
Long Term Follow Up of the Lived Attenuated Japanese Encephalitis Vaccine (SA-14-14-2) Efficacy assessment of a Prime-Boost Vaccination in Children among Four Hyper-Endemic Provinces of Northern Thailand, 2016
*Corresponding Author(s):
Gonzalez JP6,Department Of Microbiology And Immunology, Georgetown University, School Of Medicine, Washington DC, United States
Tel:+66 3013322237,
Email:jpgonzalez@vet.k-state.edu
Abstract
Beside a strategic immunization campaign against Japanese Encephalitis (JE) that started in 1992, prioritizing high endemic areas, more tens of encephalitis cases are still annually registered in Thailand. Moreover, JE appears with higher incidence in Northern region (0.07 per 100,000 cases /year) as compared to the Central Southern region of the country (0.01 per 100,000 case/year). In 2012 an extensive JEV immunization campaign was order targeting the Northern Region of Thailand. Also, most of the JE Virus (JEV) recently isolated strains in country belonged to the JEV genotypes I and III, while the genotype II was isolated one time in 1983. It was then decided to use the newly live attenuated SA 14-14-2 Japanese encephalitis vaccine of a JEV genotype III developed in Thailand.
A year after such priming JEV immunization campaign, a second JEV immunization campaign was applied to deliver a booster dose of vaccine among children of four provinces of the Northern Region of Thailand including Chiang Rai, Phayao, Mae Hong Son, and Nan.
In order to evaluate the long-term efficacy of the SA 14-14-2 JE vaccine in Thailand, the immune response in children was timely studied following such second immunization campaign.
For each province, the recipients of the JEV vaccine booster dose were selected accordingly to the time of the vaccine campaign including: 1/ Children who received the second booster dose of vaccine less than one year before the present study (N=30 by province); 2/ children who received the second dose of vaccine more than one year before the present study (N=70 by province).
Altogether, including the four provinces, a total of 400 children were enrolled in this project. After receiving the second vaccine booster dose, blood samples were collected from each recipient and tested for neutralizing antibodies against JEV (Genotypes I and III).
As a result, all recipients presented neutralizing antibodies cross reacting against the two prevalent JEV genotypes I and III. Most of the children from the first group had a seropositivity rate of 94.2% (113/120) against homologous vaccine strain. Among them, an optimal rate of 100% seropositivity (30/30) was find at the Phayao Province against both heterologous JEV genotypes I and homologous genotypes I (SA 14-14-2 JEV strain). Geometric Mean Titer (GMT) of neutralizing antibody against SM1, SA 14-14-2 JEV, and Beijing JEV strains were consistently high, respectively as 239±50, 323±201.2 and 177±36.7 PRNT50. For the second group, seropositivity rate was lower against SM1, SA 14-14-2 and Beijing JEV strains with a rate of 94.3, 90% and 86.8% responding respectively to a GMT of 154±32, 90±23 and 82±13.6 PRNT50 against each JEV strain.
Although, the immunity seems to decrease within the period of 2.5 years, neutralizing antibody rate appears consistent with a high potential to protect children against the JEV infection.
Keywords
Encephalitis; Lived Attenuated Japanese Encephalitis Vaccine; SA (14-14-2).
INTRODUCTION
Japanese Encephalitis (JE) is the leading cause of viral encephalitis in the World including 50,000 cases of acute nervous system infections leading to over 15,000 of deaths yearly, while 23% of survivors develop serious permanent neurological sequelae [1]. The mosquito, Culex tritaeniorhynchus, transmits the virus to humans and to other amplifying hosts, such as swine, birds, and other vertebrates [2].
Though JE occurred within a variety of environment in Asia, JE epidemics in Thailand are confined to the Northern region of the country [3]. However, more than ten encephalitis cases are still registered annually in Thailand (Annual Epidemiological Surveillance Report 2015 Bureau of Epidemiology, Ministry of Public Health) that prompted the Thai Ministry of Public Health to implement strategic immunization campaign prioritizing high endemic areas stating in 1992 [4].
Although inactivated, Mouse Brain-Derived JE Vaccine (MBDV) was found to be highly effective and was included in the Expanded Program of Immunization (EPI) for children in several countries including Thailand [5] , due to safety concerns and the need for multiple injections, new vaccine generation were expected to replace MBDV [6]. In this matter, the SA14-14-2 live-attenuated JE vaccine (Chengdu Institute of Biological Products, People’s Republic of China) was licensed in Thailand.
The live JE vaccine (SA14-14-2) has been used in more than 300 million children since its large-scale production began in 1989. The safety of live JE vaccine is due to its high degree of neuro attenuation and other characteristics (e.g. homogeneous cell culture plaques; stable after tissue culture passages) as well as the absence of viremia in the recipients and replication in JEV mosquito vector. Therefore, the exceptional safety, stability, immunogenicity and long-term protective efficacy present a strong case for the expended use of this live attenuated JE vaccine [7]. The safety of this vaccine was demonstrated [8], while several field trials implemented in China and Thailand suggested the efficacy of a single dose of the SA14-14-2 live-attenuated JE vaccine was expected to reach 95% protection, and a two doses with one year interval may exceeded 98% [7-9].
While these results were based on neutralization test using a JEV strain genotype III homologous to the derived SA14-14-2 live-attenuated JE vaccine, JEV genotype I strain actively circulate in Thailand and was isolated from the neighboring country of Lao People’s Democratic Republic [10]. In 2012, the Ministry of Public Health of Thailand decided to administer the live-attenuated JE vaccine SA14-14-2 as a two doses campaign of immunization among children of Northern Thailand of eight JE potentially endemic Provinces. Then in 1992, the National Vaccine Institute Department of Communicable Diseases, Ministry of Public Health requested a JE vaccine SA14-14-2 vaccine efficacy study before including it in the present vaccine in EPI program.
From 2014 to 2015, a first dose vaccination campaign was extended to four Northern provinces of Thailand including: Chiang Rai, Phayao, Mae Hong Son, and Nan. Then a second dose was administered from 2015 to 2016 among the same cohorts of children, after which the present the present study on the efficacy was implemented.
MATERIALS AND METHODS
Study population and area: The children who received the second booster dose of live attenuated SA 14-14-2 Japanese Encephalitis (JE) Vaccine (Chengdu Institute of Biological Products, People’s Republic of China) where selected among 4 provinces of Chiang Rai, Phayao, Mae Hong Son and Nan among the Northern Region of Thailand (Table1) (Figure1).
|
District |
Province |
LAT |
LONG |
|
Mae Suai |
Chiang Rai |
19.63 |
99.524 |
|
Dok Khamtai |
Phayao |
19.195 |
100.034 |
|
Sop Moei |
Mae Hong Son |
18.113 |
98.166 |
|
Ban Luang |
Nan |
18.907 |
100.425 |
Table1: Selected study areas among four districts of four Provinces endemic for Japanese Encephalitis Virus of the Northern Region of Thailand, 2016.
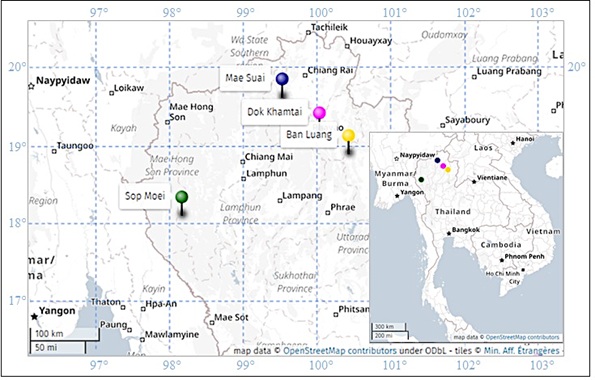 Figure 1: Study area of the Northern Provinces of Thailand.
Figure 1: Study area of the Northern Provinces of Thailand.
Note: Each colored pin refers to a study site (Purple = Mae Suai; Cyan = Dok Khamtai; Yellow = Ban Luang; Green = Sop Moe).
For each province, the children cohort (Retrospective study) was separated into two arms: The first arm of 30 children who received a prime dose less than 1 year before the study. The second arm of 70 children who receive the prime vaccine dose more than 1 year before the study. A total of 400 children were enrolled. Both sexes and age above 2.5-3.5 year were represented and 2 ml blood samples of taken. All samples were tested for JEV antibody by Plaque Reduction Neutralization Test (PRNT) against JEV genotype I and III.
Ethics: Oral consent was obtained from each child assisted by their parents or relative under the guidance of the Mahidol University Ethical Committee (MU: 2016-006).
Plaque Reduction Neutralization Test (PRNT). PRNT is one of the most specific serological tests for flavi virus eliciting a protective immune response that, therefore, shows a correlation with the neutralizing antibodies serum levels to the protection against JEV infection [11]. As previously described, PRNT involves virus–antibody interaction in a test tube, and then measures antibody effects on viral infectivity by plating the mixture on virus-susceptible cells. Briefly, LLC-MK2 cells were used for virus production of the SM1 JEV (Genotype I) strain, SA14-14-2 live-attenuated JE vaccine (Genotype III) and, and Beijing JEV (genotype III) strains [12]. Data were interpreted using the probity model with the SPSS program, and PRNT end point titers were expressed as the reciprocal value of the last serum dilution calculated on a 50% reduction in plaque counts on LLC-MK2 monolayer infected cells (PRNT50). Geometric Mean Titer (GMT) was expressed as a function of Excel program (Microsoft Office 2010).
RESULTS
The children from the first group (First vaccine dose less than 1 year) had a seropositivity rate of 90-100% (Table 2). The participants from the Phayao province had a high seropositivity rate of 100% against both heterologous (SM1 JEV) and homologous (SA 14-14-2) strains. Most of GMT for SM1, SA 14-14-2 and Beijing strains were consistently high about from 239±50, 323±201.2 and 177±36.7 respectively. Phayao province had highest GMT (707) against SA 14-14-2 strain.
The second group (First vaccine dose more than 1 year), when compared to the first group, showed a slightly reduced seropositivity rate (Table 3) between 86.9 to 94.3%, but nevertheless showing a consistent seropositivity rate corresponding to a GMT of 154±32, 90±23 and 82±13.6 respectively against SM1, SA 14-14-2 and Beijing JEV strains (Table 2).
|
Previous dose 1 |
Site |
PRNT 2 |
||
|
SM1 |
SA14-14-2 |
Beijing |
||
|
<1 year |
Chiang Rai |
28/30 (93.3) |
27/30 (90.0) |
27/30 (90.0) |
|
Phayao |
30/30 (100) |
30/30 (100) |
27/30 (90.0) |
|
|
Mae Hong Son |
27/30 (90.0) |
27/30 (90.0) |
27/30 (90.0) |
|
|
Nan |
29/30 (96.7) |
29/30 (96.7) |
27/30 (90.0) |
|
|
Total |
114/120 (95.0) |
113/120 (94.2) |
108/120 (90.0) |
|
|
>1 year |
Chiang Rai |
67/70 (95.7) |
67/70 (95.7) |
67/70 (95.7) |
|
Phayao |
65/70 (92.9) |
63/70 (90.0) |
58/70 (82.9) |
|
|
Mae Hong Son |
66/70 (94.3) |
60/70 (86.0) |
59/70 (84.3) |
|
|
Nan |
66/70 (94.3) |
62/70 (88.6) |
59/70 (84.3) |
|
|
Total |
264/280 (94.3) |
252/280 (90.0) |
243/280 (86.8) |
Table 2: Neutralizing antibody among Children sequentially immunized with two doses of the Japanese Encephalitis virus vaccine (SA14-14-2) within four highly endemic provinces of Thailand, 2015.
Legend: 1Blood sampling time after receiving the 2 vaccine doses; 2 Number positive/Totals (Percent)
This first study confirmed this vaccine safety while the administration of two doses provided a strong crossing immunogenicity against JE virus genotype I (SM1) and genotype III (SA 14-14-2 and Beijing).
|
Previous dose 1 |
Geometric Mean Titer (GMT) |
|||
|
Site |
SM1 |
SA14-14-2 |
Beijing |
|
|
<1 year |
Chiang Rai |
249 |
326 |
198 |
|
Phayao |
170 |
707 |
232 |
|
|
Mae Hong Son |
311 |
218 |
139 |
|
|
Nan |
246 |
217 |
154 |
|
|
Total ± SD2 |
239±50 |
323±201.2 |
177±36.7 |
|
|
>1 year |
Chiang Rai |
168 |
71 |
83 |
|
Phayao |
103 |
81 |
105 |
|
|
Mae Hong Son |
180 |
86 |
68 |
|
|
Nan |
180 |
131 |
77 |
|
|
Total ± SD |
154±32 |
90±23 |
82±13.6 |
|
Table 3: Geometric Mean Titer (GMT) of Japanese Encephalitis virus neutralizing antibody among Children sequentially immunized with two doses of the Japanese Encephalitis virus vaccine (SA14-14-2) within four highly endemic provinces of Thailand, 2015.
Legend: 1 Bleeding time after received 2 doses; 2 SD = Standard Deviation
DISCUSSION
Accordingly, with previous implementation of Japanese encephalitis campaign of SA14-14-2 live-attenuated JE vaccine, no adverse events where recorded in the present boosting dose vaccine campaign [13]. In year 2006, one hundred and fifty Thai children were vaccinated with one dose of this vaccine. The immunogenicity of a single dose vaccination was showed a GMT of 66.1 [9]. From our study the GMT was of 323 and 90 respectively after a booster dose period < 1 year and > 1 year. Either cohort showed a consistent GMT of 2, 3 and 1, 4-fold higher than after the first immunization campaign done in Thailand [9]. Moreover, after children received a second JEV vaccine dose they showed a consistent and efficient seropositivity rate (% 87%) against homologous as well as heterologous JEV genotypes I and III circulating in Thailand. Because the nature of this flavivirus based vaccine, the risk of Antibody-Dependent Enhancement (ADE) was also considered. Mice experimentally immunized with SA14-14-2 live-attenuated JE vaccine showed a IgG antibody cross-reactive response against Zika Virus (ZIKV) antigen without neutralizing antibody activity, moreover subsequent ZIKV challenge (C57BL/6 mice in Ifnar-/- 188 mice) after SA14-14-2 vaccination slightly lengthened the median survival. Although, ADE in SA14-14-2-immune sera was detected in vitro on cell culture ( C57BL/6 IFNAR/118 mice) [14-19]. Despite an ADE activity was detected in vitro with sera from with SA14-14-2-immunized mice [14-19], neither clinical nor biological signs with a potential harmful effect were recorded in the present study. Although, the immunity decreases, antibody neutralizing titer and GMT appears to be at an efficient protective rate against JE virus infection. Moreover, assuming (i.e. Sero prevalence) of Thai children of the class of age of < 2 years-old and 2 to 9 years old by having respectively 32% and 69% natural immunity JEV sero prevalence, will reinforce the vaccine immunity rate [15-16]. Nevertheless, a long term follows up of the protective immune response is needed to evaluate the necessity of a third dose to protect all children these class of age particularly vulnerable to JE [17-18].
ACKNOWLEDGEMENT
Funding for this research study was provided by National Vaccine Institute Department of Diseases Control, Ministry of Public Health. Thank you for staff of Center for Vaccine Development, Institute of Molecular Biosciences, Mahidol University at Salaya and the staffs of Chiang Rai Provincial Health Office, Phayao Provincial Health Office, Mae Hong Son Provincial Health Office and Nan Provincial Health Office, Ministry of Public Health, Thailand.
REFERENCES
- Solomon T, Dung NM, Kneen R, Gainsborough M, Vaughn DW, et al. (2000) Japanese encephalitis. J Neurol Neurosurg Psychiatry 68: 405-415.
- Gould DJ, Edelman R, Grossman RA, Nisalak A, Sullivan, MF (1974) Study of Japanese encephalitis virus in Chiangmai Valley, Thailand. Vector studies. Am J Epi 100: 49-56.
- Yamada T, Rojanasuphot S, Takagi M, Wungkobkiat S, Hirota T (1971) Studies on an epidemic of Japanese encephalitis in the northern region of Thailand in 1969 and 1970. Biken J 14:267-296.
- Muangchana C, Thamapornpilas P, Karnkawinpong O (2010) Immunization policy development in Thailand: The role of the Advisory Committee on Immunization Practice. Vaccine 1: 104-109.
- Muangchana C, Henprasertthae N, Nurach K, Theppang K, Yoocharoen P, Varinsathien P, et al. (2012) Effectiveness of mouse brain-derived inactivated Japanese encephalitis vaccine in Thai National Immunization Program: a case-control study. Vaccine 30: 361-367.
- Lindsey NP, Staples JE, Jones JF, Sejvar JJ, Griggs A, et al., (2010) Adverse event reports following Japanese encephalitis vaccination in the United States, 1999-2009.Vaccine. 29: 58-64.
- Sohn YM, Tandan JB, Yoksan S, Ji M, Ohrr H (2008) A 5-year follow-up of antibody response in children vaccinated with single dose of live attenuated SA14-14-2 Japanese encephalitis vaccine: immunogenicity and anamnestic responses. Vaccine. 26: 1638-1643.
- Xin YY, Ming ZG, Peng GY, Jian A, Min LH (1988) Safety of a live-attenuated Japanese encephalitis virus vaccine (SA14-14-2) for children. Am J Trop Med Hyg 39: 214-217.
- Chotpitayasunondh T, Sohn YM, Yoksan S, Min J, Ohrr H (2011) Immunizing children aged 9 to 15 months with live attenuated SA14-14-2 Japanese encephalitis vaccine in Thailand. J Med Assoc Thai 94:195-203.
- Aubry F, Vongsouvath M, Nougairède A, Phetsouvanh R, Sibounheuang B et al.,(2013) Complete Genome of a Genotype I Japanese Encephalitis Virus Isolated from a Patient with Encephalitis in Vientiane, Lao PDR. Genome Announc 1: 157-12.
- Russell PK, Nisalak A, Sukhavachana P, Vivona S (1967) A plaque reduction test for dengue virus neutralizing antibodies. J Immunol 99: 285-290.
- Nitatpattana N, Apiwathnasorn C, Barbazan P, Leemingsawat S, Yoksan S, et al. (2005) First isolation of Japanese encephalitis from Culex quinquefasciatus in Thailand. Southeast Asian J Trop Med Public Health 36: 875-876.
- World Health Organization (WHO) Weekly epidemiological record, No. 7, 14 February 2014
- Wang R, Zhen Z, Turtle L, Hou B, Li Y, et al., (2020) An Cross-Protection Against Zika Virus Infection Conferred by a Live Attenuated Japanese Encephalitis SA14-14-2 Vaccine.
- Yongxin Yu (2013). Development of Japanese Encephalitis Attenuated Live Vaccine Virus SA14-14-2 and its Characteristics. In Tech Open Science.
- Tsai TF, Yu Y (1995) Japanese encephalitis vaccines. In: Plotkin SA, Mortimer EA (eds) Vaccines, 2nd Edition, Philadelphia WB: Saunders: 671-713.
- Nitatpattana N, Dubot-Pérès A, Gouilh MA, Souris M, Barbazan P, et al. (2008) Change in Japanese encephalitis virus distribution, Thailand. Emerg Infect Dis 14: 1762-1765.
- Muangchana C, Thamapornpilas P, Karnkawinpong O (2010) Immunization policy development in Thailand: The role of the Advisory Committee on Immunization Practice. Vaccine 1: 104-109.
- Tsai TF, Yu YX, Jia LL, Putvatana R, Zhang R, et al.,(1998) Immunogenicity of live attenuated SA14-14-2 Japanese encephalitis vaccine-a comparison of 1- and 3-month immunization schedules. J Infect Dis 177: 221-223.
Citation: Nitatpattana N, Nakgoi K, , Juntarapornchai S, Ruangdachsuwan S, et al. (2020) Long Term Follow Up of the Lived Attenuated Japanese Encephalitis Vaccine (SA-14-14-2) Efficacy assessment of a Prime-Boost Vaccination in Children among Four Hyper-Endemic Provinces of Northern Thailand, 2016. J Vaccines Res Vaccin 6: 012.
Copyright: © 2020 Nitatpattana N, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

