
Vancomycin Polymethylmethacrylate Bead Plombage Thoracoplasty for Complex Empyema
*Corresponding Author(s):
Albert H Olivencia-YurvatiDepartment Of Surgery, University Of North Texas Health Science Center, Texas, United States
Tel:+1 8177355450,
Fax:+1 8177355454
Email:albert.yurvati@unthsc.edu
Abstract
A 51-year-old man was initially treated for necrotizing Mycobacterium avium infection and subsequently developed a chronic bronchopleural fistula. This complication was initially treated with an Eloesser flap and negative pressure vacuum therapy. The open thoracoplasty became chronically infected with methicillin resistant Staphylococcus aureus. This complicated intrathoracic infection was treated with a novel approach by the use of intrathoracic antibiotic impregnated Polymethylmethacrylate (PMMA) beads, in combination with negative pressure vacuum therapy. After 6 weeks of therapy the beads were extracted and subsequent cultures were negative for residual bacteria. This case report describes a novel protocol for the treatment of complex empyema not responsive to a more traditional approach.
Keywords
INTRODUCTION
Complicated and recurrent empyema challenges the thoracic surgeon as a complicated and protracted process of treatment. Empyema’s that develop as a result of drug resistant organisms are especially difficult to treat. The orthopedic, and in some respect plastic surgery literature, is replete with evidence supporting the use of antibiotic impregnated beads to combat musculoskeletal infections [1].
Advantages of using beads include the ability to provide local slow release therapy directly to the infected site in concentrations above minimum inhibitory concentration while avoiding systemic toxicity [2].
However, the thoracic literature has a very limited number of reported cases utilizing a similar technique. Thoracic empyema does have some well-established treatment modalities, including the Eloesser flap [3], originally described in 1935 and more recently, the use of negative pressure vacuum therapy [4,5]. Clinicians may encounter a subset of patients who may not completely respond to the described therapy alone or in combination. In addition, recurrent or even initial intrathoracic infections may present with organisms, which are difficult to eradicate. Failure to provide maximally effective therapy to these patients may result in longer antibiotic treatment duration, longer hospitalization and risk of repeated procedures all of which are associated with at least a theoretical risk of additive morbidity and mortality. Intravenous antibiotics have variable and sometimes poor penetration for these types of infection, as they tend to be organized collections of purulent and furious material [6].
REPORT OF A CASE
A 51-year-old man with a past medical history significant for bullous emphysema, obstructive sleep apnea and bronchitis, began to have severe dyspnea with worsening progression. His occupation was a bridge painter and was exposed to multiple noxious substances. He initially presented to his primary care physician with worsening pulmonary status and was eventually referred to the university cardiothoracic surgery clinic. The patient had a prior CT scan of the chest, which showed apical bullous disease with abscess.
He initially underwent a right thoracotomy with decortications. Intraoperatively, he was found to have extensive inflammatory changes with trapping of the lower lobe. The upper lobe had a large pocket consistent with a ruptured bulla with bronchopleural fistula and abscess present. The abscess was drained and irrigated with vancomycin solution and the fistula was repaired with tissue sealant. Due to the patients asthenic state and lack of an adequate latissimus dorsalis muscle a muscle flap was not a feasible alternative. Intraoperative cultures were obtained which resulted as Mycobacterium kansasii (>9/field); aerobic, anaerobic and fungal cultures were all negative at this initial operation.
The following day, he underwent repeat thoracotomy for continued and increasing air leak with massive subcutaneous emphysema. He was found to have an abscess pocket which was opened, as well as a large fistula and multiple smaller fistulas which were oversewn with prolene suture. An Eloesser flap was then performed at the 5th-6th ribs. Pathology of the resected ribs showed acutely inflamed fibrin debris consistent with a fistulous tract. The patient progressed well for the remainder of this initial hospital stay and was eventually discharged, then was subsequently lost to follow up.
He presented again approximately three years later to the infectious disease specialist with complaints of increasing shortness of breath and evidence of recurrent intrathoracic infection (Figure 1). He underwent repeat thoracotomy with decortication and placement of negative pressure vacuum therapy. Organisms identified via intraoperative cultures included methicillin resistant Staphylococcus aureus, Achromobacter xylosidans and Stenotrophomonas maltophilia. During re-operation, as the patient had an Eloesser flap in place, but was cachectic and there was no appreciable local muscle available. He lacked by CT scan a significant amount of omentum to utilize as a transfer flap. Therefore in an effort to attempt to control his chronic infection as well as continued bronchopleural fistula, he underwent plombage thoracoplasty fwith PMMA (Polymethylmethacrylate) beads impregnated with a total of ten grams of vancomycin.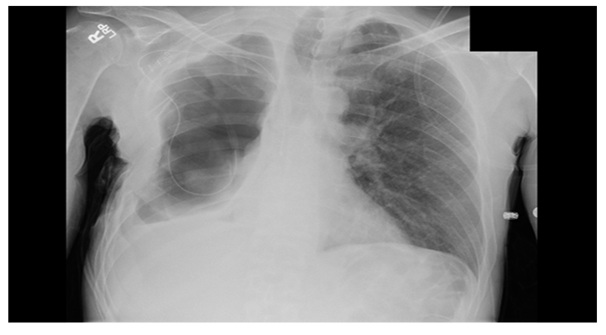
Figure 1: Large right hydropneumothorax with complete right lung collapse, as well as right pleural thickening.
The beads were placed in the pleural space and affixed to the chest wall for easy retrieval (Figure 2). A negative pressure vacuum therapy device was then affixed to the operative site. The wound vac was changed every third day. The patient progressed well post operatively and was eventually discharged to a rehab facility with plans to remove the antibiotic beads after a few weeks. Repeat cultures showed clearing of the MRSA infection at that time, and the patient returned to the operating room for removal of implanted hardware. He remains free of infection. The open Eloesser flap remains as chronic 2.5 cm fistula, without purulent drainage.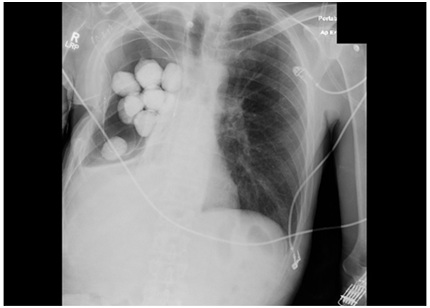
Figure 2: Post-operative film with PMMA beads in place.
DISCUSSION
Unfortunately, there is not a significant amount of literature supporting use of antibiotic impregnated beads in the thoracic setting. To our knowledge there are no published in vivo controlled trials in human subjects. A study by Liu et al., used penicillin G impregnated Poly(D,L)-lactide-co-glycolide (PLGA) beads and evaluated the release of antibiotic in vitro in phosphate-buffered saline and in vivo in the pleural cavities of New Zealand white rabbits. The results of the study confirmed the release of a fairly large amount of the antibiotic within the first day (40%) with the remainder being released over the following 30 days. However, the results showed drug concentrations remained significantly higher than the minimum inhibitory concentration throughout the study period [7].
The only other randomized controlled trial using antibiotic beads to treat empyema was conducted in 1988 by Mavroudis et al., [8]. This trial separated guinea pigs inoculated with intrathoracic Staphylococcus aureus into three groups; group I was the control group, group II had placement of placebo PMMA beads and group III animals were treated with intrathroacic tobramycin impregnated PMMA. The results of the trial showed that the group III animals did have a higher sterilization rate than either of the other two groups with, a statistically significant p value (<0.05), there was no difference in rate of early death among the various groups. These animals did not have concomitant tube thoracostomy or IV antibiotics, both of which would have been utilized in human subjects. Serum tobramycin levels were also drawn and were found to be negligible, supporting the fact that this type of local antibiotic delivery can be effective without systemic toxicity.
While the results of the study by Mavroudis et al were promising, PMMA beads are not biodegradable and must be removed with an additional operation. Human trials Should be undertaken, it would certainly be more advantageous to use a PLGA based delivery system, as this compound is entirely biodegradable and no additional intervention is required to remove the delivery system.
CONCLUSION
Though strictly a single case report, we believe the usage of antibiotic impregnated beads to treat empyema is an interesting alternative modality that deserves further investigation. Based on our report we recommend the use of PMMA beads for complex empyema at this time.
REFERENCES
- Gogia, JS, Meehan JP, Di Cesare PE, Jamlai AA (2009) Local Antibiotic Therapy in Osteomyelitis. Semin Plast Surg. 23: 100-107.
- Wininger DA, Fass RJ (1996) Antibiotic-impregnated Cement and Beads for Orthopedic Infections. Antimicrob Agents Chemother 40: 2675-2679.
- Thourani VH, Lancaster RT, Mansour KA, Miller JI (2003) Twenty-Six Years of Experience With the Modified Eloesser Flap. Ann Thorac Surg 72: 401-406.
- Sziklavari Z, Grosser C, Neu R, Schemm R, Szoke T, et al. (2013) Minimally Invasive Vacuum-Assisted Closure Therapy in the Management of Complex Pleural Empyema. Interact Cardiovasc Thorac Surg 17: 49-53.
- Palmen M, van Breugel HN, Geskes GG, van Belle A, Swennen JM, et al. (2009) Open Window Thoracostomy Treatment of Empyema Is Accelerated By Vacuum-Assisted Closure. Ann Thorac Surg 88: 1131-1136.
- Huttner AC, Harbarth S (2012) Beads vs Bugs?. Chest 141: 1136-1137.
- KS, Liu SJ, Chen HY, Huang YK, Peng YJ, et al. (2012) Steady Antibiotic Release From Biodegradable Beads in the Pleural Cavity: An In vitro and In vivo Study. Chest 141: 1197-1202.
- Mavroudis C, Katzmark SL, Ganzel BL, Gray LA, Polk HC (1988) Successful Treatment of Empyema Thoracis with Polymethylmethacrylate Antibiotic-Impregnated Beads in the Guinea Pig. Ann Thorac Surg 46: 615-618.
Citation: Olivencia-Yurvati AV, Buczek R (2017) Vancomycin Polymethylmethacrylate Bead Plombage Thoracoplasty for Complex Empyema. J Clin Stud Med Case Rep 4: 042.
Copyright: © 2017 Albert H Olivencia-Yurvati, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

