Pharmacol Toxicol 120: 115-119.
- Venturini MA, Zappa S (2015) Magnesium-oral supplementation to reduce pain in patients with severe peripheral arterial occlusive disease: the MAG-PAPER randomised clinical trial protocol. BMJ Open 5:
- Boothby LA, Doering PL, Hatton AR (2003) Serious Abuse Potential. Hospital Pharmacy 38: 337 -345.
- Dart R C, Surratt HL, Cicero TJ, Parrino MW, Severtson SG (2015) Trends in opioid analgesic abuse and mortality in the United States. New England Journal of Medicine 372: 241-248.
- Gonzalez LA, Gatch MB (2009) Abuse Potential of Soma®: the GABAA Receptor as a Target. Mol Cell Pharmacol 1: 180-186.
- Alan FS. Charles DB (2015) Manual of clinical psychopharmacology. American Psychiatric Pub.
- Zhang L, Hammond DL (2010) Cellular basis for opioid potentiation in the rostral ventromedial medulla of rats with persistent inflammatory nociception. PAIN 149: 107-116.
- Bellville JW, Forrest WH, Shroff P, Brown BW (1971) The hypnotic effects of codeine and secobarbital and their interaction in man. Clinical Pharmacology & Therapeutics 12: 607-612.
- Maurer PM, Bartkowski RR (1993) Drug interactions of clinical significance with opioid analgesics. Drug Saf 8: 30-48.
- Pechnick RN, Terman GW (1987) The role of opiate receptors in the potentiation of pentobarbital sleeping time by the acute and chronic administration of opiates. Neuropharmacology 26: 1589-1593.
- Eisenberg RM (1982) Short-term tolerance to morphine: effects of diazepam, phenobarbital, and amphetamine. Life sciences 30: 1615-1623.
- Forbes JA, Jones KF, Smith WK, Gongloff CM (1986) Analgesic Effect of an Aspirin?codeine?butalbital?caffeine Combination and an Acetaminophen?codeine Combination in Postoperative Oral Surgery Pain. Pharmacotherapy 6: 240-247.
- Hough L, Nalwalk J, Chen Y, Schuller A, Zhu Y, et al., (2000) Improgan, a cimetidine analog, induces morphine-like antinociception in opioid receptor-knockout mice. Brain Res 880: 102-108.
- Bluhm R, Zsigmond E, Winnie A (2016) Potentiation of opioid analgesia by H1 and H2 antagonists. Life Sci 31: 1229-1232.
- Halcomb S, Sivilotti M, Goklaney A, Mullins M (2005) Pharmacokinetic Effects of Diphenhydramine or Oxycodone in Simulated Acetaminophen Overdose. Acad Emerg Med 12: 169-172.
- CarrK, Hiller J, Simon E (1985) Diphenhydramine potentiates narcotic but not endogenous opioid analgesia. Neuropeptides 5: 411-414.
- Lynch K, Shapiro B, Coffa D, Novak S (2015) Promethazine use among chronic pain patients. Drug Alcohol Depend 150: 92-97.
- Shapiro B, Lynch K, Toochinda T, Lutnick A, Cheng H, et al., (2013) Promethazine Misuse Among Methadone Maintenance Patients and Community-Based Injection Drug Users. J Addict Med 7: 96-101.
- Tamadonfard E, Mojtahedein A (2015) Effect of Chlorpheniramine On Formalin-Induced Pain in Mice: Its Relation with Opioid System. PlantaMedica 76.
- Togashi Y, Umeuchi H, Okano K, Ando N, Yoshizawa Y, et al., (2012) Antipruritic activity of the κ-opioid receptor agonist, TRK-820. Eur J Pharmacol 435: 259-264.
- Laffey J, Boylan J (2014) Cyclizine and droperidol have comparable efficacy and side effects during patient-controlled analgesia. Irish Journal of Medical Science 171: 141-144.
- Carrascosa M, Caviedes J, Lucena M, Cuadrado Lavin A (2012) Syndrome of inappropriate antidiuresis in doxylamine overdose. BMJ Case Rep.
- Kim S, Lee G, Kim E, Jung H, Chang J, et al., (2017) Quetiapine misuse and abuse: Is it an atypical paradigm of drug-seeking behavior. J Res Pharm Pract 6: 12-15.
- Mattson ME, Albright VA, Yoon J, Council CL (2015) Emergency department visits involving misuse and abuse of the antipsychotic quetiapine: results from the Drug Abuse Warning Network (DAWN). Subst Abuse 9: 39-46.
- Torigoe K, Mori T, Shibasaki M, Yoshizawa K, Narita M, et al., (2012) Olanzapine suppresses the rewarding and discriminative stimulus effects induced by morphine. Synapse 66: 174-179.
- Torigoe K, Nakahara K, Rahmadi M, Yoshizawa K, Horiuchi H, et al., (2012) The usefulness of olanzapine as an adjunct to opioid treatment and for the treatment of neuropathic pain. Anesthesiology 116: 159-169.
- Fischer BA, Boggs DL (2010) The role of antihistaminic effects in the misuse of quetiapine: a case report and review of the literature. Neurosci Biobehav Rev 34: 555-558.
- Dalal S, Melzack AR (1998) Potentiation of Opioid Analgesia by Psychostimulant Drugs. J Pain Symptom Manage 16: 245-253.
- Forrest WJ, Byron Wm, Brown CR (1977) Dextroamphetamine with morphine for the treatment of postoperative pain. New England Journal of Medicine 296: 712-715.
- Trujillo KA, Smith ML, Guaderrama AM (2012) Powerful Behavioral Interactions Between Methamphetamine and Morphine. Pharmacol Biochem Behav 99: 451-458.
- Ellinwood EH JR, Eibergen RD, Kilbey MM (1976) Stimulants: interaction with clinically relevant drugs. Ann N Y Acad Sci 281: 393-408.
- Leri F, Bruneau J, Stewart J (2003) Understanding polydrug use: review of heroin and cocaine co-use. Addiction 98: 7-22.
- Jenny Wei Y, Zhu Y, Liu W (2018) Prevalence of and Factors Associated With Long-term Use of Stimulants and Opioids Among Adults With Attention-Deficit/Hyperactivity Disorder. JAMA Network Open 1: e181152.
- Tennant F (2011) Simultaneous Use of Stimulants and Opioids. Practical Pain Management 11: 1-3.
- Melzak R, Mount BM (1980) The Brompton mixture versus morphine solution given orally: effects on pain. Can Med Assoc J 120: 435-438.
- Nicak A (1965) The influence of serotonine and amphetamine on analgesic effect of morphine after reserpine premedication in rats and mice. Med Pharmacol Exp Int J Exp Med 13: 43-48.
- Beaver MT (1984) Combination analgesics. Am J Med 77: 38-53.
- Dubuisson D, Dennis S (1977) The formalin test: a quantitative study of the analgesic effects of morphine, meperidine, and brain stem stimulation in rats and cats. Pain 4: 161-174.
- Goetzl F, Burrill D, Ivy A (1944) The analgesic effect of morphine alone and in combination with dextroamphetamine. Proc Soc Exper Biol Med 55: 248-250.
- Nott MW (1968) Potentiation of morphine analgesia by cocaine in mice. Eur J Pharmacol 5: 93-99.
- Sasson S, Unterwald EM, Kornetsky C (1986) Potentiation of morphine analgesia by d-amphetamine. Psychopharmacology 90: 163-165.
- Evans Wo, Bergner Dp (1964) A comparison of the analgesic potencies of morphine, pentazocine, and a mixture of methamphetamine and pentazocine in the rat. J New Drugs 4: 82-85.
- Preston KL, Jasinski DR (1986) Evaluation of mixtures of morphine and d-amphetamine for subjective and physiological effects. Drug Alcohol Depend 17:1-13.
- McCance-Katz EF (2005) Treatment of opioid dependence and co infection with HIV and hepatitis C virus in opioid-dependent patients: the importance of drug interactions between opioids and antiretroviral agents. Clin Infect Dis 41: 89-95.
- Bruce RD, McCance-Katz E, Kharasch ED, Moody DE, Morse GD, et al., (2006) Pharmacokinetic interactions between buprenorphine and antiretroviral medications. Clinical Infectious Diseases 43: 216-223.
- Neuman MG, Monteiro M, Rehm J (2006) Drug interactions between psychoactive substances and antiretroviral therapy in individuals infected with human immunodeficiency and hepatitis viruses. Substance use misuse 41: 1395-1463.
- Cunningham CO (2018) Opioids and HIV infection: from pain management to addiction treatment. Top Antivir Med 25: 143.
- McCance-Katz EF, Moody DE, Morse GD, Ma Q, DiFrancesco R, et al., (2007) Interaction between buprenorphine and atazanavir or atazanavir/ritonavir. Drug and alcohol dependence 91: 269-278.
- Moody DE, Liu F, Fang WB (2015) Azole antifungal inhibition of buprenorphine, methadone and oxycodone in vitro metabolism. Journal of Analytical Toxicology 39: 374-386.
- Oak AS, Baddley JW, ElewskiBE (2018) Systemic Antifungals. In Biologic and Systemic Agents in Dermatology Page no: 425-450.
- Kosten TR, Oliveto A, Sevarino KA, Gonsai K, Feingold A, et al., (2002) Ketoconazole increases cocaine and opioid use in methadone-maintained patients. Drug Alcohol Depend 66: 173-180.
- Peyton LR, Gallagher S, Hashemzadeh M (2015) Triazole antifungals: a review. Drugs Today (Barc) 51: 705-718.
- Okokon EO, Verbeek JH, Ruotsalainen JH, Ojo OA (2015) Topical antifungals for seborrheic dermatitis. Cochrane Database of Systematic Reviews 5.
- Heiskanen T, Kalso E (2012) Non-analgesic effects of opioids: interactions between opioids and other drugs. Curr Pharm Des 18: 6079-6089.
- Venekamp RP, Sanders SL, Glasziou PP, Del Mar CB (2015) Antibiotics for acute otitis media in children. Cochrane database of systematic reviews 6.
- Ament PW, Bertolino JG, Liszewski JL (2000) Clinically significant drug interactions. Am Fam Physician 61: 1745-1754.
- Spagnolo PA, Badiani A, Nencini P (2013) Polydrug Abuse by Intravenous Use of Heroin and Tropicamide-Containing Eyedrops. Clinical neuropharmacology 36: 100-101.
- Sharma JN, Jawad NM (2005) Adverse Effects of COX-2 Inhibitors. The Scientific World Journal 5: 629 -645.
Citation: Valeriy Z, Tregubenko P (2019) Using Over-the-Counter and Other Prescription Medications to Potentiate Opiates in the USA: Literature Review. Medical and Public Health Aspects of OTC Medication Misuse. J Alcohol Drug Depend Subst Abus 5: 012.
Copyright: © 2019 Valeriy Z, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

