
A Comparison of Physico-Chemical Properties of Free and Chitosan Bead-Immobilized Pectin Lyase from Bacillus Cereus
*Corresponding Author(s):
Reena GuptaDepartment Of Biotechnology, Himachal Pradesh University, Summer Hill, Shimla-171005, India
Tel:+91 1772831948,
Fax:+91 1772831948
Email:reenagupta_2001@yahoo.com
Abstract
Keywords
INTRODUCTION
MATERIALS AND METHODS
Materials
Purification
Enzyme immobilization
Enzyme assay
Binding efficiency
Characterization of free and immobilized pectin lyase from Bacillus cereus
Statistics
RESULTS AND DISCUSSION
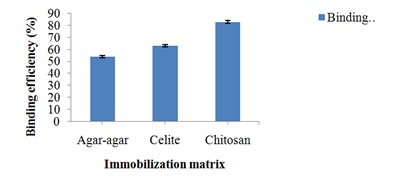
Binding efficiency of pectin lyase onto various supports
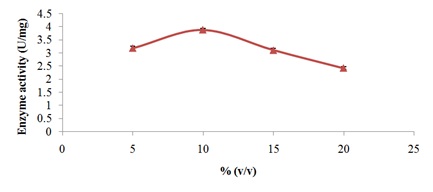
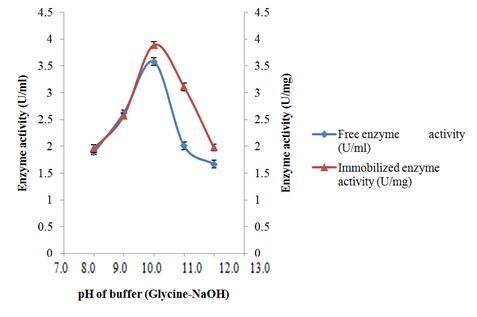
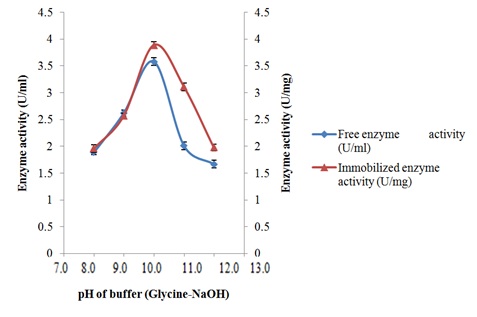
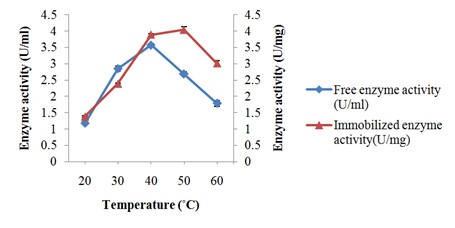
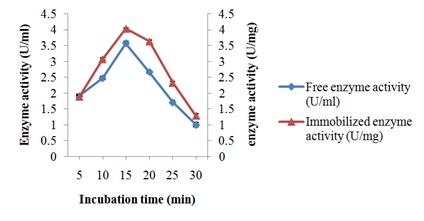
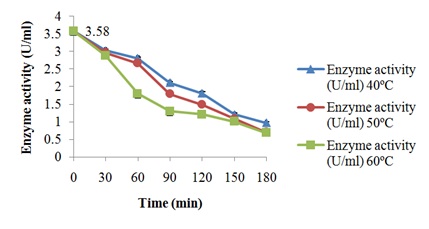
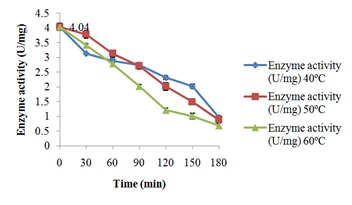
Characterization of free and immobilized pectin lyase from Bacillus cereus
| Substrate (0.5%, w/v) | Free enzyme activity (U/ml) | Immobilized enzyme activity (U/mg) |
| Apple pectin | 0.69±0.017 | 0.71±0.041 |
| Pectin | 1.41±0.032 | 1.80±0.028 |
| Polygalacturonic acid | 2.43±0.031 | 3.24±0.042 |
| Citrus pectin | 3.58±0.027 | 4.04±0.021 |
| Substrate concentration (%, w/v) | Free enzyme activity (U/ml) | Immobilized enzyme activity (U/mg) |
| 0.25 | 1.58±0.025 | 2.58±0.015 |
| 0.5 | 3.58±0.028 | 4.04±0.023 |
| 0.75 | 2.95±0.032 | 2.99±0.028 |
| 1 | 1.89±0.019 | 1.95±0.031 |
| 1.25 | 1.41±0.026 | 1.19±0.039 |
| 1.5 | 0.82±0.017 | 0.87±0.026 |
Effect of metal ions: Pectin lyase from Bacillus cereus showed maximum enzyme activity in the presence of Mg2+ ions for both free (3.59 U/ml) and immobilized enzyme (4.06 U/mg). However, Fe3+ also enhanced the activity of free and bound pectin lyase (Table 3). In a study, the Fe3+ ascorbic acid and Ca2+strongly activated pectin lyase, and Na+, Zn4+ and Mg2+ partially activated pectin lyase, K+ partially inhibited pectin lyase and Mn4+ had no effect on pectin lyase activity [16].
| Metal ion (1mM) | Free enzyme activity (U/ml) | Immobilized enzyme activity (U/mg) |
| Ca2+ | 1.79±0.02 | 1.98±0.03 |
| Cu2+ | 2.45±0.05 | 3.11±0.07 |
| Mg2+ | 3.59±0.02 | 4.06±0.02 |
| Zn2+ | 1.94±0.07 | 3.14±0.06 |
| Mn2+ | 1.41±0.03 | 1.91±0.04 |
| Fe3+ | 1.06±0.05 | 1.39±0.05 |
| Control | 0.95±0.07 | 1.01±0.02 |
| Cycle | Immobilized enzyme activity (U/mg) |
| 1 | 4.04±0.03 |
| 2 | 3.76±0.05 |
| 3 | 2.91±0.01 |
| 4 | 2.05±0.05 |
| 5 | 1.56±0.04 |
| 6 | 1.01±0.07 |
| 7 | 0.78±0.02 |
Table 4: Reusability of the immobilized pectin lyase from Bacillus cereus.
CONCLUSION
The covalent binding of pectin lyase on chitosan beads was found to be promising technique for the immobilization of pectin lyase. The thermostability of pectin lyase was increased after immobilization as compared to free enzyme. It exhibited reusability and retained more than 50% activity even after reusing it in the reaction 4 times. The improvement of temperature stability as well as reusability of pectin lyase after immobilization enhanced the potential of pectin lyase to be applicable in various industrial preparations.
ACKNOWLEDGEMENT
The Financial support from Department of Biotechnology, Ministry of Science and Technology, Government of India, to Department of Biotechnology, Himachal Pradesh University, Shimla (India), is thankfully acknowledged. The financial assistance to Ms. Pooja Kohli as RGNF from UGC and to Kamal Kumar Bhardwaj as CSIR fellow is thankfully acknowledged.
REFERENCES
- Zhou X, Liu Y, Yuan Q, Liang H (2016) Preparation of multi-enzyme co-immobilized nanoparticles by bis-aryl hydrazone bond conjugation. Biotechnol Appl Biochem 63: 214-219.
- Sharma T, Sharma A, Kanwar SS (2016) Purification and Characterization of an Extracellular High Molecular Mass Esterase from Bacillus pumilus. J Adv Biotechnol Bioeng 4.
- Sood S, Sharma A, Sharma N, Kanwar SS (2016) Carboxylesterases: Sources, Characterization and Broader Applications. Insights Enzyme Res 1.
- Hassan B, Ali S (2016) A Review on Biotechnological Impact of Pectinases in Industries. J Sci Res Phar Chem Bio Sci 1: 1-16.
- Mohnen D (2008) Pectin structure and biosynthesis. Curr Opin Plant Biol 11: 266-277.
- Jayani RS, Saxena S, Gupta R (2005) Microbial pectinolytic enzymes: A review. Process Biochem 40: 2931-2944.
- Bogra P, Kumar A, Kuhar K, Panwar S, Singh R (2013) Immobilization of tomato (Lycopersicon esculentum) pectinmethylesterase in calcium alginate beads and its application in fruit juice clarification. Biotechnol Lett 35: 1895-1900.
- Rehman HU, Aman A, Zohra RR, Qader SA (2014) Immobilization of pectin degrading enzyme from Bacillus licheniformis KIBGE IB-21 using agar-agar as a support. Carbohydr Polym 102: 622-626.
- Krajewska B (2004) Application of chitin- and chitosan-based materials for enzyme immobilizations: a review. Enzym Micro Tech 35: 126-139.
- Çetinusa ?A, Öztop HN (2000) Immobilization of catalase on chitosan film. Enzyme Microb Tech 26: 497-501..
- Nedjma M, Hoffmann, Belarbi A (2001) Selective and sensitive detection of pectin lyase activity using a colorimetric test: application to the screening of microorganisms possessing pectin lyase activity. Anal Biochem 291: 290-296.
- Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72: 248-254.
- Rehman HU, Aman A, Silipo A, Qader SAU, Molinaro A, et al. (2013) Degradation of complex carbohydrate: Immobilization of pectinase from Bacillus licheniformis KIBGE-IB21 using calcium alginate as a support. Food Chem 139: 1081-1086.
- Cerreti M, Markošová K, Esti M, Rosenberg M, Rebroš M (2017) Immobilisation of pectinases into PVA gel for fruit juice application. Int J Food Sci Tech 52: 531-539.
- Rajdeo K, Harini T, Lavanya K, Fadnavis NW (2016). Immobilization of pectinase on reusable polymer support for clarification of apple juice. Food Bioprod Process 99: 12-19.
- Poturcu K, Ozmen I, Biyik HH (2017) Characterization of an Alkaline Thermostable Pectin Lyase from Newly Isolated Aspergillus niger _WHAK1 and Its Application on Fruit Juice Clarification. Arab J Sci Eng 42: 19-29.
- Spagna G, Pifferi PG, Tramontini M (1995) Immobilization and stabilization of pectinlyase on synthetic polymers for application in the beverage industry. J Mol Catal A-Chem 101: 99-105.
- Wu R, He B, Zhao G, Li X (2014) Immobilization of pectinase on polyethyleneimine-coated pulp fiber for treatment of whitewater from papermaking. J Mol Catal B-Enzym 99: 163-168.
- Alkorta I, Garbisu C, Llama MJ, Serra JL (1996) Immobilization of pectin lyase from Penicillium italicum by covalent binding to nylon. Enzyme Microb Tech 18: 141-146.
- Namika, Lakhanpal A, Gupta R (2014) Immobilization of commercial pectinase on silica and its applications in pulm juice clarification. J Sci Technol 3: 38-47.
- Saxena S, Shukla S, Thakur A, Gupta R (2008) Immobilization of polygalacturonase from Aspergillus niger onto activated polyethylene and its application in apple juice clarification. Acta Microbiol Immunol Hung 55: 33-51.
- Kant S, Vohra A, Gupta R (2013) Purification and physicochemical properties of polygalacturonase from Aspergillus niger MTCC 3323. Protein Expr Purif 87: 11-16.
- Das A, Bhattacharya S, Reddy NV, Sundara RS (2013) Improved Production and Purification of Pectinase from Streptomyces sp. GHBA10 isolated from Valapattanam mangrove habitat, Kerala, India. Int Res J Biological Sci 2: 16-22.
- Ling XM, Wang XY, Ma P, Yang Y, Qin JM, et al. (2016) Covalent Immobilization of Penicillin G Acylase onto Fe3O4@Chitosan Magnetic Nanoparticles. J Microbiol Biotechnol 26: 829-836.
- Shukla SK, Saxena S, Thakur J, Gupta R (2010) Immobilization of polygalacturonase from Aspergillus niger onto glutaraldehyde activated nylon-6 and its application in apple juice clarification. Acta Aliment Hung 39: 277-292.
Citation: Dogra A, Kohli P, Bhardwaj KK, Gupta R (2018) A Comparison of Physico-Chemical Properties of Free and Chitosan Bead-Immobilized Pectin Lyase from Bacillus Cereus. Adv Ind Biotechnol 1: 002
Copyright: © 2018 Adarsh Dogra, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

