
Monitoring of Persistent Organic Pollutants in Human Milk of Lazio Region
*Corresponding Author(s):
Fabio BusicoIstituto Zooprofilattico Sperimentale Delle Regioni Lazio E Toscana M Aleandri, Via Appia Nuova 1411, Rome, Italy
Tel:+039 0679099402,
Email:fabio.busico@izslt.it
Abstract
Direct or indirect prolonged exposure to organic contaminants diffused into the environment may lead to chronic toxicity through human milk. Infants and young children exposure during breast feeding can have a negative impact on adulthood. For this reason, some scientists express reservations on the benefits of lactation, despite the World Health Organization (WHO) and the American Academy of Pediatrics (AAP) continue to recommend breastfeeding. However, the human milk is the best way to monitor the level of exposure of these substances in the environment and in the food. This study studied the levels of Polychlorinated Dibenzodioxins, Polychlorinated Dibenzofurans (PCDD/F) and Polychlorinated Biphenyls (PCBs) in human milk in the Italian region of Lazio, which includes the provinces of Rome, Rieti, Viterbo, Latina, Frosinone, and correlated them with eating habits and residence of the donor mothers. We found no difference in Dioxins and PCB levels in human milk among the provinces of Lazio, with the exception of Rome, and no relationship between the levels of contamination detected in the milk and eating habits, confirming previously published literature. The levels of PCDD/F were compared with data obtained in other European countries between 1998 and 2015. We found a reduction of POPs in human milk compared to what has been detected in recent years in Italy and in other European countries and we conclude that the strict rules and the official controls in order to restrain the diffusion of those substances into the environment and into human food are gradually getting good results.
Keywords
INTRODUCTION
Persistent Organic Pollutants (POPs) are a group of toxic substances that are persistent, bio-accumulative and can be diffused in the environment even over long distances. POPs are resistant to chemical and biological degradation, and may remain in the soil, waters, and living organisms for long periods of time due to their chemical-physical properties. Dioxins (polychlorinated dibenzodioxins and polychlorinated dibenzofurans), polychlorinated biphenyls, and chlorinated pesticides belong to a list originally comprised of 12 POPs that are regulated under the Stockholm Convention on POPs [1]. Within individual dioxin congeners, the toxicity is considerably diverse. Because of the Cl-substitution at the 2,3,7,8 positions, only 17 of the 210 congeners have significant toxicity. The most toxic congener is the 2,3,7,8 TCDD, which has chlorine at positions 2,3,7,8 and is taken as a reference to toxicity [2-4]. POPs are lipophilic compounds, stable against degradation and able to bio-accumulate, thus they are highly persistent and ubiquitous in the environment and in the food chain [4]. POPs cause chronic toxicity at low doses when absorbed daily with food (meat, fish, eggs, milk),main source of contamination. Humans are at the top of the food chain with risk of accumulation of pollutants within the lipid fraction of tissues. Because, women have higher percentage fat tissue than men [5], they are at higher risk. It is not easy to demonstrate the existence of a connection between the long-term exposure to a specific POP (or of a group of POPs) and its impact on human health. However, exposure during the fetal and nursing period is particularly critical. The main effects of the substances of the study, PCDD/F and Polychlorinated biphenyls are carcinogenicity, genotoxicity, mutagenicity and teratogenicity [6]. Furthermore, these contaminants may mimic the activity of endogenous substances, which interact with aryl hydrocarbons receptor, an intracellular soluble protein able to bind to numerous aromatic, halogenated and non-halogenated substances. As 2,3,7,8-TCDD has the maximum affinity to the receptor, thus the highest toxicity, the affinity decreases with increasing Cl-substituents. This is also indicated by the lower TE factors for higher chlorinated PCDD/F [7-10].
These substances may travel from the mother to the fetus during the gestational and the nursing period. Breast milk monitoring is a non-invasive mean to determine the levels of the chemicals of interest in humans [11]. Over the last decade, various international studies have been conducted to provide information on POPs burden in women of reproductive age, together with the perinatal exposure of the fetus and the nursing infant [12]. These studies highlighted a significant reduction in the levels of PCDD/F-PCBs in most European countries following the Stockholm Convention. Today in breast milk there is an average of 5pg/g fat PCDD/ F and PCB TEQ [13-24].
The Italian studies in the provinces of Venice, Rome, Milan, Piacenza and Naples demonstrated variable values between 4.37 and 12.56pg WHO-TEQ2005/g fat [25,26]. The aim of this study was to define the levels of these pollutants in breast milk, and to correlate the types of pollutants with the mother’s dietary habits within the Lazio region. This information could be used by the Ministry of Health to inform women on the benefits of a correct diet, thus reducing the exposure to these pollutants.
MATERIALS AND METHODS
Samples
Chemical analisys
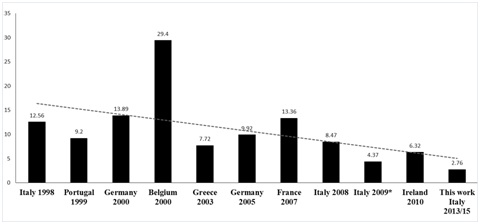 Figure 1: Concentrations of PCDD/F in human milk in several European countries found in the literature [12,16,17,19-21,24-26,29].
Figure 1: Concentrations of PCDD/F in human milk in several European countries found in the literature [12,16,17,19-21,24-26,29].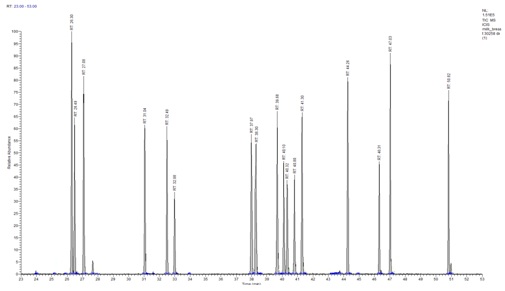 Figure 2: MID chromatogram in HRGC-HRMS, which represents the PCDD and PCDF content of a human milk sample. Tetra-substituted congeners (RT 26.30-27.08), penta-substituted congeners (RT 31.04-32.98), hexa-substituted congeners (RT 37.97-41.30), hepta-substituted congeners (RT 44.26-47.03), octa-substituted congeners (RT 50.82-50.91).
Figure 2: MID chromatogram in HRGC-HRMS, which represents the PCDD and PCDF content of a human milk sample. Tetra-substituted congeners (RT 26.30-27.08), penta-substituted congeners (RT 31.04-32.98), hexa-substituted congeners (RT 37.97-41.30), hepta-substituted congeners (RT 44.26-47.03), octa-substituted congeners (RT 50.82-50.91).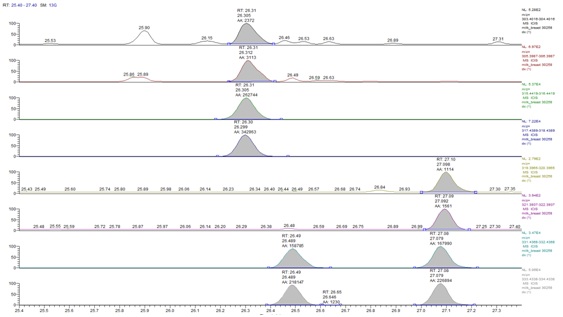 Figure 3: Ion extract of a breast milk sample shows the peaks of the native 2,3,7,8-TCDD (RT 27.10), the 2,3,7,8-TCDD labeled (RT 27.08), the 2,3,7 , 8-TCDF and 2,3,7,8-TCDF labeled (RT 26,30-26.31). At RT 26.49, the13C6 1,2,3,4 TCDD the recovery standard.
Figure 3: Ion extract of a breast milk sample shows the peaks of the native 2,3,7,8-TCDD (RT 27.10), the 2,3,7,8-TCDD labeled (RT 27.08), the 2,3,7 , 8-TCDF and 2,3,7,8-TCDF labeled (RT 26,30-26.31). At RT 26.49, the13C6 1,2,3,4 TCDD the recovery standard.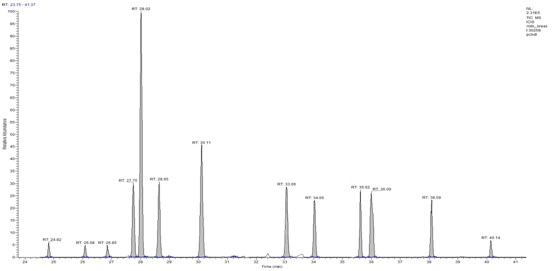 Figure 4: MID chromatogram in HRGC-HRMS, which represents the PCBs content of a human milk sample. Tetra-substituted congeners (RT 24.82-26.85), penta-substituted congeners (RT 27.75, 33.06), hexa-substituted congeners (RT 34.05-38.09), hepta-substituted congener (RT 40.14).
Figure 4: MID chromatogram in HRGC-HRMS, which represents the PCBs content of a human milk sample. Tetra-substituted congeners (RT 24.82-26.85), penta-substituted congeners (RT 27.75, 33.06), hexa-substituted congeners (RT 34.05-38.09), hepta-substituted congener (RT 40.14).Statistical analysis
|
Parameter |
N |
Value |
SD |
Min |
Max |
|
|
Mother age |
81 |
33 |
4.91 |
22 |
42 |
|
|
Body Mass Index (BMI) |
81 |
22.5 |
3.14 |
16.7 |
29.6 |
|
|
Newborns age (days) |
81 |
16.5 |
10.9 |
3 |
43 |
|
|
|
N |
Children |
N women |
Percentage |
||
|
Number of pregnancies |
|
1 |
24 |
33.3 |
||
|
|
|
2 |
34 |
47.2 |
||
|
|
|
>2 |
14 |
19.4 |
||
|
Resident in rural areas |
81 |
|
29 |
39.2 |
||
|
Smokers |
81 |
|
2 |
2.74 |
||
Expression of results and quality control
|
Pollutants |
Concentration of the Pollutants |
|||||
|
Average |
Median |
25th-75th Percentile |
Min. |
Max. |
||
|
2,3,7,8-T4CDD |
0.232 |
0.102 |
0.0176 |
0.393 |
0.064 |
1.22 |
|
1,2,3,7,8-P5CDD |
0.809 |
0.769 |
0.148 |
1.28 |
0.01 |
3.4 |
|
1,2,3,4,7,8-H6CDD |
0.367 |
0.305 |
0.0656 |
0.48 |
0.010 |
2.9 |
|
1,2,3,6,7,8-H6CDD |
1.86 |
1.51 |
0.779 |
2.62 |
0.01 |
7.58 |
|
1,2,3,7,8,9-H6CDD |
0.244 |
0.0481 |
0.0222 |
0.361 |
0.01 |
2.58 |
|
1,2,3,4,6,7,8-H7CDD |
2.29 |
1.69 |
0.531 |
2.58 |
0.01 |
44.4 |
|
O8CDD |
17.5 |
14.3 |
7.6 |
19.4 |
0.117 |
195 |
|
2,3,7,8-T4CDF |
0.330 |
0.230 |
0.0353 |
0.495 |
0.01 |
1.79 |
|
1,2,3,7,8-P5CDF |
0.155 |
0.064 |
0.0290 |
0.243 |
0.07 |
0.668 |
|
2,3,4,7,8-P5CDF |
2.65 |
2.44 |
1.42 |
3.96 |
0.0221 |
8.48 |
|
1,2,3,4,7,8-H6CDF |
0.766 |
0.694 |
0.294 |
1.13 |
0.01 |
4.37 |
|
1,2,3,6,7,8-H6CDF |
0.763 |
0.776 |
0.230 |
1.08 |
0.01 |
3.16 |
|
1,2,3,7,8,9-H6CDF |
0.196 |
0.0383 |
0.0231 |
0.204 |
0.01 |
1.25 |
|
2,3,4,6,7,8-H6CDF |
0.242 |
0.0514 |
0.0275 |
0.383 |
0.012 |
2.03 |
|
1,2,3,4,6,7,8-H7CDF |
0.792 |
0.585 |
0.215 |
0.911 |
0.01 |
7.06 |
|
1,2,3,4,7,8,9-H7CDF |
0.043 |
0.0257 |
0.0146 |
0.0449 |
0.05 |
0.433 |
|
O8CDF |
0.133 |
0.0332 |
0.0165 |
0.156 |
0.04 |
1.27 |
|
WHO-TEQ1998-PCDD-PCDF |
2.76 |
2.50 |
0.851 |
3.79 |
0.03 |
10.4 |
|
WHO-TEQ2005-PCDD-PCDF |
2.26 |
2.00 |
1.15 |
3.24 |
0.023 |
8.71 |
|
Pollutants |
Concentration of the Pollutants |
|||||
|
Average |
Median |
25th-75th Percentile |
Min. |
Max |
||
|
PCB 77 |
50,7 |
22,3 |
2.57 |
55.3 |
0.18 |
0.78 |
|
PCB 81 |
6,90 |
2,2 |
0.577 |
4.74 |
0.08 |
86.2 |
|
PCB 126 |
13,3 |
11,1 |
1.74 |
19.9 |
0.11 |
71.1 |
|
PCB 169 |
11,9 |
8,22 |
3.41 |
13.9 |
0.10 |
82 |
|
PCB 105 |
1090 |
896 |
583 |
1230 |
7.43 |
6930 |
|
PCB 114 |
293 |
229 |
146 |
416 |
0.16 |
1240 |
|
PCB 118 |
4420 |
3790 |
2150 |
5740 |
30.5 |
17500 |
|
PCB 123 |
42,5 |
40.2 |
23 |
57.9 |
0.18 |
126 |
|
PCB 156 |
2560 |
1980 |
1190 |
3830 |
18.1 |
7030 |
|
PCB 157 |
506 |
414 |
233 |
699 |
3.63 |
2390 |
|
PCB 167 |
685 |
557 |
371 |
948 |
5.93 |
23.20 |
|
PCB 189 |
258 |
184 |
113 |
382 |
1.85 |
1460 |
|
WHO-TEQ1998-PCBdl |
3.63 |
3.33 |
2.04 |
4.65 |
0.04 |
14.2 |
|
WHO-TEQ2005-PCBdl |
1.88 |
1.53 |
0.63 |
2.09 |
0.02 |
9.00 |
|
PCB-ndl (6 indicators sum) (ng/g fat) |
72.5 |
61.6 |
37.7 |
96.7 |
20.1 |
359 |
Table 2: Concentrations of congeneric of PCDD-PCDF and PCB-dl and their sum found in samples of human milk collected within 2013 and 2015 from 81 donors resident in the five provinces of Lazio region expressed as pg/g of fat and as pg/g of fat TEQ-WHO.
RESULTS
Eighty-one different samples of human breast milk have been analyzed for dioxin, dioxin like PCBs and non-dioxin like PCB (six indicators). The obtained data has been represented in table 1, values of toxicity (WHO-TEQ1998-PCDD-PCDF, WHO-TEQ1998-dl-PCBWHO-TEQ2005-PCDD-PCDF, WHO-TEQ2005-dl-PCB) have been calculated by multiplying obtained results for each congeneric with both factors (TEF) proposed by World Health Organization 1997 [2], and reviewed in 2005 [4], in order to compare the obtained data with the ones published in literature.
The average concentrations of contaminants expressed in WHO-TEQ2005 were comparable for the province of Rieti, Viterbo, Latina and Frosinone, while the province of Rome showed higher values (Figure 5).
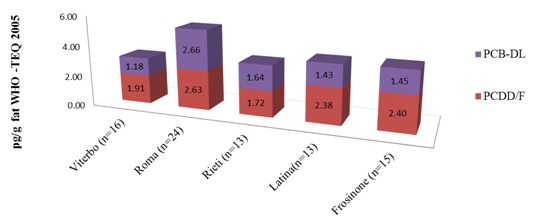
Figure 5: Average content of PCDD – PCDF – PCB-dl in the five provinces of Lazio expressed in pg/g fat TEQ –WHO2005
To compare the profiles of the congeners obtained in our study with those found in literature, the most abundant component (O8CDD for the dioxins and PCB118 for dioxin like Polychlorinated biphenyls) is 100% and the other congeners reported according to their abundance (normalization to 100). This profile is consistent to all Lazio provinces (Figure 6 and 7), and 99% similar with the profile of human milk analyzed in Ireland in 2010 (Figure 8) [21]. The average concentrations in pg WHO-TEQ1998/g fat for PCDD /F in the breast milk of Lazio were compared with data reported in the literature from 1998 to 2015 (Figure 1). There were not significant associations between provinces and the consumption of milk (p=0.166), cheese (p=0.123), fish (p=0.086) and meat (p=0.716), and between the consumption of food and the rural or urban living. Statistical analysis of the data showed no correlation between the levels of contamination of milk and feeding of donor mothers, as previously reported in the literature [26]. An important negative correlation was present between the number of pregnancies and the concentration of WHO-TEQ2005 PCDD/F (r=-0.966, p<0.05) and the sum of WHO-TEQ2005 PCB-dl+WHO2005-TEQ PCDD/F (r=-0.867, p<0.05).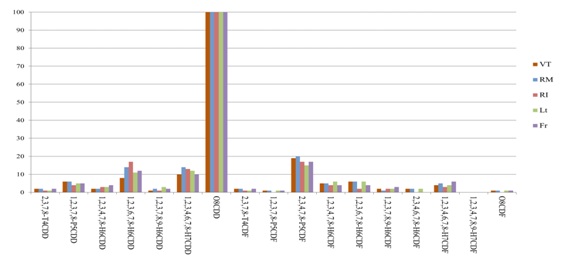
Figure 6: Comparison profile of PCDD/F congeners normalized for the five provinces of Lazio.
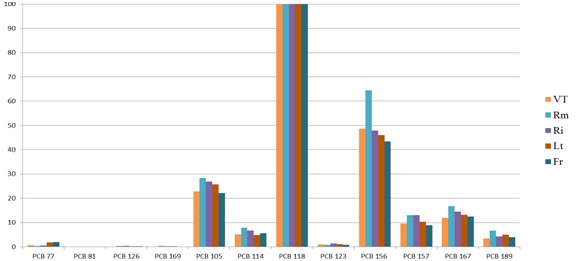 Figure 7: Comparison profile of PCB-dl congeners normalized for the five provinces of Lazio.
Figure 7: Comparison profile of PCB-dl congeners normalized for the five provinces of Lazio.
 Figure 8: Match found between the profile of PCDD/F to Lazio and to a study carried out in Ireland in 2010. Comparing the profiles of PCDD/F content in human milk of Lazio and Ireland.
Figure 8: Match found between the profile of PCDD/F to Lazio and to a study carried out in Ireland in 2010. Comparing the profiles of PCDD/F content in human milk of Lazio and Ireland.
DISCUSSION
The purpose of our study was to evaluate whether levels of PCDD-PCDF and PCB-dl in human milk of the Lazio provinces are different from the Italian and European average. The level of PCDD-PCDF and of dl-PCB found in Lazio’s human milk samples seemed to be lower than what previously reported in the national and European average, confirming a reduction of those contaminants in human milk during the last decade, with an estimation of 57% and 20.5% reduction of WHO-TEQ PCDDs/Fs and PCB-dl values, respectively [28] and a reduction of 80% in the last 20 years [3]. However, those values are higher than maximum levels allowed by European regulation of baby food. Dioxin and the amount of PCB-dl in the area found in our research show a lower value than other Italian regions, showing a favorable trend over time [25,26,29]. The lack of an evident correlation of dioxin and PCB-dl concentration with food habits agrees with previously published results obtained from the city of Rome in 2008 [26], and could be correlated to the globalization of food market. We found no difference in Dioxins and PCB-dl levels in human milk among the provinces of Lazio, with the exception of Rome, which had higher level, probably due to the bigger extension of the territory together with higher population compared to the other provinces and a lower consumption of locally produced foods. Furthermore,in other provinces of Lazio more local food is consumed. The data obtained in this work are part of the temporal trend that leads to the reduction of pollutants in human milk in Italy and in other countries of the European community (Figure 1) [26,30]. Decrease reported here likely reflects the gradual reduction of dioxin levels in the environment and subsequent decline in food, resulting in reduction of human exposure. The decreasing trend is probably also due to the consumption of food of animal origin containing lesser amounts of dioxins and PCBs coming from farms in which the level of pollutants in feed is kept under control. The content of these pollutants in food for farm animals is controlled in Italy by the "National Plan for Animal Feeding", which keeps the content of these contaminants in check by removing food with pops content above the legal limits from the market.
CONCLUSION
The human milk continues to prove the best way to monitor exposure to these substances. Nevertheless, the recommendation to breastfeed children according to the WHO recommendations is still valid, independently of the maternal diet. Thus, from this type of study we can provide a valid element to raise the awareness of society for the environment safety to reduce the human milk contamination. Which it has been noticed is that in Lazio region the contaminants are more present in the city center, for the reasons linked to the geographic characteristics (more populated is more polluted). Moreover, this study has noted a reduction in the level of contaminants of these substances in the environment and in human foods over the last decade, probably due to the implementation of stricter legislation and controls in the European community.
ACKNOWLEDGMENT
This research was funded by The Ministry of Health and approved by the ethic committee of Hospital Bambino Gesù. The authors are grateful to the operators of breast milk collection centers and donor mothers.
REFERENCES
- Listing of POPs in the Stockholm Convention Stockholm Convention.
- Van den Berg M, Birnbaum L, Bosveld AT, Brunström B, Cook P, et al. (1998) Toxic equivalency factors (TEFs) for PCBs, PCDDs, PCDFs for humans and wildlife. Environ Health Perspect 106: 775-792.
- World health Organization (WHO) and International Agency for Research on Cancer (IARC) (1997) IARC Monographs on the Evaluetion of Carcinogenic Risks to Humans-Polychlorinated dibenzo-para-dioxins and Polichlorinated dibenzofurans. Volume 69.
- Van den Berg M, Birnbaum LS, Denison M, De Vito M, Farland W, et al. (2006) The 2005 World Health Organization reevaluation of human and mammalian toxic equivalency factors for dioxins and dioxin-like compounds. Toxicol Sci 93: 223-241.
- Blaak E (2010) Gender differences in fat metabolism. Curr Opin Clin Nutr Metab Care 4: 499-502.
- IARC (2012) A review of human carcinogens-Part F: Chemical agents and related occupations. In: IARC Monographs on the Evaluation of Carcinogenic Risks to Humans, Volume 100F. IARC Press, Geneva, Switzerland.
- Schecter A, Birnbaum L, Ryan JJ, Constable JD (2006) Dioxins: An overview. Environ Res 101: 419-428.
- Lundqvist C, Zuurbier M, Leijs M, Johansson C, Ceccatelli S, et al. (2006) The effects of PCBs and dioxins on child health. Acta Paediatr Suppl 95: 55-64.
- Yamada T, Mishima K, Fujiwara K, Imura H, Sugahara T (2006) Cleft lip and palate in mice treated with 2,3,7,8-tetrachlorodibenzo-p-dioxin: A morphological in vivo study. Congenit Anom (Kyoto) 46: 21-25.
- Imura H, Yamada T, Mishima K, Fujiwara K, Kawaki H, et al. (2010) Effect of 2,3,7,8-tetrachlorodibenzo-p-dioxin suggests abnormal palate development after palatal fusion. Congenit Anom (Kyoto) 50: 77-84.
- WHO (2007) Fourth WHO-Coordinated Survey of Human Milk for Persistent Organic Pollutants in Cooperation with UNEP-Guidelines for Developing a National Protocol, Geneva, Switzerland.
- Focant JF, Pirard C, Thielen C, De Pauw E (2002) Levels and profiles of PCDDs, PCDFs and PCBs in Belgian breast milk. Estimation of infant intake. Chemosphere 48: 763-770.
- Chovancová J, ?onka K, Ko?an A, Sejáková ZS (2011) PCDD, PCDF, PCB and PBDE concentrations in breast milk of mothers residing in selected areas of Slovakia. Chemosphere 83: 1383-1390.
- Kami?ska J, Ligocka D, Zieli?ski M, Czerska M, Jakubowski M (2014) The use of PowerPrep and HRGC/HRMS for biological monitoring of exposure to PCDD, PCDF and dl-PCB in Poland. Int J Hyg Environ Health 217: 11-16.
- Lignell S, Aune M, Darnerud PO, Cnattingius S, Glynn A (2009) Persistent organochlorine and organobromine compounds in mother’s milk from Sweden 1996–2006: Compound-specific temporal trends. Environ Res 109: 760-767.
- Raab U, Preiss U, Albrecht M, Shahin N, Parlar H, et al. (2008) Concentrations of polybrominated diphenyl ethers, organochlorine compounds and nitro musks in mother’s milk from Germany (Bavaria). Chemosphere 72: 87-94.
- Reis MF, Sampaio C, Aguiar P, Maurício Melim J, Pereira Miguel J, et al. (2007) Biomonitoring of PCDD/Fs in populations living near portuguese solid waste incinerators: Levels in human milk. Chemosphere 67: 231-237.
- Schecter A, Piskac AL, Grosheva EI, Matorova NI, Ryan JJ, et al. (2002) Levels of dioxins and dibenzofurans in breast milk of women residing in two cities in the Irkutsk region of Russian Siberia compared with American levels. Chemosphere 47: 157-164.
- Wittsiepe J, Fürst P, Schrey P, Lemm F, Kraft M, et al. (2007) PCDD/F and dioxin-like PCB in human blood and milk from German mothers. Chemosphere 67: 286-294.
- Costopoulou D, Vassiliadou I, Papadopoulos A, Makropoulos V, Leondiadis L (2006) Levels of dioxins, furans and PCBs in human serum and milk of people living in Greece. Chemosphere 65: 1462-1469.
- Pratt IS, Anderson WA, Crowley D, Daly SF, Evans RI, et al. (2012) Polychlorinated Dibenzo-P-Dioxins (PCDDs), Polychlorinated Dibenzofurans (PCDFs) and Polychlorinated Biphenyls (PCBs) in breast milk of first-time Irish mothers: impact of the 2008 dioxin incident in Ireland. Chemosphere 88: 865-872.
- Croes K, Colles A, Koppen G, Govarts E, Bruckers L, et al. (2012) Persistent Organic Pollutants (POPs) in human milk: A biomonitoring study in rural areas of Flanders (Belgium). Chemosphere 89: 988-994.
- Dimitriadou L, Malarvannan G, Covaci A, Lossifidou E, Tzafettas J, et al. (2016) Levels and profiles of brominated and chlorinated contaminants in human breast milk from Thessaloniki, Greece. Sci Total Environ 539: 350-358.
- Focant JF, Fréry N, Bidondo ML, Eppe G, Scholl G, et al. (2013) Levels of polychlorinated dibenzo-p-dioxins, polychlorinated dibenzofurans and polychlorinated biphenyls in human milk from different regions of France. Sci Total Environ 452-453: 155-162.
- Giovannini A, Rivezzi G, Carideo P, Ceci R, Diletti G, et al. (2014) Dioxinslevels in breastmilk of women living in Caserta and Naples: Assessment of environmental risk factors. Chemosphere 94: 76-84.
- Abballe A, Ballard TJ, Dellatte E, Di Domenico A, Ferri F, et al. (2008) Persistent enviromental contaminants in Human milkconcentrations and time trends in Italy. Chemosphere 73: 220-227.
- EPA (1994) Method 1613, Tetra- through Octa-Chlorinated Dioxins and Furans by Isotope Dilution HRGC/HRMS.
- Weiss J, Päpke O, Bignert A, Jensen S, Greyerz E, et al. (2003) Concentrations of dioxins and other organochlorines (PCBs, DDTs, HCHs) in human milk from Seveso, Milan and a Lombardian rural area in Italy: A study performed 25 years after the heavydioxin exposure in Seveso. Acta Pediatr 92: 467-472.
- Ulaszewska MM, Zuccato E, Capri E, Iovine R, Colombo A, et al. (2011) The effect of waste combustion on the occurrence of Polychlorinated Dibenzo-P-Dioxins (PCDDs), Polychlorinated Dibenzofurans (PCDFs) and Polychlorinated Biphenyls (PCBs) in breast milk in Italy. Chemosphere 82: 1-8.
- Zietz BP, Hoopmann M, Funcke M, Huppmann R, Suchenwirth R, et al. (2008) Long-term biomonitoring of polychlorinated biphenyls and organochlorine pesticides in human milk from mothers living in northern Germany. Int J Hyg Environ Health 211: 624-638.
- EPA 1613, Tetra-through Octa-Chlorinated Dioxins and Furans by Isotope Dilution HRGC/HRMS. October 1994.
- EPA 1668, Method 1668B Chlorinated Biphenyl Congeners in Water, Soil, Sediment, Biosolids, and Tissue by HRGC/HRMS. November 2008.
Citation: Busico F, Salvatori G, Pannone V, Berretta S, Mattei G, et al. (2019) Monitoring of Persistent Organic Pollutants in Human Milk of Lazio Region. J Toxicol Cur Res 3: 009.
Copyright: © 2019 Fabio Busico, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

