
Journal of Animal Research & Veterinary Science Category: Agriculture
Type: Case Report
Guinea Pig Laparoscopic Ovariectomy Using Two Portal Sites: 3-mm and 5-mm
*Corresponding Author(s):
Angelo E. Tapia-ArayaVeterinary Surgeon At Laparoendovet, Mobile Laparoscopy And Endoscopy Service, Barcelona, Spain
Tel:+34 661009555,
Email:angelo.tapia@gmail.com
Received Date: Jun 05, 2019
Accepted Date: Jun 14, 2019
Published Date: Jun 21, 2019
Abstract
The report describes the first veterinary Laparoscopic Ovariectomy (LapOve) in a 2 year old, 0.8kg, rescue, and sexually intact female guinea pig (Cavia porcellus) using two portal sites: 3-mm and 5-mm. Abdominal access was obtained using Hasson technique, allowing us to place the ports in the middle line. Carbon dioxide insufflation and abdominal exploration was made until both ovarian pedicles and horns were observed. The ovaries were fixed with sutures to the abdominal wall and cauterized with bipolar forceps, as well as the suspensory ligament, the proper ligament, mesovarium and the horns; they were then extracted once the trocar was removed. A two-port incision laparoscopic ovariectomy appears to be an easy minimal invasion technique, without postsurgical complications, that can be used routinely for guinea pig spays. Therefore, deeper studies should be carried out to evaluate this approach in clinical veterinary practice.
Keywords
Guinea pig; Laparoscopic; Minimal invasion; Ovariectomy; Ports
INTRODUCTION
A 0.8-kg, 2-year-old, sexually intact female rescue guinea pig (Cavia porcellus) was brought to a private veterinary center for spaying. Spaying is a common procedure in small animals and is also becoming common in exotic animals. Indications for spaying include prevention of fertile mating and prevention of reproductive diseases such as ovarian tumors, pyometra, endometrial hyperplasia, among others. The risk of postoperative infection in this species is high (33%) [1] compared to other small animals like dogs and cats (1-3%) [2]. Laparoscopic or “minimal invasion” surgery is well known for increasing visualization of surgeons, reducing postoperative pain, reducing surgical site infection, and shorter recovery times. The present report was conducted to examine the feasibility and safety of a laparoscopic procedure through two portals in order to establish it as a routine surgery for guinea pigs.
CASE REPORT DESCRIPTION
All procedures were performed in accordance with the Spanish Government for Animal Care guidelines (RD 53/2013).
The animal was classified as ASA class I (American Society of Anesthesiologists). It was premedicated [3] with medetomidine (Doctor; Norvet, Spain), 0.3 mg/kg Body Weight (BW), IM; butorphanol (Torbugesic 1%; Zoetis, Spain), 1 mg/kg BW, IM and alfaxan (Alfaxan 10mg/ml; Jurox; Crawley; UK) 1 mg/kg BW, IM. Marbofloxacin (Marbocyl 10%; Vetoquino; Spain), 4 mg/kg BW, SC and meloxicam (Metacam; Boehringer Ingelheim, Barcelona, Spain) 0.5 mg/kg BW, 30 minutes before surgery. The patient was maintained under general anesthesia using isofluorane (Isofluorano DCI 100%, Inibsa, Spain) in 100% oxygen (Figures 1-2).
The animal was classified as ASA class I (American Society of Anesthesiologists). It was premedicated [3] with medetomidine (Doctor; Norvet, Spain), 0.3 mg/kg Body Weight (BW), IM; butorphanol (Torbugesic 1%; Zoetis, Spain), 1 mg/kg BW, IM and alfaxan (Alfaxan 10mg/ml; Jurox; Crawley; UK) 1 mg/kg BW, IM. Marbofloxacin (Marbocyl 10%; Vetoquino; Spain), 4 mg/kg BW, SC and meloxicam (Metacam; Boehringer Ingelheim, Barcelona, Spain) 0.5 mg/kg BW, 30 minutes before surgery. The patient was maintained under general anesthesia using isofluorane (Isofluorano DCI 100%, Inibsa, Spain) in 100% oxygen (Figures 1-2).
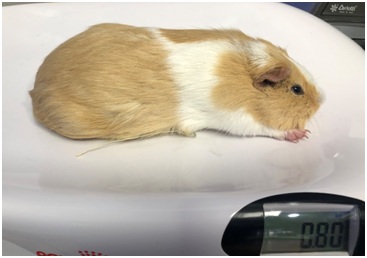
Figure 1: The animal during clinical examination.
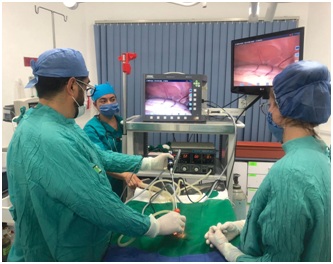 Figure 2: Surgeon performing the procedure.
Figure 2: Surgeon performing the procedure.Following induction, the patient was positioned in dorsal recumbency and reverse Trendelenburg position; the ventral abdomen was clipped and aseptically prepared with chlorhexidine 2% cranially from the xiphoid to the pubis. Abdominal access was obtained using Hasson technique, a 5mm incision was made - 1cm between the umbilicus and the pubis, through the skin and subcutaneous tissue down to the linea alba, where a 5-mm trocar was inserted inside the abdomen. Then pneumoperitoneum was established by connecting the trocar to the high flow CO2 (carbon dioxide) insufflators until the pressure of 5 mm Hg [4] was achieved. A 5-mm, 30-degree rigid laparoscope connected to the camera and light source was inserted into the abdomen and a 360 degree scan was performed to check all organs. A second 3mm incision was made midway cranial to the umbilicus and a 3-mm portal was inserted under direct visualization to prevent injury to the abdominal organs. The patient was then tilted 30 degrees either to the left and right lateral recumbency to perform the right or left OVE, respectively. With the rigid laparoscope the left and right kidney were identified and following this, the horns were visualized. The proper ligament was grasped with the 3-mm grasping forceps and elevated to the ventral part of abdomen. Then a 3/8 circle curved cutting needle was used passing through the body wall to suture with 2/0 poliglycolic acid (Monosyn, Braun) percutaneously the ovary. Following this, the 2.7 mm 30-degree rigid telescope was introduced via the cranial 3-mm portal and the ovarian pedicle, proper ligament and mesovarium were cauterized and cut using a 5-mm bipolar vessel sealing device (Ligasure, Valleylab, Tyco Healthcare UK). The same procedure was repeated with the contralateral ovary. When both ovaries were cut, the grasping forceps holding the resected ovary was brought directly towards the camera and simultaneously pulled backwards until the forceps entered inside the cannula and was observed from the outside. Finally, the trocar was pulled out while the ovary was removed from the abdomen and both incisions were closed using intradermic absorbable 4/0 monofilament suture (Figure 3).
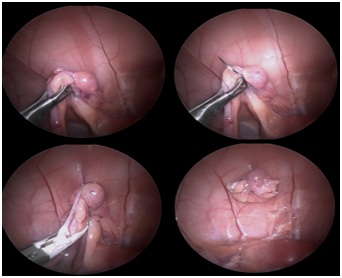
Figure 3: The sequence of steps during the procedure.
Total surgical time from the access to the removal of the second ovary and closure of incisions was 24 minutes. The guinea pig woke up 10 minutes after the surgery was finished and immediately food was offered to avoid hypoglycemia. The patient was maintained in the hospital and kept warm until full recovery. Six hours later, it was taken home. The wound was cleaned every day with saline solution 0.9% and no infection was seen. Post-surgery medication was meloxicam (Metacam; Boehringer Ingelheim, Barcelona, Spain) 0.5 mg/kg BW and enrofloxacino (Baytril 25 mg/ml, Bayer Hispania, Barcelona) 5 mg/kg BW bid 5 days. The owner reported the animal was active, eating and drinking that same night and the following days (Figures 4-6).
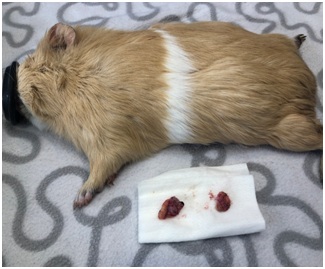 Figure 4: Image of the ovaries post-surgery.
Figure 4: Image of the ovaries post-surgery.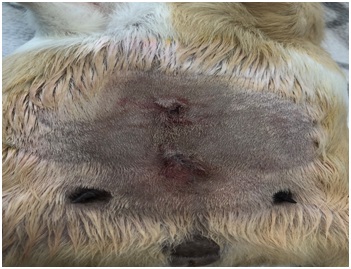 Figure 5: Image showing both incisions post-surgery.
Figure 5: Image showing both incisions post-surgery. Figure 6: The animal recovering after surgery.
Figure 6: The animal recovering after surgery.DISCUSSION AND CONCLUSION
The report describes the success of LapOve using minimal invasion of two portal sites. This is the first report of a laparoscopic ovariectomy in a guinea pig. Perioperative complications were not encountered, and the guinea pig was without clinical signs from the same day of surgery to the telephone conversation with the owner 2 weeks following the surgery (Video).
Guinea pigs as rodents can easily become hypothermic, so it is essential to provide supplementary heat during and after the surgery [5]. If the recovery is prolonged, dehydration and hypoglycemia can happen. The short surgical time and the minimal invasion of the procedure helped to avoid these possible complications.
Among the advantages of laparoscopic surgery, we have decreased postoperative pain, rapid return to preoperative activity, decreased postoperative ileus, and preserved immune function in contrast to open surgery which can lead to infections, eviscerations, wound dehiscence, hemorrhage, among others [6]. However, the cost of laparoscopy instruments, surgeon’s limited precision, poor ergonomics and shortage of learning curve are probable disadvantages to be considered [7].
A variety of methods for laparoscopic OVE with different numbers of portals have been described in veterinary medicine [8,9]. In order to enhance the feasibility and cosmetics of the techniques, two portals rather than three portals were chosen in the present study, as it was recommended previously.
In exotic species, the 2.7-mm telescope and 3-mm instrumentation are preferred for laparoscopic abdominal procedures in animals weighing less than 10 kg (e.g., many primates, felids, rabbits, and rodents) [10] making it less traumatic with minor incision. The 2.7-mm telescope significantly decreased the laparoscopic view but no complications were seen using the 2.7-mm 30° telescope.
We conclude that LapOve with two trocars is a minimally invasive technique that is feasible, safe and without postsurgical complications that could be considered as an alternative for the sterilization of female guinea pigs. Therefore, deeper studies should be carried out to evaluate this approach in clinical veterinary practice.
REFERENCES
- Johnson D (2009) Essential exotic companion mammal surgeries. Proc Assoc Exot Mammal Vet 57-59.
- Eugster S, Schawalder P, Gaschen F, Boerlin P (2004) A prospective study of postoperative surgical site infections in dogs and cats. Vet Surg 33: 542-550.
- Kuo W, Ko J, Collins BR, Keegan RD (2001) Evaluation of anesthetic and cardiorespiratory effects of tiletamine-zolazepam-ketamine-xylazine-butorphanol and medetomidine-butorphanol-ketamine in guinea pigs. Vet Anaesth Analg 28: 107-108.
- Mora MC, Bittner KR, Wong KE, Moriarty KP, Tashjian DB, et al. (2017) Laparoscopic Pediatric Inguinal Hernia Repair: A Pilot Study in a Novel Guinea Pig Animal Model. J Laparoendosc Adv Surg Tech A 27: 639-644.
- Olson ME, Bruce J (1986) Ovariectomy, ovariohysterectomy and orchidectomy in rodents and rabbits. Can Vet J 27: 523-527.
- Shariati E, Bakhtiari J, Khalaj A, Niasari-Naslaji A (2014) Comparison between two portal laparoscopy and open surgery for ovariectomy in dogs. Vet Res Forum 5: 219- 223.
- Amodeo A, Linares Quevedo A, Joseph JV, Belgrano E, Patel HR (2009) Robotic laparoscopic surgery: Cost and training. Minerva Urol Nefrol 61: 121-128.
- Tapia-Araya AE, Diaz-Guemes Martin-Portugues I, Bermejo LF, Sánchez-Margallo FM (2015) Laparoscopic ovariectomy in dogs: comparison between laparoendoscopic single-site and three-portal access. J Vet Sci 16: 525-530.
- Case JB, Marvel SJ, Boscan P, Monnet EL (2011) Surgical time and severity of postoperative pain in dogs undergoing laparoscopic ovariectomy with one, two, or three instrument cannulas. J Am Vet Med Assoc 239: 203-208.
- Divers SJ (2015) Endoscopic ovariectomy of exotic mammals using a three-port approach. Vet Clin North Am Exotic Animal Pract 18: 401-415.
Citation: Tapia-Araya AE, Sanz IT, Gómez JV (2019) Guinea Pig Laparoscopic Ovariectomy Using Two 3-mm and 5-mm Portal Sites. J Anim Res Vet Sci 3: 015.
Copyright: © 2019 Angelo E. Tapia-Araya, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
© 2026, Copyrights Herald Scholarly Open Access. All Rights Reserved!

