
Calcar-Replacing Total Hip Replacement for a Giant Cell Tumor of the Proximal Femur: A Case Report and A Review of Literature
*Corresponding Author(s):
Juan Paulo PantiDepartment Of Orthopedic Surgery, University Of The Philippines - Philippine General Hospital, Taft Ave, Manila, Philippines
Tel:+63 423314601,
Email:docpaulopanti@gmail.com
Abstract
Giant Cell Tumor of the bone (GCTb) is a relatively benign but locally aggressive tumor, capable of producing debilitating lytic bone lesions with a rare likelihood of producing lung metastases. The proximal femur is an uncommon location for its occurrence, with more than 50% of these epi-metaphyseal lesions occurring around the knee joint. The reported incidence of GCTb in the proximal femur is only reported to be around 3-4%. However, selecting the appropriate treatment is challenging due to the proximity with the hip joint, and the risk of damaging the insertion of several essential hip muscles. Joint-salvaging surgery primarily involves extended curettage, with or without adjuvant therapy, bone graft, or cement packing. Conversely, there is still considerable high recurrence rate even after extended curettage with cementing (0-29%). As such, hip replacement surgery is a suitable treatment for patients with pathologic fracture and for those with an extensive lesion of the proximal femur. We report our experience of a young patient with a pathologic femoral neck fracture, secondary to GCTb of the proximal femur. A calcar replacing stem was utilized since the tumor and surgical resection involved the lesser trochanter.
Keywords
LEVEL OF EVIDENCE
CASE REPORT
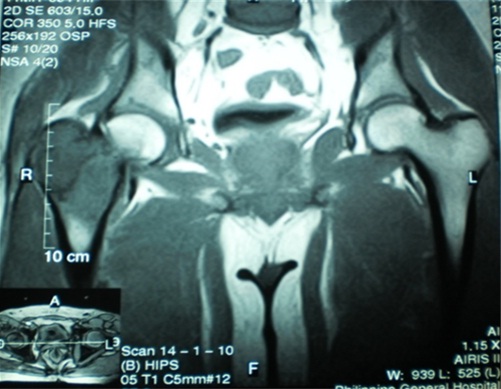
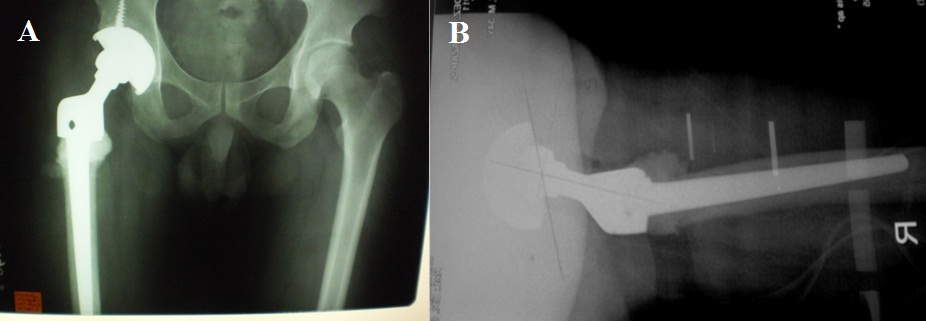
Two weeks prior to surgery, the patient’s pain increased in intensity up to a VAS score of 9/10. He did not recall any incident of fall or trauma. His limp worsened and was unable to put weight on his right lower extremity. He was eventually brought to our institution’s out-patient clinic. After initial assessment from the clinic’s triage, the patient was referred to our institution’s orthopedic and musculoskeletal tumor service. Physical examination done both by the orthopedic-musculoskeletal tumor specialist and orthopedic resident, indicated leg length shortening of 0.5 cm on the right lower extremity, right hip tenderness upon palpation, and a positive heel pound test. No skin lesions, palpable mass, nor lymphadenopathy were noted. The modified Musculoskeletal Tumor Society rating functional (MSTS) score was 4/30. Repeat plain radiographs of the hip revealed a pathologic fracture of the right femoral neck at the area of the lesion (Figure 1). The proximal femoral lesion was characterized as epi-metaphyseal, osteolytic with well demarcated borders, with endosteal expansion, cortical break, and has soft tissue extension anteriorly; but without any matrix. The lesion extended from the femoral neck up to the base of the lesser trochanter. The patient was admitted for further work-up and his right lower extremity was placed in skin traction. Magnetic resonance imaging of the hip confirmed a well delineated homogenous 8x7 cm lesion from the greater trochanter up to the level of the base of the lesser trochanter (Figure 2). A pre-operative tru-cut biopsy was done in the ward to exclude other differential diagnosis such as aneurysmal bone cyst and other primary bone tumor. The results confirmed giant cell tumor of the bone. The radiologic grade of the tumor was a Campanacci III due to the anterior cortical break and soft tissue extension, while the clinical and histologic grade was an Enneking 3 (for benign lesions). A pre-operative chest X-ray was done to rule out pulmonary metastasis, and showed negative results. The authors decided to pursue wide excision and perform total hip replacement due to the patient’s age and function, high recurrence for GTCb, along with the extensiveness of the lesion and the cortical break. Prosthesis specifications that were considered included a custom-made proximal femur endoprosthesis or a proximal femoral allograft composite. However, due to financial constraints and unavailability of a proximal femur allograft, a calcar-replacing femoral stem was selected due to the involvement and possible excision of the calcar femorale and lesser trochanter.
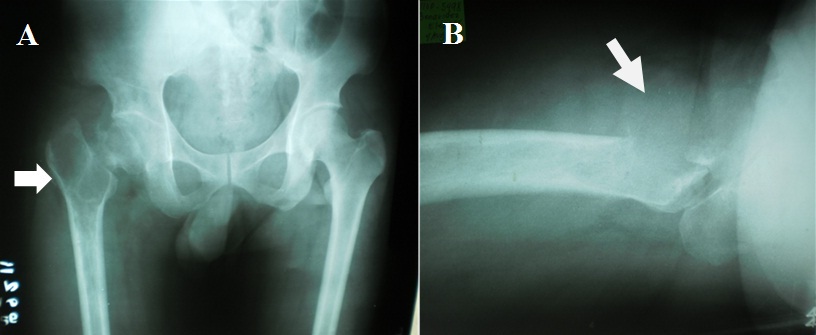
B) Arrow pointer showing cortical break at the anterior cortex of the greater trochanter and soft tissue extension of the tumor.
SURGICAL TECHNIQUE
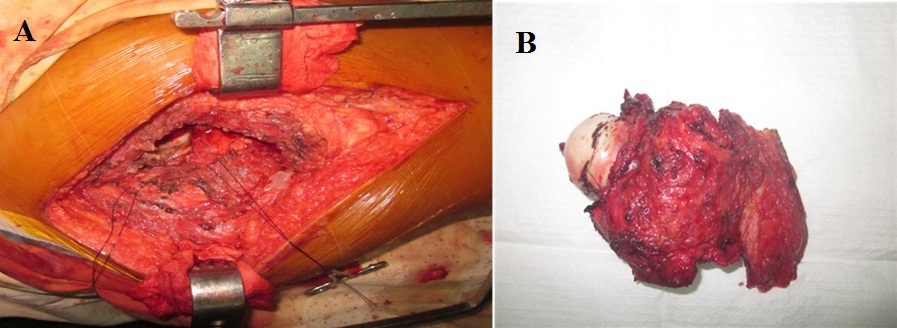
Figure 3: En-block resection of proximal femur up to 1.5 cm distal to the base of the lesser trochanter including insertion of iliopsoas, vastus lateralis, gluteus medius, short external rotators and proximal portion of tensor fascia lata.
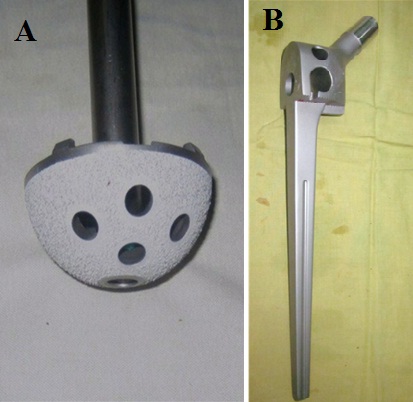
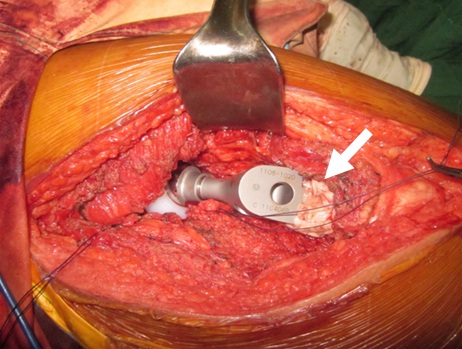
Figure 5: Arrow pointer showing excess cement that was moulded around the junction between the femoral shaft and the calcar component of the implant.
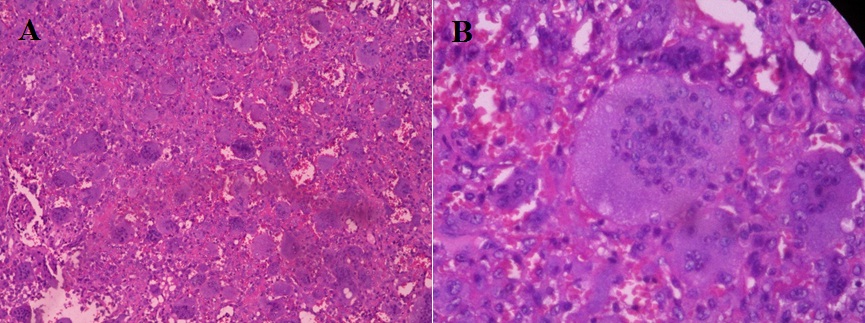
B) High power magnification: hematoxylin-eosin, original magnification x 100.
DISCUSSION
Joint salvaging surgery is primarily contemplated especially if the patient is relatively young, and the lesion is small and well contained [15-17]. This classically involves extended curettage, +/- adjuvant, and packing the defect either with bone graft or bone cement. Local recurrence rate after extended curettage however varies in literature, as it is 4.5-78% without cementing and 0-29% after cementing [18-20]. En-bloc resection of the lesion will certainly decrease, if not, eliminate the risk of tumor recurrence [21]. Additionally, it is presumed that there is no role for joint salvaging measures if the patient has a displaced pathologic fracture, or if the tumor is uncontained. In the contrary, Sim and Lang (1997) published a case report about a proximal femur GCTb with a transcervical fracture treated with internal fixation using a dynamic hip screw combined with valgus osteotomy, curettage, and bone grafting [17]. The latest follow-up was at 35 months, and the patient did not show any evidence of metastasis nor implant failure. However, at the latest follow-up there was a noted deformation of the femoral head that was absent prior to surgery. Furthermore, pre-operatively their patient did not have an extensive cortical break or soft-tissue extension unlike in our case. For our case, we opted for tumor resection and reconstruction due to the patient’s pathologic fracture, cortical break, soft-tissue extension, better tumor control and the lesser incidence of recurrence. The options for hip reconstruction after resection, includes excisional arthroplasty, hemi-arthroplasty or total hip replacement.
Hip joint reconstruction following excision is valuable for maintaining the stability and normalization of gait. However, there is always the risk of sacrificing vital tendinous insertions of hip abductors, adductors, flexors, and external rotators if the excision involves removing either the greater or the lesser trochanter. Furthermore, there is also the risk of taking out the dense vertically oriented calcar femorale at the posteromedial portion of the proximal femur, which support majority of the hip axial load. As with our patient, we expected to resect distal to the base of the lesser trochanter so we opted for a calcar-replacing long femoral stem. The aim is to have proximal portion of the femoral implant sitting on top of the femoral cortex, while the long stem provides stability and diaphyseal fixation. There are also designed holes on the proximal part of the femoral implant for re-attachment of the hip abductors and the iliopsoas (Figure 4B). Another option for hip reconstruction after excision of the proximal femur, would be an Allograft-prosthesis Composite (APC). Malhotra et al., reported a case series of 18 patients diagnosed with GCTb of the proximal femur treated with excision of the proximal femur, and APC using irradiated fresh-frozen allograft. With a mean follow-up of 54 months, all patients had complete graft union and excellent Harris hip scores in 13 patients [22]. We did consider using a proximal femoral allograft composite prior to the operation, however there was none available at our institution at that time. We also decided to do a cemented femoral fixation not only for stability, but also since the use PMMA bone cement seems to reduce tumor recurrence rate.
A review of literature investigating the treatment of proximal femur GCTb with excision and joint replacement was done (Table 1). The objectives of the literature search included analysis of the clinical outcomes, check for any tumor recurrence, and to determine any complications. The articles mostly were case report or case series, and there was heterogeneity with the type of implants used amongst the studies. All but one study stated no tumor recurrence on the latest follow-up. Out of the 51 patients with proximal femur GCTb treated with joint replacement in literature, there were only two patients who have had tumor recurrence. All studies reported improvement of latest functional outcome scores (Harris hip or MSTS score).
| Authors/ Year | No. of patients | Type of resection | Implant used | Mean follow-up (months) | Clinical outcome |
| Kulkarni SS et al., [23] | 1 | Wide excision | Charnley type low-friction THA | 32 | No tumor recurrence; Patient pain-free, full-weight bearing, but with mild Trendelenburg lurch |
| Sakayama K et al., [24] | 4 | Wide excision | Two THA (Implant not specified) and two bipolar hemi-arthroplasty | 77.5 | No tumor recurrence. The mean MSTS functional score was 93%. |
| Khan SA et al.,[25] | 12 | Wide excision | Customized, bipolar, cemented proximal femoral megaprosthesis | 57 | No tumor recurrence; No incidence of aseptic loosening or dislocation. The mean MSTS score is 28.3. |
| Nakano S et al., [26] | 1 | Wide excision | Cemented custom-made prosthesis | 384 (32 years) | No tumor recurrence, and with signs of bone formation at muscle re-attachment sites to the prosthesis. Mean Harris hip score of 100. |
| Dhatt S et al., [27] | 1 | Wide excision | Custom made megaprosthesis | 36 | No tumor recurrence nor any metastasis; no implant loosening. The MSTS score is 26.7. |
| Malhotra R et al., [22] | 18 | Wide excision | Uncemented Acetabular cup; ceramic-on-ceramic bearings; proximal femoral allograft-prosthesis composite | 54 | No tumor recurrence; Harris hip scores: 13 excellent and 5 good outcomes. All allografts united. |
| Wisbek AE et al., [28] | 14 | Wide excision | Total hip replacement (8 pxs), hemi-arthroplasty (1 px), endoprosthetic joint replacement (5 pxs) | 108.3 |
Mean MSTS scores for THR was 25.7 and endoprosthesis was 24.7. Complications: Two patients treated with THR had tumor recurrence; 1 THR and 1 endoprosthesis had dislocation; 1 hemi-arthroplasty was revised to a THR and 1 THR had an acetabular cup revision. |
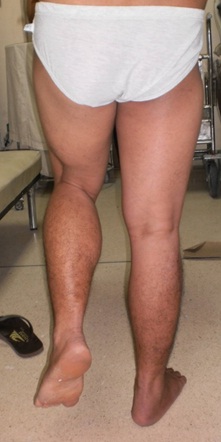
Comparable to data reported on literature, our patient had significant improvement in his MSTS score. Albeit the short term follow-up, it is important to note that the patient did not have any tumor recurrence or any mechanical signs of implant failure. It would be interesting to note the long term survivorship of this implant and follow-up the functional outcome of the patient in the future.
CONCLUSION
REFERENCES
- Szendröi M (2004) Giant-cell tumor of bone. J Bone Joint Surg Br 86: 5-12.
- Turcotte RE, Wunder JS, Isler MH, Bell RS, Schachar N, et al. (2002) Giant cell tumor of long bone: a Canadian Sarcoma Group study. Clin Orthop Relat Res 397: 248-258.
- Campanacci M, Baldini N, Boriani S, Sudanese A (1987) Giant-cell tumor of bone. J Bone Joint Surg Am 69: 106-114.
- Bloem JL, Reidsma II (2012) Bone and soft tissue tumors of hip and pelvis. Eur J Radiol 81: 3793-3801.
- Mirra JM (1989) Giant Cell Tumors. In: Mirra JM (ed.). Bone Tumors, Clinical Radiologic and Pathologic correlations, Volume 2. Lea and Febiger, Philadelphia, USA. Pg no: 942.
- Malone S, O’Sullivan B, Catton C, Bell R, Fornasier V, et al. (1995) Long-term follow-up of efficacy and safety of megavoltage radiotherapy in high-risk giant cell tumors of bone. Int J Radiat Oncol Biol Phys 33: 689-694.
- Bell RS, Harwood AR, Goodman SB, Fornasier VL (1983) Supervoltage radiotherapy in the treatment of difficult giant cell tumors of bone. Clin Orthop Relat Res. Pg no: 208-216.
- Bennett CJ Jr, Marcus RB Jr, Million RR, Enneking WF (1993) Radiation therapy for giant cell tumor of bone. Int J Radiat Oncol Biol Phys 26: 299-304.
- Zhou M, Yang H, Chen K, Wang G, Lu J, et al. (2013) Surgical treatment of giant cell tumors of the sacrum and spine combined with pre-operative transarterial embolization. Oncol Lett 6: 185-190.
- El Hadidi YN, Ghanem AA, Helmy I (2015) Injection of steroids intralesional in central giant cell granuloma cases (giant cell tumor): Is it free of systemic complications or not? A case report. Int J Surg Case Rep 8: 166-170.
- Kaban LB, Mulliken JB, Ezekowitz RA, Ebb D, Smith PS, et al. (1999) Antiangiogenic therapy of a recurrent giant cell tumor of the mandible with interferon alfa-2a. Pediatrics 103: 1145-1149.
- de Lange J, van den Akker HP, van den Berg H, Richel DJ, Gortzak RA (2006) Limited regression of central giant cell granuloma by interferon alpha after failed calcitonin therapy: a report of 2 cases. Int J Oral Maxillofac Surg 35: 865-869.
- Tse LF, Wong KC, Kumta SM, Huang L, Chow TC, et al. (2008) Bisphosphonates reduce local recurrence in extremity giant cell tumor of bone: a case-control study. Bone 42: 68-73.
- Mak IW, Evaniew N, Popovic S, Tozer R, Ghert M (2014) A translational study of the neoplastic cells of giant cell tumor of bone following neoadjuvant denosumab. J Bone Joint Surg Am 96: 127.
- Schajowicz F (1981) Giant cell tumor (Osteoclastoma). In: Schajowicz F (ed.). Tumors and tumorlike lesions of bone and joints, Springer - Verlag, New York, USA. Pg no: 205-242.
- Moon MS, Kim SS, Moon JL, Kim SS, Moon H (2013) Treating giant cell tumors with curettage, electrocautery, burring, phenol irrigation, and cementation. J Orthop Surg (Hong Kong) 21: 209-212.
- Sim E, Lang S (1997) Joint salvaging surgery for an extensive giant cell tumor of the proximal femur complicated by a transcervical fracture. Arch Orthop Trauma Surg 116: 431-434.
- O’Donnell RJ, Springfield DS, Motwani HK, Ready JE, Gebhardt MC, et al. (1994) Recurrence of giant-cell tumors of the long bones after curettage and packing with cement. J Bone Joint Surg Am 76: 1827-1833.
- Komiya S, Inoue A (1993) Cementation in the treatment of giant cell tumor of bone. Arch Orthop Trauma Surg 112: 51-55.
- Oda Y, Miura H, Tsuneyoshi M, Iwamoto Y (1998) Giant cell tumor of bone: oncological and functional results of long-term follow-up. Jpn J Clin Oncol 28: 323-328.
- Saikia KC, Bhattacharyya TD, Bhuyan SK, Bordoloi B, Durgia B, et al. (2011) Local recurrences after curettage and cementing in long bone giant cell tumor. Indian J Orthop 45: 168-173.
- Malhotra R, Kiran Kumar GN, Digge VK, Kumar V (2014) The clinical and radiological evaluation of the use of an allograft-prosthesis composite in the treatment of proximal femoral giant cell tumors. Bone Joint J 96: 1106-1110.
- Kulkarni SS, Dogra AS, Bhosale PB (1996) Total hip arthroplasty for giant cell tumor. J Postgrad Med 42: 82-84.
- Sakayama K, Sugawara Y, Kidani T, Miyawaki J, Fujibuchi T, et al. (2007) Diagnostic and therapeutic problems of giant cell tumor in the proximal femur. Arch Orthop Trauma Surg 127: 867-872.
- Khan SA, Kumar A, Inna P, Bakhshi S, Rastogi S (2009) Endoprosthetic replacement for giant cell tumor of the proximal femur. J Orthop Surg (Hong Kong) 17: 280-283.
- Nakano S, Enishi T, Hasan MY, Hanaoka N, Kawasaki Y, et al. (2009) Arthroplasty using a custom-made cemented total hip prosthesis for an extensive giant cell tumor of the proximal femur: report of a patient followed up for over 30 years. Arch Orthop Trauma Surg 129: 1171-1175.
- Dhatt S, Tahasildar N, Tripathy SK, Shashidhar BK, Tamuk T (2010) Excision and endoprosthesis implantation for proximal femur giant cell tumor. WebmedCentral ORTHOPAEDICS 1: WMC001236.
- Wijsbek AE, Vazquez-Garcia BL, Grimer RJ, Carter SR, Abudu AA, et al. (2014) Giant cell tumor of the proximal femur: Is joint-sparing management ever successful? Bone Joint J 96: 127-131.
Citation: Panti JP, Dimayuga C, Bernardo P (2015) Calcar-replacing Total Hip Replacement for A Giant Cell Tumor of the Proximal Femur: A Case Report and A Review of Literature. J Orthop Res Physiother 1: 010.
Copyright: © 2015 Juan Paulo Panti, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

