
Outpatient Asthma Management without Rescue Bronchodilators?
*Corresponding Author(s):
Luis Javier NaniniFaculty Of Medical Sciences, Rosario National University, Rosario, Argentina
Tel:+54 341 4362300,
Email:ljnannini@hotmail.com
Abstract
Overreliance on Short Acting Beta2 Agonists (SABA) is associated with increased risk of death from asthma in a dose-response fashion.
Objective
We wonder if there any subpopulation of asthmatic individuals that live without SABA. To face this possibility, we studied the rescue strategy of individuals that consecutively came for a first outpatient visit.
Methods
Individuals that did not have SABA for use as rescue medication in the last three months constituted the “NO SABA group”; and those who used SABA at least once in the last 3 months: “SABA group”. They completed the Asthma Control Test (ACT) and performed spirometry.
Results
“SABA group” aged 38.82±15.13 yrs (n=65, 26 males) used inhaled salbutamol MDI as rescue therapy. The NO SABA group” (n=21, 8 males; mean (SD) aged 39.91±17.77 yrs) used for symptoms relief as follows: salmeterol/fluticasone (n=5), formoterol/budesonide (n=3); salbutamol/beclomethasone (n=2); budesonide (n=8); intramuscular dexamethasone (n=1); nebulized normal saline solution (n=1); and one individual did nothing. NO SABA group showed statistically significant better ACT (median= 18 vs SABA=11; p=0.0056, Mann Whitney test). NO SABA group had higher number of individuals under regular ICS (10/21 vs 21/65; p=0.0418; Fisher's exact test); and with higher doses (mean: 221.23 micrograms [95%CI: 59-384]; vs SABA group: 123.9 [95% CI: 63-184]; p=0.0318). Also, the number of individuals with poorly controlled asthma was lower in the NO SABA group.
Conclusion
We observed that asthmatic individuals could live at least 3 months without SABAs and with better outcomes. Increased preventer adherence could be a cause or a consequence.
Keywords
INTRODUCTION
PATIENTS AND METHODS
RESULT
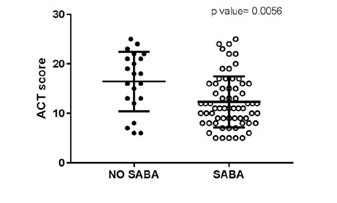 Figure 1: Graphical representation.
Figure 1: Graphical representation.|
Variables |
No SABA n=21 |
SABA n= 65 |
p |
|
Age Yrs mean±SD |
39.91±17.77 |
38.82±15.13 |
0.78 NS* |
|
Male/Female |
8/16 |
26/39 |
0.574 NS# |
|
History of asthma (yrs mean±SD) |
26.78±20.11 |
25.52±15.67 |
0.81 NS* |
|
Reason for visit Not due to asthma (n) |
8 |
20 |
0.6 NS# |
|
ACT mean±SD |
15.70±6.80 |
12.32±5.27 |
0.0056* |
|
FEV1% predicted |
78.94±20.01 |
75.20±17.65 |
0.3676 NS* |
|
Percent FEV1 Change post bronchodilator |
9.69±10.63 |
15.83±14.46 |
0.088 NS* |
|
Hospital/E Room visit 12 m (n) |
4 |
22 |
0.5 NS# |
|
Subjects with regular ICS (n) |
10 |
21 |
0.0418# |
|
ICS dose microgr Mean (95%CI) |
221.23 (59-384) |
123.9 (63-184) |
0.0318* |
|
Poorly controlled asthma (ACT<16) |
8/16 |
48/17 |
0.0041# |
|
Daily maximum of rescue medication mean (95%CI) |
1.27 (0.45-2.1) |
12.32 (6.37-18.27) |
<0.0001* |
|
*Mann Whitney test #Fisher's exact test |
|||
DISCUSSION
Overreliance on SABA and relief behavior became an upward spiral of SABA consumption that was associated with increased risk of asthma death; and furthermore, it conspired to the pathogenic essence of asthma treatment; namely anti-inflammatory therapy and its adherence. Indeed, first step in asthma recommendations should not keep blank[9]. The strategy of Combined ICS/formoterol as needed for GINA step 3 to 5 is not widely accepted[5].
As observational design, this study had several limitations. Firstly, investigators implemented a “SABA free asthma management”[10]. The second bias is that as one of the questions of ACT was rescue therapy, the NO SABA group had lower use even when we explained to the individuals that their response should include all the agents used to relief symptoms. Anyway, the exclusion from the ACT of the item 4 did not change the results at all. The high rate of drop outs for a first outpatient visit and the low percentage of well controlled individuals constituted a faithful measure of how far away were GINA recommendations from daily practice.
CONCLUSION
AUTHORS CONTRIBUTION
CONFLICT OF INTEREST
REFERENCES
- Luis Javier Nannini received fees as speaker and advisory board for AstraZeneca, Argentina SA and Novartis Argentina SA. Nadia S Neumayer, Octavio M Fernández and Daniela M Flores had no conflict of interest to declare.
- GBD 2015 Chronic Respiratory Disease Collaborators (2017) Global, regional, and national deaths, prevalence, disability-adjusted life years, and years lived with disability for chronic obstructive pulmonary disease and asthma, 1990-2015: a systematic analysis for the Global Burden of Disease Study 2015. Lancet Respir Med 5: 691-706.
- Suissa S, Ernst P, Boivin JF, Horwitz RI, Habbick B, et al. (1994) A cohort analysis of excess mortality in asthma and the use of inhaled beta-agonists. Am J Respir Crit Care Med 149: 604-610.
- Nannini LJ (2018) Overreliance link between asthmatic subjects and short-acting β2 agonists. Pulm Pharm Ther 50: 80-81.
- Global Initiative for asthma (2018) Global strategy for asthma management and prevention, 2018.
- Nathan RA, Sorkness CA, Kosinski M, Schatz M, Li JT, et al. (2004) Development of the Asthma Control Test: a survey for assessing asthma control. J Allergy Clin Immunol 113:59-65.
- Rodrigo GJ, Arcos JP, Nannini LJ, Neffen H, Gonzalez Broin M, et al. (2008) Reliability and factor analysis of the Spanish version of the Asthma Control Test. Ann Allergy Asthma Immunol 100: 17-22.
- Miller MR, Hankinson J, Brusasco V, Burgos F, Casaburi R, et al. (2005)Standardisation of spirometry. ATS/ERS Task Force: standarisation of lung function testing. Eur Respir J26:319-338.
- Nannini LJ, Neumayer NS (2018) Treatment Step 1 for Asthma Should Not Be Left Blank, and SABA-Only Might Not Be a Treatment Step 1 Option for Asthma. Respiration95:212-214.
- Nannini LJ (2019) Treat to target approach for asthma. J Asthma 23:1-4.
Citation: Nannini LJ, Neumayer NS, Fernandez OM, Flores DM(2019) Outpatient Asthma Management without Rescue Bronchodilators?. J Pulm Med Respir Res 5: 024.
Copyright: © 2019 Luis Javier Nanini, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

