
Validation of the Origami “Foldscope” Microscope as a Preliminary Screening Tool for Male Infertility
*Corresponding Author(s):
Joaquín ErrázurizDepartamento De Ginecologia Y Obstetricia, Facultad De Medicina, Clínica Alemana, Universidad Del Desarrollo, Santiago, Chile
Tel:+56 222101111,
Email:jerrazurizv@gmail.com
Abstract
Background: Male infertility is a problem that affects a large percentage of the male population worldwide. Currently, the standard method for semen quality evaluation is conventional microscopy, but this method has several disadvantages, including high costs, the need for specialized personnel, and cultural stigmas. To solve these problems, multiple devices for semen analysis at home have been developed and commercialized, including the foldable microscope, which is accessible and low-cost. The aim of this study was to evaluate the efficacy of a foldable microscope for semen analysis and compare the results obtained with those from a conventional microscope.
Methods: Semen samples collected between January and June 2022 were analyzed using a conventional microscope. Then, a video was recorded using a smartphone adapted to a Foldscope. Finally, the videos were evaluated and compared with the conventional results in a blind and random manner.
Results: Analyzing 188 semen samples in a fertility clinic demonstrated that the Foldscope is capable of effectively analyzing spermograms and providing an accurate evaluation of sample quality with an accuracy of 91%.
Conclusion: The Foldscope microscope has a strong correlation with the conventional microscope in terms of semen quality evaluation. With its accessibility and lowcost, it has significant potential to reduce waiting times and allow for early detection of male infertility.
Keywords
Foldscope; Male infertility; Semen analysis; Sperm concentration; Sperm motility
Introduction
Studies from around the world suggest that infertility affects 15% of couples of reproductive age, with approximately 40% of cases being attributed to male factors [1]. Sperm abnormalities are markers of male infertility and have been linked to conditions such as varicocele, hormonal imbalances, and genetic causes such as deletions of the Y chromosome [2]. More recent evidence has shown that sperm abnormalities can also be correlated with lifestyle and environmental effects [3]. Semen analysis is a cornerstone in the evaluation of male infertility, but men often feel embarrassed to visit urologists, and women bear the burden of infertility most of the time [4]. Furthermore, disparities in health care related to economic, cultural, social, geographic, and religious factors pose significant barriers to accessing infertility care worldwide, especially in developing countries, making it one of the most underestimated and neglected health care issues [4].
Manual microscope-based tests remain the current standard for evaluating spermograms. Over time and with technological advancements, multiple alternatives to the traditional microscope have emerged. Computer-Assisted Semen Analysis (CASA) systems or kits, which include a phone microscope and other accessories for semen sample analysis, are marketed directly to the public for home semen evaluation [5-8]. However, traditional microscopes are expensive and require highly trained technicians, while home semen analysis kits are lowcost but often inaccessible in low-resource countries [9].
The Foldscope is an optical microscope developed by Manu Prakash in 2014. It was created based on the principle of origami; it can be assembled from folded papers, making it highly flexible, portable, and lightweight (weighing only 8 grams), and is equipped with a lens capable of providing magnification from 140X to 2000X. Additionally, it does not require external power and is very low cost compared to conventional field microscopes [10]. One of its great advantages is that it can be attached to a smartphone using a magnet, allowing for capturing photos and videos that can be sent for analysis by specialists [10,11]. All these features could provide significant collaboration in the field of reproductive medicine for spermogram studies, especially in areas with limited access to care. The aim of this study was to compare the results of spermograms, specifically motility and concentration, using a Foldscope microscope and a traditional microscope.
Materials And Methods
- Study design
All men who underwent a spermogram for fertility evaluation from January to June 2022 at the Reproductive Medicine Unit of Clínica Alemana de Santiago were selected. Demographic and clinical data were collected anonymously, and fresh samples were obtained from the patients. The samples were analyzed by a single embryologist using a traditional microscope (Nikon®, Labaphot 2). An aliquot of 10 μL was prepared, and the sperm motility (A: progressive motility, B: nonprogressive motility, C: immotility) and concentration (millions/mL) were evaluated using a Neubauer chamber, with the values recorded for subsequent comparison. Then, a 1-minute video of another prepared aliquot of the semen sample was recorded using the Foldscope paper microscope attached to an iPhone 12 smartphone. Finally, a number was assigned to each sample. The data were collected weekly by the embryologist using an Excel® database to record the values of the evaluated parameters and assign a sequential number to the samples. The data were sent to a collaborator who randomized them with different codes, and the videos were provided to the embryologist for evaluation. At the end of the designated study period, the results were compared.
- Patient selection
For this study, men over 18 years old with no history of previous vasectomy who came to the Reproductive Medicine Unit for a spermogram as medically indicated for infertility evaluation were selected.
- Foldscope microscope
The Foldscope is an origami-based microscope that can be easily purchased online. It consists of a basic kit with paper cards, an enlarged spherical lens (140X), an LED flashlight with a battery for illumination, glass slides, and magnets for attaching it to a smartphone, allowing the user to capture magnified images (Photo 1). Its assembly is straight forward according to the provided instructions.
 Photo 1: Assembled Foldscope microscope.
Photo 1: Assembled Foldscope microscope.
Source: Prepared by the authors.
- Semen preparation
A total of 200 human semen samples obtained through masturbation in the sample collection room of the Reproductive Medicine Unit were included in the analysis. The samples were collected in a sterile container and delivered to the laboratory. After 30 minutes of liquefaction, the samples were thoroughly mixed using a pipette and transferred to a calibrated tube for volume determination. An aliquot of 10 μL was placed on a glass slide with a 22x22 mm cover slip for subsequent evaluation. The evaluation was performed under a conventional microscope using a 40x objective. Then, a video was taken and an evaluation was performed using the Foldscope microscope. The distance between the Foldscope's single-ball lens and the smartphone camera was 2.7 mm.
- Semen analysis
Semen analysis was conducted according to the standards of the World Health Organization's Laboratory Manual for the Examination and Processing of Human Semen (5th edition, 2010). The lower reference limit for sperm concentration was used as defined in the manual, which sets a lower reference limit of 15 x 10^6 sperm/mL [2]. Motility was assessed according to the suggested parameters, separating motility into A: progressive motility, B: nonprogressive motility, and C: immotility. Two aliquots of 10 μL of semen were evaluated for motility using the conventional microscope, and the value was recorded if the difference between the two measurements fell within the accepted range defined by the manual [2]. Sperm count was performed using a Neubauer chamber, using a 1:20 dilution and applying the dilution factor as indicated in the manual [2]. Additionally, vitality and morphology were evaluated, but these parameters were not included in the analysis.
The measurements using the Foldscope were conducted through video recordings using a computer and a cell counter (Photo 1). The video evaluation was performed in duplicate by the embryologist, and the value was recorded if the difference between the measurements fell within the allowed ranges defined by the WHO manual. The concentration assessment was performed by calculating the sperm count per field in the video, and each sample was assigned a rating based on whether it was 15 x 10^6/ml.
The measurements from the conventional microscope and the Foldscope were grouped and compared by another collaborator of the study.
- Statistical analysis
Continuous variables were analyzed using the Shapiro-Wilk test to determine their distribution and expressed as median and interquartile range or mean ± standard deviation if the distribution was nonparametric or parametric, respectively. To standardize the results of both methods in relation to sperm concentration, the conventional microscopy and Foldscope results were stratified into three categories: azoospermia, concentration 15 million/ml.
To compare the evaluation of sperm motility between both methods, Lin's concordance correlation coefficient was used for quantitative variables. For the comparison of sperm concentration, the Kappa correlation test was used for categorical variables. Conventional microscopy was considered the gold standard for both tests. All analyses were performed and figures were generated using Stata®/SE 16.1 software (StataCorp, College Station Texas, United States).
Results
Out of the 200 recruited patients, 188were included in the final analysis. The median age of the participants was 35 (18-55) years, with a semen volume of 2.6 (1.7-3.9) mL. Table 1 describes the semen analysis parameters using both methods, including motility and sperm concentration. Table 2 describes the agreement assessment between conventional microscopy and the Foldscope for evaluating sperm motility. While the agreement for the progressive sperm category was acceptable (CCC = 0.69; r = 0.72, Figure 1), the values for the nonprogressive and immotile categories were unacceptable. The agreement for sperm count between both methods was 91%, with a kappa index of 0.66 (p < 0.0001).
|
Variable |
Conventional microscope |
Foldscope |
p Value |
|
Progressive |
48.7 ± 1.3 |
46.1 ± 1.6 |
0.2 |
|
Nonprogressive |
10.7 ± 0.3 |
13.1 ± 0.4 |
< 0.0001 |
|
Immotile |
39.1 ± 1.2 |
37.3 ± 1.6 |
0.37 |
|
Sperm concentration |
|
|
< 0.0001 |
|
Azoospermia |
3 (1.6) |
1 (0.5) |
|
|
< 15 million/mL |
23 (12.2) |
32 (17) |
|
|
> 15 million/mL |
162 (86.2) |
155 (82.5) |
|
Table 1: Comparison of traditional microscopy and foldscope for sperm count analysis.
Notes:
Continuous variables are expressed as the mean ± standard deviation.
Categorical variables are expressed as n (%).
|
Variable |
CCC |
Standard error |
Pearson coefficient |
Average difference by Bland-Altman |
Concordance limit (95%) B-A |
|
Progressive |
0.69 |
0.036 |
0.72 (<0.001) |
2.66 |
-28.33-33.65 |
|
Nonprogressive |
0.24 |
0.06 |
0.28 (<0.001) |
-2.36 |
-13.47-8.74 |
|
Immotile |
0.51 |
0.05 |
0.54 (<0.001) |
1.93 |
-36.10-39.96 |
Table 2: Correlation analysis of foldscope versus traditional microscopy
Abbreviations: B-A: Bland-Altman; CCC = Concordance Correlation Coefficient.
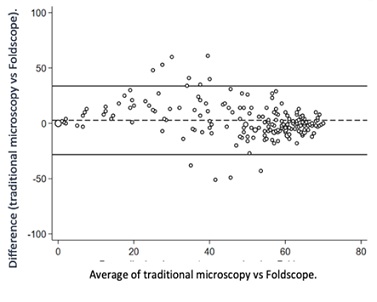 Figure 1: Bland-Altman plot for progressive sperm count with traditional microscopy and foldscope.
Figure 1: Bland-Altman plot for progressive sperm count with traditional microscopy and foldscope.
Discussion
This study demonstrated that the Foldscope microscope is an effective instrument for accurately analyzing sperm concentration and motility, providing a reliable assessment of sample quality with 91% precision. In the field of reproductive medicine, where patients seek less invasive methods, medical technology is constantly evolving. Home spermogram tests have become a trend in this area, and smartphones and internet access have great potential to support this approach. Currently, there are various options for home testing, but many of them are expensive or not available in all countries [12-14]. However, these ambulatory tests have not completely replaced laboratory analysis [15]. The Foldscope, a low-cost paper-based microscope with an approximate cost of $7, is a convenient and highly economical alternative for at-home male fertility tests compared to other home tests. Research findings indicate that the accuracy of home spermogram tests compares favorably with standard methods, with correlations ranging from 83% to 98%, which is consistent with the results obtained in the present study [6]. Although the Foldscope allows for the measurement of multiple parameters, it is important to note that it cannot evaluate other important aspects, such as sperm morphology, oxidation-reduction potential, and DNA fragmentation index. Therefore, despite the convenience and low cost of home semen tests, it remains crucial for the evaluation to be performed by a physician. Men's participation in a detailed examination can be challenging, but medical supervision is essential. This study has certain limitations that must be highlighted. One limitation is the need for the proper collection and handling of samples to obtain accurate results, which can be challenging for some patients. Additionally, there is no rigorous quality control compared to analyses performed in a laboratory. Therefore, it is important to consider these limitations when interpreting the results obtained using the Foldscope. In conclusion, Foldscope microscope technology has the potential to have significant applications in the field of reproductive medicine by providing an affordable and straightforward option for evaluating semen sample quality. This could facilitate early detection of abnormal semen parameters and improve patient involvement in assessing their fertility. However, it is crucial to bear in mind that the results of these home tests should always be interpreted and supervised by a qualified health care professional since the Foldscope is not a medical diagnostic device and has certain limitations in terms of the variables it can evaluate.
Disclosures
The authors report no conflict of interest.
Funding
This study received no funding
Corresponding Author
Analyses were performed and the first draft of the manuscript was prepared by JE. ASM participated in the statistical analysis. JE and ASM were involved in the data interpretation. JE was involved in planning and designing the study and data interpretation. All authors reviewed and edited subsequent drafts of the manuscript and approved the final version.
References
- Foresta C, Moro E, Ferlin A (2001) Y chromosome microdeletions and alterations of spermatogenesis. Endocr Rev 22: 226-239.
- World Health Organization (2010) WHO laboratory manual for the examination and processing of human semen (5thedn). WHO, Geneva, Switzerland.
- Bhongade MB, Prasad S, Jiloha RC, Ray PC, Mohapatra S, et al. (2014) Effect of psychological stress on fertility hormones and seminal quality in male partners of infertile couples. Andrologia 47: 336-342.
- Petok WD (2015) Infertility counseling (or the lack thereof) of the forgotten male partner. Fertil Steril 104: 260-266.
- Schaff UY, Fredriksen LL, Epperson JG, Quebral TR, Naab S, et al. (2017) Novel centrifugal technology for measuring sperm concentration in the home. Fertil Steril 107: 358-364.
- Kobori Y, Pfanner P, Prins GS, Niederberger C (2016) Novel device for male infertility screening with single-ball lens microscope and smartphone. Fertil Steril 106: 574-578.
- Agarwal A, Panner Selvam MK, Sharma R, Master K, Sharma A, et al. (2018) Home sperm testing device versus laboratory sperm quality analyzer: Comparison of motile sperm concentration. Fertil Steril 110: 1277-1284.
- Chen YA, Chen KC, Tsai VF, Huang ZW, Hsieh JT, et al. (2013) Direct characterization of motion-dependent parameters of sperm in a microfluidic device: Proof of principle. Clin Chem 59: 493-501.
- Switz NA, D'Ambrosio MV, Fletcher DA (2014) Low-cost mobile phone microscopy with a reversed mobile phone camera lens. PLoS One 9: 95330.
- Cybulski JS, Clements J, Prakash M (2014) Foldscope: origami-based paper microscope. PLoS One 9: 98781.
- Breslauer DN, Maamari RN, Switz NA, Lam WA, Fletcher DA (2009) Mobile phone based clinical microscopy for global health applications. PLoS One 4: 6320.
- Dendere R, Myburg N, Douglas TS (2015) A review of cellphone microscopy for disease detection. J Microsc 260: 248-259.
- Yu S, Rubin M, Geevarughese S, Pino JS, Rodriguez HF, et al. (2018) Emerging technologies for home-based semen analysis. Andrology 6: 10-19.
- Smith ZJ, Chu K, Espenson AR, Rahimzadeh M, Gryshuk A, et al. (2011) Cell-phone-based platform for biomedical device development and education applications. PLoS One 6: 17150.
- Drobnis EZ, Rothmann SA (2018) A semen analysis phone app does not replace laboratory testing. Fertil Steril 110: 1255-1256.
Citation: Errázuriz J, Castillo C, Sepúlveda-Martínez A, Filselcker I, Alwane E, et al. (2023) Validation of the Origami “Foldscope” Microscope as a Preliminary Screening Tool for Male Infertility. J Reprod Med Gynecol Obstet 8: 144.
Copyright: © 2023 Joaquín Errázuriz, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

