
Prognostic Value of the Tumor Marker Index Based on Preoperative Serum Carcinoembryonic Antigen and Cytokeratin-19 Fragment Levels for Overall and Disease-Free Survival in Patients with Stage I Non-Small Cell Lung Cancer
*Corresponding Author(s):
Gaku YamaguchiDepartment Of Thoracic Surgery, Chemotherapy Research Institute, Kaken Hospital, Kohnodai, Ichikawa-shi, Chiba, Japan
Tel:047-375-1111,
Fax:047-373-4921
Email:ugaku@hotmail.com
Abstract
Purpose
Increased preoperative serum Carcinoembryonic Antigen (CEA) and Cytokeratin-19 Fragment (CYFRA 21-1) levels are associated with a poor postoperative prognosis in early-stage Non-Small Cell Lung Cancer (NSCLC). The Tumor Marker Index (TMI), calculated as the mean of the CEA and CYFRA 21-1 levels normalized to their respective diagnostic cutoff values, is a predictor of prognosis. This study aimed to validate the prognostic value of the TMI for Overall Survival (OS) and Disease-Free Survival (DFS) in a large patient population.
Methods
Five-year OS and DFS based on preoperative serum CEA and CYFRA 21-1 levels and the TMI were evaluated in 454 patients (252 men and 202 women) with Stage I NSCLC (median age, 67 [range: 35-86] years) who underwent complete surgical resection. Univariate and multivariate analyses of serum CEA and CYFRA 21-1 levels, and the TMI were performed.
Results
The 5-year OS and DFS rates were significantly lower for patients with elevated serum CEA/CYFRA 21-1 levels than for patients with normal CEA/CYFRA 21-1 levels. The TMI was also markedly different, demonstrating significantly lower OS and DFS in patients with a TMI of >0.5 (P<0.001). Eighty-four patients with normal CEA and CYFRA 21-1 levels had an elevated TMI, which correlated with significantly lower OS and DFS. Multivariate analysis indicated the independent prognostic value of the TMI.
Conclusion
The TMI is an independent preoperative prognostic factor in Stage I NSCLC and should be considered in the decision making for postoperative therapy.
Keywords
INTRODUCTION
Oncofetal glycoprotein CEA is overexpressed in approximately 35-60% of patients with NSCLC [9]. A number of studies demonstrates that elevated preoperative serum CEA concentrations predict a poor prognosis in early-stage NSCLC[10-13], while others report no prognostic value for Overall Survival (OS)[14-18], but rather associated with tumor histology, smoking [14,15], or postoperative disease recurrence[16-18].
Cytokeratin 19 is expressed in the epithelial lining of the bronchial tree and is overexpressed in lung tumors [19]. The C-terminal fragment of cytokeratin 19 is detected by two specific monoclonal antibodies (Bm 19.21 and Ks 19.1). Elevated serum CYFRA 21-1 was found in lung cancer patients [20,21]. Serum CYFRA 21-1 levels are independent of sex and smoking history[22]. Several reports have demonstrated the independent prognostic value of CYFRA 21-1 for survival and tumor relapse after resection[23-26].
Studies that evaluated the prognostic significance of CEA and CYFRA 21-1in the same patient population with stage I-II NSCLC suggested that only CYFRA 21-1, and not CEA, was an independent prognostic indicator for survival [27] and metastasis[28], while others[29,30], showed that both markers have independent prognostic value.It was also noted that prognosis was dependent on tumor histology: patients with adenocarcinoma, but not squamous cell carcinoma had elevated preoperative CEA associated with shorter survival and early recurrence.In contrast, poor prognosis in squamous cell carcinoma was associated with elevated CYFRA 21-1[31]. Higher serum CEA concentrations in adenocarcinoma patients made it difficult to estimate the CEA cutoff value [32,33].
The combination of tumor markers significantly improves their prognostic ability. Cedrés et al. showed that patients with Stage III–IV NSCLC with elevated three markers, CEA, CYFRA 21-1, and cancer antigen-125,had the poorest prognosis[33]. A similar result was obtained using a combination of CEA, CYFRA 21-1, and neuron-specific enolase [34]. A combination of CYFRA 21-1 and a laminin chain gamma 2, predicted a poor OS better than any other markers associated with NSCLC[35]. The CEA and CYFRA 21-1 combination is also reported to have higher sensitivity and specificity for the clinical diagnosis of NSCLC [36-38]. CEA and CYFRA 21-1 gene expression signature was also predictive of a favorable outcome [39].
Muley et al. have shown that a geometric mean of the preoperative CEA and CYFRA 21-1 levels normalized to their respective diagnostic cutoff values, called tumor marker index (TMI), has a prognostic significance[40].The same authors [41] and subsequently Tomita et al. [42] later confirmed these findings, while Blankenburg et al. [43] found no association between elevated preoperative TMI and a poor postoperative prognosis.
To resolve this controversy, this study investigates the usefulness of the TMI in a large patient population with equal number of men and women. We demonstrate that TMI is an independent prognostic factor for postoperative 5-year OS and disease-free survival (DFS) in patients with Stage I NSCLC.
MATERIALS AND METHODS
Subjects
This retrospective study included 454 patients with Stage I NSCLC who underwent complete surgical resection, consisting of a lobectomy and lobe-specific selective lymph node dissection, at Tokyo Medical University Hospital between January 2006 and December 2009. Lymph node dissection was performed in all cases. The cohort comprised 252 men and 202 women, with a median age of 67 (range: 35-86) years. Patients were followed up for ≥5 years or until death. The overall postoperative follow-up period ranged from 46 to 2,955 days (1.5-97.2 months), with a mean of 2,004 days or 65.9 months.
Tumor size and histology
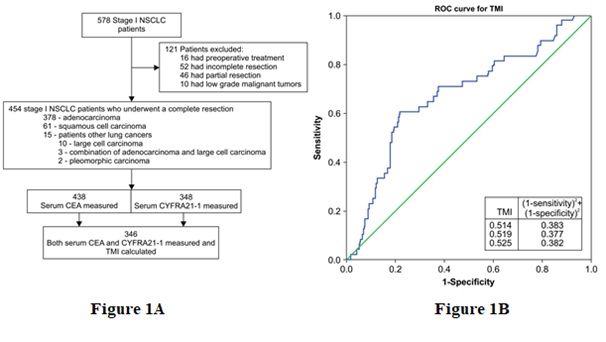 Figure 1: (A) Flow chart of patient recruitment and (B) Receiver Operating Characteristic (ROC) curve for determining the Tumor Marker Index (TMI) cutoff value.Abbreviations: CEA=carcinoembryonic antigen; CYFRA 21-1=cytokeratin-19 fragment; NSCLC=non-small cell lung cancer.
Figure 1: (A) Flow chart of patient recruitment and (B) Receiver Operating Characteristic (ROC) curve for determining the Tumor Marker Index (TMI) cutoff value.Abbreviations: CEA=carcinoembryonic antigen; CYFRA 21-1=cytokeratin-19 fragment; NSCLC=non-small cell lung cancer.Carcinoembryonic antigen and cytokeratin-19 fragment measurements
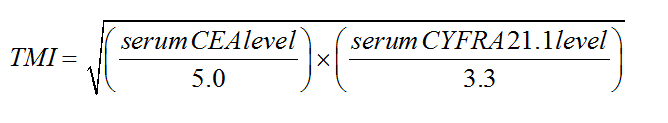
Statistical analyses
Postoperative OS and DFS curves were plotted according to the Kaplan-Meier method. OS was measured from the date of surgery to the date of death or last follow-up. A log-rank test was performed to evaluate the significance of differences in survival rates among the groups. A P
RESULTS
Patient description and marker levels
Preoperative serum CEA levels ranged from 0.2 to 137.0 ng/mL (median: 1.5 ng/mL); 23.1% of patients had elevated CEA levels of >5.0 ng/mL. CYFRA 21-1 levels ranged from 0.1 to 212.0 ng/mL (median: 1.0 ng/mL); 6.6% of patients had elevated CYFRA 21-1 levels of >3.3 ng/mL. In 346 patients, both markers had been measured, and the TMI was calculated. TMI values ranged from 0.09 to 3.87 (median: 0.50); 50.3% of patients had an elevated TMI value of >0.5. Among patients who had normal serum CEA and CYFRA 21-1 levels, 84 had an elevated TMI value. The CEA, CYFRA 21-1, and TMI data are summarized in Table 1.
|
Characteristic |
Patients (n = 454) |
|
Age (years), n (%) |
|
|
Range |
35–86 |
|
<75 |
353 (77.8) |
|
>75 |
101 (22.2) |
|
Sex, n (%) |
|
|
M |
252 (55.5) |
|
F |
202 (44.5) |
|
Smoking History, n (%) |
|
|
Y |
263 (57.9) |
|
N |
191 (42.1) |
|
Histology, n (%) |
|
|
ADC |
378 (83.3) |
|
SCC |
61 (13.4) |
|
Other |
15 (3.3) |
|
CEA Level (ng/mL), n (%)* |
|
|
Median (range) |
1.5 (0.2–137.0) |
|
<5.0 |
337 (76.9) |
|
>5.0 |
101 (23.1) |
|
CYFRA 21-1 Level (ng/mL), n (%)* |
|
|
Median (range) |
1.0 (1.0–26.0) |
|
<3.3 |
325 (93.4) |
|
>3.3 |
23 (6.6) |
|
TMI, n (%)* |
|
|
Median (range) |
0.50 (0.09–3.87) |
|
<0.5 |
172 (49.7) |
|
>0.5 |
174 (50.3) |
|
Normal CEA and CYFRA 21-1 and TMI >0.5, n (%) |
84 (24.3) |
|
*Four hundred and thirty-eight, 348, and 346 patients evaluated, respectively. |
|
|
Abbreviations: ADC=adenocarcinoma; CEA=carcinoembryonic antigen; CYFRA 21-1=cytokeratin-19 fragment; F=female; M=male; N=no; SCC=squamous cell carcinoma; TMI=tumor marker index; Y=yes. |
|
Survival and univariate and multivariate analyses
The 5-year OS and DFS rates were significantly lower for patients with elevated serum CEA levels than for patients with normal CEA levels (P<0.01 and P<0.001, respectively) (Figure 2A and Table 2). Similarly, OS and DFS rates were significantly lower for patients with elevated serum CYFRA 21-1 levels than for patients with normal CYFRA 21-1 levels (P<0.01 and P<0.05, respectively) (Figure 2B and Table 2). In addition, significantly lower OS and DFS rates were observed for patients with an elevated TMI value than for patients with a normal TMI value (P<0.001 for both OS and DFS) (Figure 2C and Table 2).
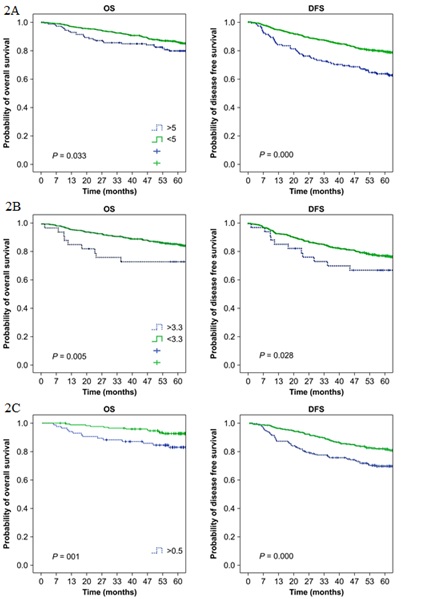 Figure 2: Kaplan-Meier Curves for (OS; Left) and (DFS; Right) According to (A) carcinoembryonic antigen, (B) Cytokeratin-19 fragment, and (C) tumor marker index values. Green curves represent normal values and blue curves represent elevated values of the markers.
Figure 2: Kaplan-Meier Curves for (OS; Left) and (DFS; Right) According to (A) carcinoembryonic antigen, (B) Cytokeratin-19 fragment, and (C) tumor marker index values. Green curves represent normal values and blue curves represent elevated values of the markers.|
Survival |
CEA |
CYFRA 21-1 |
TMI |
||||||
|
Normal |
Elevated |
P |
Normal |
Elevated |
P |
Normal |
Elevated |
P |
|
|
<5.0 ng/mL |
>5.0 ng/mL |
<3.3 ng/mL |
>3.3 ng/mL |
<0.5 |
>0.5 |
||||
|
OS Rate (%) |
89.7 |
85.8 |
.005* |
88.5 |
73.2 |
.005* |
92.7 |
82.8 |
.001* |
|
DFS Rate (%) |
80.8 |
65.7 |
.001* |
78.9 |
64.1 |
.028* |
85.6 |
70.5 |
.001* |
|
*P < .05. |
|||||||||
|
Abbreviations: DFS = disease-free survival; OS = overall survival. |
|||||||||
The clinical variables associated with a significantly poorer survival that were identified by univariate analysis included: male sex (mean survival: 1,785 days in men vs. 1,940 days in women; P<0.01), smoking history (1,771 days in smokers vs. 1,972 days in non-smokers; P<0.01), tumor size (1,651 days for tumors >3.0 cm vs. 1,978 days for tumors <3.0 cm; P<0.001), and tumor histology (1,353 days for non-adenocarcinoma vs. 1,794 days for adenocarcinoma; P<0.0001) (Table 3 and Table 4).
|
Risk Factor |
Univariate Analysis |
Multivariate Analysis |
||
|
HR (95.0% CI) |
P |
HR (95.0% CI) |
P |
|
|
Age (>75 Years) |
1.673 (0.963–2.905) |
0.068 |
– |
– |
|
Sex (M) |
2.064 (1.190–3.579) |
0.010* |
– |
– |
|
Smoking Status (Smoker vs. Ex-Smoker) |
2.928 (1.586–5.406) |
0.010* |
– |
– |
|
Tumor Size (>3.0 cm) |
2.434 (1.458–4.064) |
0.001* |
– |
– |
|
Histology (Non-ADC) |
4.242 (2.525–7.124) |
<0.001* |
3.916 (2.168-7.076) |
<0.001* |
|
CEA Level (>5.0 ng/mL) |
1.807 (1.042–3.135) |
0.035* |
– |
– |
|
CYFRA 21-1 Level (>3.3 ng/mL) |
2.968 (1.328–6.635) |
0.008* |
– |
– |
|
TMI (>0.5) |
2.699 (1.447–5.033) |
0.002* |
1.996 (1.031-3.752) |
0.040* |
|
*P < .05 |
||||
|
Abbreviations: ADC = adenocarcinoma; CEA = carcinoembryonic antigen; CYFRA 21-1 = cytokeratin-19 fragment; CI = confidence interval; HR = hazard ratio; M = male; TMI = tumor marker index. |
||||
|
Risk Factor |
Univariate Analysis |
Multivariate Analysis |
||
|
HR (95.0% CI) |
P |
HR (95.0% CI) |
P |
|
|
Age (>75 Years) |
1.483 (0.965–2.277) |
0.072 |
– |
– |
|
Sex (M) |
1.705 (1.135–2.561) |
0.010* |
– |
– |
|
Smoking Status (Smoker vs. Ex-Smoker) |
1.863 (1.454–2.385) |
<0.010* |
– |
– |
|
Tumor Size (>3.0 cm) |
2.190 (1.731–2.771) |
<0.001* |
1.568 (1.130–2.176) |
0.007* |
|
Histology (Non-ADC) |
2.071 (1.747–2.455) |
<.001* |
1.882 (1.466–2.416) |
<0.001* |
|
CEA Level (>5.0 ng/mL) |
1.948 (1.471–2.579) |
<0.001* |
– |
– |
|
CYFRA 21-1 Level (>3.3 ng/mL) |
1.897 (1.152–3.066) |
0.012* |
– |
– |
|
TMI (>0.5) |
1.989 (1.461–2.709) |
<0.001* |
1.563 (1.125–2.172) |
0.008* |
|
*P < .05 |
||||
|
Abbreviations: ADC = adenocarcinoma; CEA = carcinoembryonic antigen; CYFRA 21-1 = cytokeratin-19 fragment; CI = confidence interval; HR = hazard ratio; M = male; TMI = tumor marker index |
||||
Univariate analysis of preoperative tumor markers confirmed their significance for OS (Table 3). The mean survival of patients with serum CEA levels of >5.0 ng/mL was 1,454 days compared to 1,869 days for patients with serum CEA levels of <5.0 ng/mL (P<0.05). The mean survival of patients with serum CYFRA 21-1 levels of >3.3 ng/mL was 1,472 days compared to 1,845 days for patients with serum CYFRA 21-1 levels of <3.3 ng/mL (P<0.01). Patients in whom the calculated TMI value was >0.5 had a lower mean survival than patients with a TMI value of <0.5 (1,702 vs. 1,944 days, respectively; P<0.01).
Multivariate analysis identified the TMI as being an independent prognostic factor not only for OS (hazard ratio: 1.966, 95.0% confidence interval: 1.031–3.752; P<0.05), but also for DFS (hazard ratio: 1.563, 95.0% confidence interval: 1.125-2.172; P<0.01) (Table 4). According to the multivariate analysis, tumor size was an independent prognostic factor for DFS only and not OS.
When survival analysis was performed on patients (n=84) who had normal preoperative serum CEA and CYFRA 21-1 levels, but an elevated TMI value, significant differences in the OS and DFS rates were found compared to patients who had all three parameters that were normal (Figure 3A). The 5-year OS rate was 82.8% in patients with a TMI value of >0.5 and 92.6% in patients with a TMI value of <0.5 (P<0.05). The 5-year DFS rate was 70.9% in patients with a TMI value of >0.5 and 86.0% in patients with a TMI value of <0.5 (P<0.01) (Figure 3B).
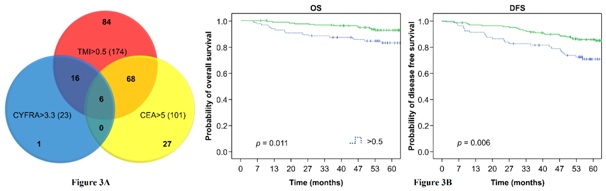 Figure 3: (A) Patients (84) with (CEA) and (CYFRA 21-1) and elevated Tumor Marker Index (TMI). (B) Overall Survival (OS; Left) and Disease-Free Survival (DFS; Right) for the above 84 patients. Green curves represent normal values and blue curves represent elevated values.
Figure 3: (A) Patients (84) with (CEA) and (CYFRA 21-1) and elevated Tumor Marker Index (TMI). (B) Overall Survival (OS; Left) and Disease-Free Survival (DFS; Right) for the above 84 patients. Green curves represent normal values and blue curves represent elevated values.The DFS and OS are significantly different from the patient who had normal preoperative serum CEA and CYFRA211 levels but an elevated TMI (mean DFS and OS are 16111 days and 1737days), the patients with low three indicators and the patient with elevated CEA or CYFRA211 and TMI (mean DFS and OS are 1383 days and 1461 days).
DISCUSSION
In accordance with several previous reports [5,6] our results demonstrate that male sex, smoking history, tumor size and histology are independent prognostic factors in NSCLC. We validated the significance of the TMI for the prediction of OS in a much larger cohort than those of previous studies[40-43]. The number of patients included in the study was 454 compared to 153, 261, and 293 patients in previous studies. Furthermore, our patient population consisted of an almost equal number of men and women (252 vs. 202). This is important given the sex differences in survival in patients with NSCLC[46].
All patients included in this study underwent lobe-specific selective lymph node dissection (known as ND2a-1 in Japan), which results in high OS[47]. In our study5-year survival rates were approximately 80.0%. Moreover, the proportion of patients with large cell carcinoma, who usually have a poor prognosis, was relatively low in our study (<20.0%). Despite high survival rates, we were able to demonstrate the effectiveness of the TMI in predicting a poor prognosis. In a previous study, Blankenburg et al. reported that the survival rates in 40.0% of patients with large cell carcinoma were <55.0%. The low OS rates may explain their inability to demonstrate the prognostic significance of the TMI[43].
To further investigate the prognostic value of the TMI, we plotted Kaplan-Meier curves of DFS according to the TMI (Figure 2C and Table 2) and performed univariate and multivariate analyses (Table 4). Our data demonstrated that the TMI is an independent prognostic indicator not only for OS but also for DFS, suggesting that it could be utilized for the prediction of potential disease recurrence, and thus could contribute to the decision making for postoperative chemotherapy.
Our study for the first time demonstrates advantage of TMI compared serum CEA and CYFRA 21-1 levels separately. We show that individual CEA and CYFRA 21-1 measurements often produce non-overlapping results in the same patient population. For example, among 101 patients with elevated serum CEA levels and 23 patients with elevated CYFRA 21-1 levels, the levels of both markers were simultaneously elevated in only 6 patients. Similarly, among 325 patients with normal CYFRA 21-1 levels, 95 patients had elevated serum CEA levels. Furthermore, we found that among 174 patients with an elevated TMI value, 84 had normal CEA and CYFRA 21-1 levels (Figure 3A).
We standardized the prognostic threshold of the TMI as 0.5 using ROC analysis. Previous studies [40-43], used a critical level approach that resulted in various TMI cutoff values for the prediction of survival, complicating the comparison of results between different reports. For instance, Blankenburg et al. defined several prognostic cutoff values for the TMI based on survival curves[43]. They also used a different diagnostic threshold for CEA assay, which may explain the discrepancy between their results and our own.
In accordance with our findings, the TMI has been shown to correlate with imaging characteristics of high-resolution computed tomography, predicting a poor prognosis in early-stage lung adenocarcinoma[47]. Elevated TMI also correlated with a poor survival in aggressive adenosquamous lung carcinoma with a relatively high frequency of EGFR mutations[48].
The TMI has also been calculated for different tumor markers, e.g. for preoperative CEA and Krebs von den Lungen-6 levels in NSCLC[49]. In general, the utilization of a combination of several markers appears to be a more advantageous approach for the diagnosis and assessment of chemotherapy response in lung cancer and other cancer types[50]. For example, a comparison of the diagnostic sensitivity of various combinations of serum tumor markers, including CEA and CYFRA 21-1, in patients with esophageal squamous cell carcinoma suggested that combinations of markers that included Chitinase-3-like-1 protein and squamous cell carcinoma antigen had better diagnostic capabilities than CEA and CYFRA 21-1 for this type of cancer[51]. The usefulness of a prognostic marker index that is calculated as a combination of preoperative serum CEA and plasma tissue inhibitor of metalloproteinases-1 levels for the assessment of the risk of disease recurrence has been demonstrated in patients with early-stage colorectal cancer[52].
Several limitations of our study should be considered. First, false-positive results associated with elevated CEA levels due to smoking could not be excluded[53]. Moreover, this retrospective study did not include the findings of a preoperative clinical examination of the patients, which could influence serum CEA levels. Second, variations in the rates of CEA and CYFRA 21-1 synthesis and elimination in highly heterogeneous lung tumors were not taken into consideration, and third, tumor depth and nodal status were not examined.
CONCLUSION
DISCLOSURE STATEMENT
SOURCE(S) OF SUPPORT
ACKNOWLEDGEMENTS
REFERENCES
- Sagawa M, Nakayama T, Tanaka M, Sakuma T, JECS Study Group, et al. (2012) A randomized controlled trial on the efficacy of thoracic CT screening for lung cancer in non-smokers and smokers of <30 pack-years aged 50-64 years (JECS study): research design. Jpn J Clin Oncol 42: 1219-1221.
- Midthun DE (2016) Early detection of lung cancer. F1000Res 5: 739.
- Saisho S, Yasuda K, Maeda A, Yukawa T, Okita R, et al. (2013) Post-recurrence survival of patients with non-small-cell lung cancer after curative resection with or without induction/adjuvant chemotherapy. Interact Cardiovasc Thorac Surg 16: 166-172.
- Cho S, Song IH, Yang HC, Kim K, Jheon S (2013) Predictive factors for node metastasis in patients with clinical stage I non-small cell lung cancer. Ann Thorac Surg 96: 239-245.
- Holdenrieder S, Pagliaro L, Morgenstern D, Farshid D (2016) Clinically meaningful use of blood tumor markers in oncology. Biomed Res Int 2016: 9795269.
- Wallerek S, Sørensen JB (2015) Biomarkers for efficacy of adjuvant chemotherapy following complete resection in NSCLC stages I-IIIA. Eur Respir Rev 24: 340-355.
- Grunnet M, Sorensen JB (2012) Carcinoembryonic antigen (CEA) as tumor marker in lung cancer. Lung Cancer 76: 138-143.
- Matsuguma H, Nakahara R, Igarashi S, Ishikawa Y, Suzuki H, et al. (2008) Pathologic stage I non-small cell lung cancer with high levels of preoperative serum carcinoembryonic antigen: clinicopathologic characteristics and prognosis. J Thorac Cardiovasc Surg 135: 44-49.
- Kawachi R, Nakazato Y, Takei H, Koshi-ishi Y, Goya T (2009) Clinical significance of preoperative carcinoembryonic antigen level for clinical stage I non-small cell lung cancer: can preoperative carcinoembryonic antigen level predict pathological stage? Interact Cardiovasc Thorac Surg 9: 199-202.
- Ozeki N, Fukui T, Taniguchi T, Usami N, Kawaguchi K, et al. (2014) Significance of the serum carcinoembryonic antigen level during the follow-up of patients with completely resected non-small-cell lung cancer. Eur J Cardiothorac Surg 45: 687-692.
- Wang XB, Li J, Han Y (2014) Prognostic significance of preoperative serum carcinoembryonic antigen in non-small cell lung cancer: a meta-analysis. Tumour Biol 35: 10105-10110.
- Tomita M, Matsuzaki Y, Edagawa M, Shimizu T, Hara M, et al. (2004) Prognostic significance of preoperative serum carcinoembryonic antigen level in lung adenocarcinoma but not squamous cell carcinoma. Ann Thorac Cardiovasc Surg 10: 76-80.
- Kato T, Ishikawa K, Aragaki M , Sato M, Okamoto K, et al. (2013) Optimal predictive value of preoperative serum carcinoembryonic antigen for surgical outcomes in stage I non-small cell lung cancer: differences according to histology and smoking status. J Surg Oncol 107: 619-624.
- Gaspar MJ, Diez M, Rodriguez A, Ratia T, Martin Duce A, et al. (2003) Clinical value of CEA and CA125 regarding relapse and metastasis in resectable non-small cell lung cancer. Anticancer Res 23: 3427-3432.
- Liu H, Gu X, Lv T, Wu Y, Xiao Y, et al. (2014) The role of serum carcinoembryonic antigen in predicting responses to chemotherapy and survival in patients with non-small cell lung cancer. J Cancer Res Ther 10: 239-243.
- Maeda R, Suda T, Hachimaru A, Tochii D, Tochii S, et al. (2017) Clinical significance of preoperative carcinoembryonic antigen level in patients with clinical stage IA non-small cell lung cancer. J Thorac Dis 9: 176-186.
- Kosacka M, Jankowska R (2009) Comparison of cytokeratin 19 expression in tumor tissue and serum CYFRA 21-1 levels in non-small cell lung cancer. Pol Arch Med Wewn 119: 33-37.
- Wieskopf B, Demangeat C, Purohit A, Stenger R, Gries P, et al. (1995) Cyfra 21-1 as a biologic marker of non-small cell lung cancer. Evaluation of sensitivity, specificity, and prognostic role. Chest 108: 163-169.
- Pujol JL, Grenier J, Daurès JP, Daver A, Pujol H, et al. (1993) Serum fragment of cytokeratin subunit 19 measured by CYFRA 21-1 immunoradiometric assay as a marker of lung cancer. Cancer Res 53: 61-66.
- Kao CH, Hsieh JF, Ho YJ, Tsai SC, Lee JK (1999)Cytokeratin fragment 19 (CYFRA 21-1) in healthy smokers. Anticancer Res19: 4545-4546.
- Takahashi H, Kurishima K, Ishikawa H, Kagohashi K, Kawaguchi M, et al. (2010) Optimal cutoff points of CYFRA21-1 for survival prediction in non-small cell lung cancer patients based on running statistical analysis. Anticancer Res 30: 3833-3837.
- Zhao H, Shi X, Liu J, Chen Z, Wang G (2014) Serum cyfra21-1 as a biomarker in patients with non-small cell lung cancer. J Cancer Res Ther 10: 215-217.
- Xu Y, Xu L, Qiu M, Wang J, Zhou Q, et al. (2015) Prognostic value of serum cytokeratin 19 fragments (Cyfra 21-1) in patients with non-small cell lung cancer. Sci Rep 5: 9444.
- Yu Z, Zhang G, Yang M, Zhang S, Zhao B, et al. (2017) Systematic review of CYFRA 21-1 as a prognostic indicator and its predictive correlation with clinicopathological features in Non-small Cell Lung Cancer: A meta-analysis. Oncotarget 8: 4043-4050.
- Hanagiri T, Sugaya M, Takenaka M, Reinmiedl J, Gruber C, et al. (2011) Preoperative CYFRA 21-1 and CEA as prognostic factors in patients with stage I non-small cell lung cancer. Lung Cancer 74: 112-117.
- Zhang L, Liu D, Li L, Pu D, Zhou P, et al. (2017) The important role of circulating CYFRA21-1 in metastasis diagnosis and prognostic value compared with carcinoembryonic antigen and neuron-specific enolase in lung cancer patients. BMC Cancer 17: 96.
- Duan X, Cui Y, Li H, Shi G, Wu B, et al. (2015) High preoperative and postoperative levels of carcinoembryonic antigen and CYFRA 21-1 indicate poor prognosis in patients with pathological Stage I nonsmall cell lung cancer. Indian J Cancer 52: 158-163.
- Zhang ZH, Han YW, Liang H, Wang LM (2015) Prognostic value of serum CYFRA21-1 and CEA for non-small-cell lung cancer. Cancer Med 4: 1633-1638.
- Matsuoka K, Sumitomo S, Nakashima N, Nakajima D, Misaki N (2007) Prognostic value of carcinoembryonic antigen and CYFRA21-1 in patients with pathological stage I non-small cell lung cancer. Eur J Cardiothorac Surg 32: 435-439.
- Szturmowicz M, Rudzi?ski P, Kacprzak A, Langfort R, Bestry I, et al. (2014) Prognostic value of serum C-reactive protein (CRP) and cytokeratin 19 fragments (Cyfra 21-1) but not carcinoembryonic antigen (CEA) in surgically treated patients with non-small cell lung cancer. Pneumonol Alergol Pol 82: 422-429.
- Cedrés S, Nuñez I, Longo M, Martinez P, Checa E, et al. (2011) Serum tumor markers CEA, CYFRA21-1, and CA-125 are associated with worse prognosis in advanced non-small-cell lung cancer (NSCLC). Clin Lung Cancer 12: 172-179.
- Barlési F, Gimenez C, Torre JP, Doddoli C, Mancini J, et al. (2004) Prognostic value of combination of Cyfra 21-1, CEA and NSE in patients with advanced non-small cell lung cancer. Respir Med 98: 357-362.
- Korbakis D, Dimitromanolakis A, Prassas I, Davis G J, Barber E, et al. (2015) Serum LAMC2 enhances the prognostic value of a multi-parametric panel in non-small cell lung cancer. Br J Cancer 113: 484-491.
- Muley T, Dienemann H, Ebert W (2003) Increased CYFRA 21-1 and CEA levels are negative predictors of outcome in p-stage I NSCLC. Anticancer Res 23: 4085-4093.
- Okamura K, Takayama K, Izumi M, Harada T, Furuyama K, et al. (2013) Diagnostic value of CEA and CYFRA 21-1 tumor markers in primary lung cancer. Lung Cancer 80: 45-49.
- Ma L, Xie XW, Wang HY, Ma LY, Wen ZG (2015) Clinical evaluation of tumor markers for diagnosis in patients with non-small cell lung cancer in China. Asian Pac J Cancer Prev 16: 4891-4894.
- McKeegan EM, Ansell PJ, Davis G, Chan S, Chandran RK, et al. (2015) Plasma biomarker signature associated with improved survival in advanced non-small cell lung cancer patients on linifanib. Lung Cancer 90: 296-301.
- Muley T, Dienemann H, Ebert W (2008) CYFRA 21-1 and CEA are independent prognostic factors in 153 operated stage I NSCLC patients. Anticancer Res 24: 1953-1956.
- Muley T, Fetz TH, Dienemann H, Hoffmann H, Herth FJ, et al. (2008) Tumor volume and tumor marker index based on CYFRA 21-1 and CEA are strong prognostic factors in operated early stage NSCLC. Lung Cancer 60: 408-415.
- Tomita M, Shimizu T, Ayabe T, Yonei A, Onitsuka T (2010) Prognostic significance of tumour marker index based on preoperative CEA and CYFRA 21-1 in non-small cell lung cancer. Anticancer Res 30: 3099-3102.
- Blankenburg F, Hatz R, Nagel D, Ankerst D, Reinmiedl J, et al. (2008) Preoperative CYFRA 21-1 and CEA as prognostic factors in patients with stage I non-small cell lung cancer: external validation of a prognostic score. Tumour Biol 29: 272-277.
- Lemjabbar-Alaoui H, Hassan OU, Yang YW, Buchanan P (2015) Lung cancer: Biology and treatment options. Biochim Biophys Acta 1856: 189-210.
- Sangha R, Price J, Butts CA (2010) Adjuvant therapy in non-small cell lung cancer: current and future directions. Oncologist 15: 862-872.
- Sakurai H, Asamura H, Goya T, Eguchi K, Nakanishi Y, et al. (2010) Survival differences by gender for resected non-small cell lung cancer: a retrospective analysis of 12,509 cases in a Japanese Lung Cancer Registry study. J Thorac Oncol 5: 1594-1601.
- Xu Y, Sun H, Zhang Z, Song A, Wang W, et al. (2016) Assessment of relationship between CT features and serum tumor marker index in early-stage lung adenocarcinoma. Acad Radiol 23: 1342-1348.
- Zhi Q, Wang Y, Wang X, Yue D, Li K, et al. (2016) Predictive and prognostic value of preoperative serum tumor markers in resectable adenosqamous lung carcinoma. Oncotarget 7: 64798-64809.
- Tomita M, Ayabe T, Chosa E, Nose N, Nakamura K (2017) Prognostic significance of a tumor marker index based on preoperative serum carcinoembryonic antigen and krebs von den lungen-6 levels in non-small cell lung cancer. Asian Pac J Cancer Prev 18: 287-291.
- Ma R, Xu H, Wu J, Bai S, Dun B, et al. (2017) Identification of serum proteins and multivariate models for diagnosis and therapeutic monitoring of lung cancer. Oncotarget 8: 18901-18913.
- Zheng X, Xing S, Liu XM, Liu W, Liu D, et al. (2014) Establishment of using serum YKL-40 and SCCA in combination for the diagnosis of patients with esophageal squamous cell carcinoma. BMC Cancer 14: 490.
- Nielsen HJ, Christensen IJ, Brünner N (2010) A novel prognostic index in colorectal cancer defined by serum carcinoembryonic antigen and plasma tissue inhibitor of metalloproteinases-1. Scand J Gastroenterol 45: 200-207.
- Tomita M, Ayabe T, Chosa E, Nakamura K (2015) Impact of smoking on outcome of resected lung adenocarcinoma. Gen Thorac Cardiovasc Surg 63: 608-612.
- Myerson RJ, Singh A, Birnbaum EH, Fry RD, Fleshman JW, et al. (2001) Pretreatment clinical findings predict outcome for patients receiving preoperative radiation for rectal cancer. Int J Radiat Oncol Biol Phys 50: 665-674.
- Li F, Shen Z, Lu Y, Wang L, Song W (2014) How much does body mass index affect serum carcinoembryonic antigen concentration? Cancer Epidemiol Biomarkers Prev 23: 555-560.
Citation: Yamaguchi G, Haghiwara M, IkedaN (2019) Prognostic Value of the Tumor Marker Index Based on Preoperative Serum Carcinoembryonic Antigen and Cytokeratin-19 Fragment Levels for Overall and Disease-Free Survival in Patients with Stage I Non-Small Cell Lung Cancer. J Pulm Med Respir Res 5: 027.
Copyright: © 2019 Gaku Yamaguchi, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.

